Abstract
Maternal HIV-1 status and antiretroviral drug exposure may influence the haematological profiles of infants. We recruited infants from 118 uninfected control women and from 483 HIV-1 infected women who received no antiretroviral drugs (n=28), or received single-dose Nevirapine (sdNVP) (n=424) or triple-drug combination therapy (n=31) to reduce HIV-1 transmission. Blood was drawn from infants within 24 hours of delivery or 6-12 weeks post-delivery and full blood counts performed using a fully automated AcT-5-diff haematology analyser and reference controls. Exposed uninfected (EU; no NVP) differed from control infants only in having lower basophil counts and percentages. In all infant groups, leukocyte profiles showed characteristic quantitative changes with age in the first 6 weeks of life. HIV-1 infected infants displayed by 6 weeks elevations in white blood cells, lymphocyte, monocyte and basophil counts, and monocyte and basophil percentages, when compared to EU infants. At birth EU NVP-treated infants exhibited elevated monocyte percentages and counts and basophil counts that did not persist at 6 weeks. Interestingly, EU newborns of mothers with high CD4 counts (> 500 cells/μl) that had taken sdNVP had significantly elevated white blood cell, monocyte and basophil counts when compared to newborn infants of mothers with similar CD4 counts that had not taken sdNVP; this was not evident in infants of mothers with CD4 counts <200 cells/μl. These previously undescribed features may affect immune response capability in early life and clinical consequences of such changes need to be further investigated.
Keywords: Infant haematological profiles, maternal HIV-1 status, single-dose NVP.
INTRODUCTION
Immune development in neonatal life is unique, being shaped by the challenges at the maternal-foetal interface [1] and at birth by the transition to the environment outside of the uterus which is rich in foreign antigens. Issues relating to the effect the maternal immune environment (HIV-1 exposure and the associated immune dysregulation) has on the ontogenic development of an infant’s immune system have culminated in numerous studies that have highlighted the fact that there are consequences of in utero HIV-1 exposure and of the antiretroviral prophylaxis given to prevent mother-to-child transmission (MTCT) of HIV-1 on immune parameters of the newborn. These include alterations in haematopoiesis, haematological parameters, T-cell maturation, immunological reactivity, and imbalances in cell populations [2-12]. Our study, which showed in utero HIV-1 and pre-birth exposure to single-dose Nevirapine (sdNVP) to significantly increase immune activation in infants [13] suggests that the altered maternal immune environment, through HIV-1 infection and antiretroviral drugs given peripartum, influences the immune system of her unborn infant. A murine study has also shown that maternal T-helper type 1 responses induced during gestation can modulate cell phenotypes and cytokine response capabilities in the offspring [14]. The ability of some neonates to mount robust HIV-1 specific immune responses [15] suggests that at birth infant immune systems are sufficiently developed having the necessary immunocompetent cells and signalling mechanisms in place to provide the immune components necessary for developing responses that are protective from maternal HIV-1 infection.
The increased use of antiretroviral drugs to prevent MTCT of HIV-1 is associated with the added risk of haematological toxicity in the mother and infant. Concerns about the short- and long-term haematological toxicities caused by transmission prophylaxis have prompted several studies that address issues of safety and possible side effects. While results from some short-term studies are reassuring [16] some studies suggest effects on haematopoiesis can last up to 18 months following perinatal exposure [4, 17]. Results from other studies have raised concerns over the longer term adverse effects of neonatal exposure to antiretroviral drugs [6, 18, 19].
Important factors such as antigen dose, microenvironment (which includes exposure to antiretrovirals) and timing and extent of antigen exposure will likely influence the outcome of the infant’s immune response to subsequent antigenic challenge as well as responses to vaccines. During pregnancy, innate and adaptive immune responses are altered [20], and further dysregulated as a consequence of HIV-1 infection. The aim of this study was to establish if blood cell counts and differentials were altered in infants in early life as a consequence of maternal immune status (CD4 count and viral load) and antiretroviral drug exposure (particularly maternal sdNVP). Given our interest in infant cellular immune response capability in early life, we focussed on alterations of white blood cell (WBC) counts and differentials (neutrophils, lymphocytes, monocytes, eosinophils, basophils; expressed as either an absolute value (counts: percentage x total WBC) or as a percentage (differential: relative number of each type of WBC in relation to the total WBC)). To the best of our knowledge this is the first reported study showing that these parameters are affected by maternal sdNVP treatment as well as HIV-1 status, especially maternal CD4 counts.
MATERIALS AND METHODS
Study Population
Infants were recruited from mothers that formed part of the HIV-1 Protective Immunity and Perinatal Exposure Study. Participants included infants born to uninfected (control) and HIV-1 infected mothers, recruited at Chris Hani Baragwanath Hospital (CHBH), Soweto, South Africa and from Coronation Women and Children Hospital (CWCH), Johannesburg, South Africa. Women at CHBH were enrolled at delivery and blood was collected from their infants within 48 hours following delivery. Infants of women identified as part of the postpartum voluntary counselling and testing program were given sdNVP as post-exposure prophylaxis (PEP) and mothers were counselled about infant feeding options. These were women that were identified at the maternity ward as having delivered live-born infants but who had not been tested for HIV-1 at CHBH prior to delivering their infants; NVP was administered as PEP to infants of women that consented and that were identified as being positive. The study subjects who were identified as HIV-1 positive during pregnancy and who participated in routine prevention of MTCT services received sdNVP before delivery. All mothers from CWCH were identified at 6 weeks post-partum when they returned for postnatal follow-up services. These mothers received either sdNVP or combination antiretroviral therapy (ART) (lopinavir/ritonavir, stavudine (d4T), lamivudine (3TC) or efavirenz, d4T, 3TC) if identified with low CD4 counts early enough during pregnancy (combination therapy would have been initiated at any stage of pregnancy but adherence in these women may not have been complete however, NVP or other ART intervention would have been taken prior to delivery). A summary of the maternal clinical characteristics and the antiretroviral regimens given to the mothers to reduce MTCT of HIV-1 are presented in Table 1. Peripheral blood was again collected at 6 weeks (for the CHBH enrolees, CWHC participants were only recruited at 6 weeks) and 12 weeks if possible. Blood was collected from 119 uninfected children (twins born to one of the mothers) and these served as controls. Blood was processed within 12 hours of collection. Written informed consent was obtained from the participants and ethical approval was obtained from the Committee for Research on Human Subjects of the University of the Witwatersrand.
Table 1.
Cohort Summary of the Mothers of Infants Recruited at Chris Hani Baragwanath Hospital, Soweto, South Africa and from Coronation Women and Children Hospital, Johannesburg, South Africa
| ART Regimen | Controls | HIV-1 Infected Mothers | ||
|---|---|---|---|---|
| No Drug | Monotherapy (NVP Only) | Triple Therapy3 | ||
| N | 1181 | 28 | 424 | 31 |
| Mean (Standard Deviation) (n/N) | ||||
| Maternal CD4 +T-cell count | 991 (344) (6/20)2 | 575 (228) (27/28) | 482 (261) (422/424) | 221 (154) (31/31) |
| Maternal age (y) | 25 (6.4) (20/20 )2 | 28 (5.8) (27/28) | 28 (5.2) (422/424) | 29 (5.5) (31/31) |
| Infant birth weight (g) | 3002 (461) (20/20)2 | 2815 (503) (28/28) | 3049 (451) (422/424) | 3004 (504) (31/31) |
| Median (IQR) (n/N) | ||||
| Maternal viral load (log10) | uninfected | 3.9 (2.8-4.4) (28/28) | 3.98 (3.3-4.7) (419/424) | 2.6 (2.6-2.8) (31/31) |
| % (n/N) | ||||
| Primiparity | 45 (9/20)2 | 21 (6/28) | 30 (45/424) | 100 (31/31) |
| Infant sex (male) | 58 (11/20)2 | 39 (11/28) | 49 (208/424) | 45 (14/31) |
| Preterm (<37 wk) | 15 (3/20) 2 | 15 (4/28) | 4 (15/424) | 3 (1/31) |
| Vaginal delivery | 100 (20/20)2 | 86 (24/28) | 88 (370/424) | 77 (24/31) |
N: group size – no distinction is made between sampling time points i.e. birth, 6 weeks or 12 weeks or the hospital sites.
IQR: interquartile range (25th and 75th percentiles).
n/N: number of cases tested from the group.
One of the control mothers had twins i.e. infant number = 119.
Limited clinical data available on the control patients.
Mothers given triple-drugs (lopinavir/ritonavir, stavudine (d4T), lamivudine (3TC) or efavirenz, d4T, 3TC) were only recruited at 6 weeks or 12 weeks.
Note: South African PMTCT guidelines have been criticised for commencing comprehensive management for all pregnant women with highly active antiretroviral therapy (HAART) in WHO stage IV with CD4 counts ≤200 cells/µl. On November 30th 2009 the World Health Organization announced new recommendations that replace previous guidelines for low and middle-income countries that everyone diagnosed with HIV infection should start treatment when their CD4 count falls below 350 cells/µl.
Sample Processing and Testing
Infants born to mothers at CHBH were prospectively followed up after birth to determine their HIV-1 status until 12 weeks. HIV-1 DNA polymerase chain reaction (PCR) tests were carried out on infant peripheral blood samples collected at 6 weeks (Amplicor, Roche Diagnostic Systems, Inc, Branchburg, New Jersey, USA). If the 6 week PCR was positive, samples collected on the day of birth were tested to establish timing of infection. A positive result at birth was used to infer intrauterine transmission and a negative result at birth but a positive result at 6 weeks or later was used to infer intrapartum transmission. Almost all women avoided breastfeeding and those who initiated some breastfeeding had all stopped by 6 weeks. Children who remained negative at 6 weeks or 12 weeks were considered to be exposed uninfected (EU). Infants born to mothers at CWCH were tested at 12 weeks and if the PCR was positive the 6 week sample was tested. In this group, however, the timing of infection could not be determined because birth samples were not available. Maternal viral load was measured in plasma using the Roche Amplicor RNA Monitor assay (Roche Diagnostic Systems, Inc, New Jersey, USA) with the lower detection limit of <400 HIV-1 RNA copies/ml. CD4 T cell counts were determined using the FACSCount System (Becton Dickinson, San Jose, CA).
A full blood count was performed on the infant blood samples within 12 hours of blood having been drawn from patients, using a fully automated AcT-5-diff haematology analyser (Beckman Coulter Inc, Fullerton, CA) according to the manufacturer’s protocol. Reference controls (low, medium and high; Beckman Coulter Inc, Fullerton, CA) were routinely used to confirm and monitor instrument accuracy and performance by providing measurements for counting, sizing, haemoglobin determination and white blood cell differentiation. Low counts were confirmed with manual differentials. Specimen results were accepted when the control ranges fell within those specified for the reference controls. Specimens whose results fell out of the range of determination were diluted (1:2 or 1:4 in saline) and the dilution factor taken into account when leukocyte counts for these samples were analysed. A sample report was automatically generated for each specimen with the data being presented as a percentage and an absolute number per microlitre. Reference values used for the white blood cell counts (neutrophils, lymphocytes, monocytes, eosinophils and basophils) come from the Department of Haematology, National Health Laboratory Service (NHLS), Chris Hani Baragwanath and Braamfontein Laboratories Service Handbook, 2006-2007 [21]. We refer to these values as DISA reference values. DISA serves as the laboratory information system used by the NHLS. These DISA reference tables represent baseline data applicable to the South African population. Haematological parameters recorded are shown in column 1 of Table 2.
Table 2.
Haematology Profiles of Birth and of 6 Weeks Control Infants and Exposed Uninfected Infants that were Not Exposed to Maternal NVP or that Received sdNVP or Triple Antiretroviral Therapy to Reduce MTCT of HIV-1
| Infant Group | DISA Reference Ranges (Infants Birth)# | DISA Reference Ranges(Infants 1 day - 1 Week)# | Birth | DISA Reference Ranges (Infant 1 - 6 Months) | 6 Weeks | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Haematologic Parameter Median(IQR)(Min; Max) | Control No NVP | EU No NVP | EU NVP | Control No NVP | EU No NVP | EU NVP | EU Triple Therapy | |||
| No. Of Infants | 112 | 16 | 113 | 34 | 20 | 258 | 13 | |||
| Haemoglobin (g/dL) | 18.0-27.0 | 16.0-25.5 | 17 (15.5-18.6) (3.9;22.5) |
16.8 (15.5-18.8) (14.2;19.5) |
17.6 (15.9-19.5) (10.9;23) |
10.0-15.0 | 11.8 (10.5-12.8) (10.1;14) |
10.8 (9.8-11.5) (6.8;13.5) |
11.2 (10.5-12) (2;16.3) |
11.3 (10.7-11.7) (9.5;13.1) |
| MCV (fL) |
109 (Av) |
104 (Av) |
106 (102-109.8) (91;124) |
108.5 (96.5-110.8) (84;112) |
108 (102-111) (82;118) |
78 (Av) |
92 (87-95) (72;103) |
92 (86-93.8) (71;101) |
92 (88-95) (70;190) |
98 (96.5 -100) (87;105) |
| Absolute Counts (/µL) | ||||||||||
| Platelets (x103) |
140-400 | 140-400 | 235.5 (183.8-306.8) (41;467) |
242.5 (139.5-268.3) (19;569) |
237 (178.5-280.5) (13;413) |
140-400 | 427 (317.5-482.5) (177;711) |
429.5 (375-538.8) (147;840) |
420 (334-529) (3.1;805) |
421 (333.5-600) (136;692) |
| RBC (x106) |
4.8-7.1 | 4.5-6.4 | 5.1 (4.5-5.4) (1;6.7) |
4.8 (4.5-5) (3.8;6) |
4.9 (4.4 -5.3) (3;6.8) |
3.9-5.3 | 3.8 (3.5-4.1) (3.1;4.7) |
3.5 (3.1-3.7) (2.1;5.2) |
3.5 (3.2-3.9) (0.7;306) |
3.3 (3.1-3.4) (3-3.9) |
| WBC (x103) |
9.0-30.0 | 5.0-21.0 | 16 (12.9-20.4) (7.7;168) |
13.8 (10.5-18.3) (7.9;20.4) |
15.6 (12.9-19.3) (6.6;33) |
6.0-17.5 | 7.8 (6.3-9.6) (3.1;17.8) |
9.1 (7.4-12.1) (4.4;18.6) |
9.1 (7.3-11.1) (0.1;36.3) |
10.4 (7.6-11) (5.4;15.7) |
| Neutrophils (x103) |
6.0-26.0 | 1.5-10.0 | 9.1 (6.9-12.5) (2.5;20.6) |
7.9 (6.2-10.6) (4.3;13.4) |
9.1 (7-11.1 ) (2.8;20.9) |
1.0-8.5 | 1.8 (1.3-2.4) (0.3;4.1) |
2 (1.5-2.9) (0.3;4.3) |
1.8 (1.4-2.7) (0.03;11.5) |
1.9 (1.2-3.9) (0.56;4.8) |
| Lymphocytes (x103) |
2.0-11.0 | 2.0-17.0 | 3.8 (3.1-4.5) (1.4;7.4) |
3.6 (3.1-4.1) (1.3;6.9) |
3.6 (2.9-4.4) (1.5;7.3) |
4.0-13.5 | 4.6 (3.7-6.1) (2.1;12.5) |
5.7 (4.5-7.6) (2-11.5) |
5.4 (4.5-7) (0.05;24.5) |
5.1 (4.7-7.4) (4;8.7) |
| Monocytes (x103) |
0.4-3.1 | 0.3-2.7 | 1.4 (1.0-1.8) (0.3;4.3) |
1.2 (1-1.6) (0.3;2.6) |
1.6 (1.2-2.1) (0.06;4.1) |
0.1-1.3 | 0.72 (0.48-0.97) (0.26;1.9) |
0.93 (0.65 (1.3) (0.07;2.6) |
1 (0.7-1.3) (0.01;6.4) |
0.88 (0.65-1.1) (0.43;2.4) |
| Eosinophils (x103) |
0.02-0.85 | 0.07-1.10 | 0.29 (0.19-0.4) (0.1;1.3) |
0.27 (0.19-0.36) (0.11;0.53) |
0.27 (0.2-0.39) (0.04;0.6) |
0.07-0.75 | 0.34 (0.21-0.47) (0.05;1.8) |
0.24 (0.14-0.29) (0.1;0.82) |
0.24 (0.16-0.36) (0;2.3) |
0.2 (0.13-0.33) (0.06;0.8) |
| Basophils (x103) |
0-0.64 | 0-0.25 | 0.6 (0.3-1.2) (0.05;7.4) |
0.3 (0.12-0.65) (0.03;1.4) |
0.5 (0.23-1.3) (0.04;4.1) |
0-0.2 | 0.11 (0.06-0.15) (0.02;0.4) |
0.12 (0.07-0.21) (0.04;0.48) |
0.1 (0.07-0.16) (0.02;2.26) |
0.12 (0.09-0.18) (0.05-0.31) |
| Blood Differential (%) | ||||||||||
| Neutrophils | - | - | 57.2 (51.2-64.6) (6.7;74.6) |
57.9 (53.1-65.9) (49.5;77.2) |
59.3 (52.6-65) (37.3;73.9) |
- | 22.1 (18.6-28.6) (4.9;41.3) |
22.2 (17.6-29.3) (5.1;38.8) |
21.4 (17.3-27.5) (3.6;52.7) |
24.4 (12.3-31) (9.2;43.9) |
| Lymphocytes | - | - | 24.1 (18.9-30.4) (8.7;47.8) |
28.5 (19.6-32.3) (12.2;39.2) |
23.1 (18.2-28.4) (10.7;49.7) |
- | 64.1 (52.7-67.1) (43.6;74.2) |
62.5 (58.5-69.2) (44.8;71.5) |
62.6 (56.3-68) (32.8;86.8) |
65.1 (53.5-72.9) (43.2;78.3) |
| Monocytes | - | - | 8.9 (7.3-10.4) (2.5;16.1) |
8.6 (8.2-10.6) (4.2;13.3) |
10.3 (9.1-12.2) (0.9;22) |
- | 8.6 (7.5-11.2) (5.1;19.4) |
10.7 (8.3-13.2) (0.6;15.2) |
10.6 (8.8-12.9) (0.7;31.7) |
9.2 (7.7-11.8) (5.8;17.3) |
| Eosinophils | - | - | 1.7 (1.3-2.4) (0.7;9.3) |
2 (1.4-2.5) (0.9;3.8) |
1.7 (1.3-2.3) (0.5;4.9) |
- | 4.1 (2.6-5.8) (0.5;20) |
2.3 (1.4-3.6) (0.9;12.7) |
2.7 (1.7-3.8) (0.1;25.5) |
2.3 (1.9-2.6) (0.8;7.4) |
| Basophils | - | - | 3.5 (2-8.1) (0.5;41.6) |
2.1 (1.1-4.2) (0.4;7.6) |
3.2 (1.6-8.8) (0.5;21.4) |
- | 1.2 (0.85-1.7) (0.5;5.3) |
1.3 (0.8-1.8) (0.6;11.1) |
1.1 (0.9-1.4) (0.3;38.3) |
1.2 (1-1.65) (0.8;3.3) |
IQR: interquartile range (25th and 75th percentiles).
Min: lowest value for the haematologic parameter; max: highest value for the haematologic parameter.
DISA reference ranges are shown for two infant groups namely birth and 1 day to 1 week.
Av: average value.
Statistical Tests
The non-parametric Mann-Whitney U test was used to determine whether there were any significant differences in the proportions and absolute cell counts between the infant study groups. Statistical analyses were performed using SPSS software (version 17.0, SPSS Inc., Chicago, IL). All statistical tests were two-tailed and considered significant at P<0.05.
RESULTS
Clinical Characteristics of the HIV-1-Seropositive Mothers and their Infants
The maternal clinical characteristics and the antiretroviral regimens given to the mothers to reduce MTCT of HIV-1 of the infants included in this study are presented in Table 1. At birth, viral loads and CD4 counts were not significantly different between mothers that did not take NVP and those that did (P=0.688 and P=0.294, respectively). Mothers given triple drugs were only recruited at either 6 weeks or 12 weeks and their viral loads and CD4 counts were significantly lower than those of NVP unexposed (P=0.001 and P<0.001 respectively) and NVP treated mothers (P<0.001 and P<0.001 respectively).
Changes in Haematological Profiles within the First Six Weeks of Life
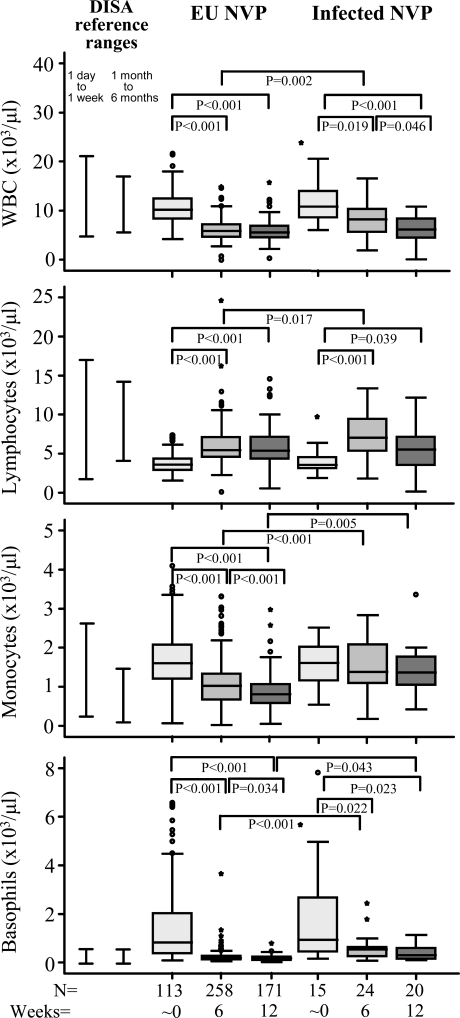
Although maturational abnormalities have been associated with intrauterine exposure to HIV-1, quantitative changes (birth to 6 weeks) in the infant cell type counts, consistent with the maturational development expected in infants after birth, was observed amongst all infant groups including those exposed to maternal NVP i.e. control, exposed uninfected (EU) no NVP, EU NVP (Table 2) and infected NVP (Table 3) infants. When comparing control and EU (no NVP) infants, blood cell profiles were not different at birth or at 6 weeks (with the exception of basophil counts (P=0.005) and percentages (P=0.009), which were lower in EU infants (no NVP) at birth) (Table 2). As birth samples were taken within 48 hours of delivery; reference ranges are therefore provided for both range categories for comparison (birth and 1 day – 1 week). Irrespective of infant infection status (controls, EU or infected) and maternal drug regimen, most infants at birth, tended to have normal ranges of neutrophil, lymphocyte, monocyte, basophil and eosinophil counts when compared to DISA reference ranges.
Table 3.
Maturational Changes (Birth to 12 Weeks) Amongst Exposed Uninfected and HIV-1 Infected Infants whose Mothers were Given NVP
| Infant Group | Birth NVP | 6 Weeks NVP | 12 Weeks NVP | |||
|---|---|---|---|---|---|---|
| Haematologic Parameter Median (IQR) (min; max) |
EU | HIV-1 Infected | EU | HIV-1 Infected | EU | HIV-1 Infected |
| No. of infants | 113 | 15 | 258 | 24 | 171 | 20 |
| Haemoglobin(g/dL) | 17.6 (15.9-19.5) (10.9;23) |
17.6 (15.8-19.1) (9.6;21.3) |
11.2 (10.5-12) (2;16.3) |
11 (9.7-12.3) (5;18) |
11.6 (10.9-12) (6;14.8) |
11 (10.6-11.4) (8.9;12.1) |
| MCV(fL) | 108 (102-111) (82;118) |
106 (103-108) (93;112) |
92 (88-95) (70;190) |
88 (85-93.8) (79;101) |
83 (80-86) (69;92) |
84 (81-85.8) (75;89) |
| Absolute Counts (/µL) | ||||||
| Platelets(x103) | 237 (178.5-280.5) (13;413) |
204 (173-261) (41;283) |
420 (334-529) (3.1;805) |
362.5 (304-451.3) (97;676) |
442 (363-532) (35;819) |
260 (190.8-383.3) (35;544) |
| RBC(x106) | 4.9 (4.4 -5.3) (3;6.8) |
5 (4.3-5.2) (2.9;5.7) |
3.5 (3.2-3.9) (0.7;306) |
3.6 (3.2-3.9) (1.5;4.9) |
4.1 (3.8-4.3) (2;5.2) |
3.8 (3.7-3.9) (3.1;4.2) |
| WBC(x103) | 15.6 (12.9-19.3) (6.6;33) |
16.3 (12.4-21.8) (9.1;31) |
9.1 (7.3-11.1) (0.1;36.3) |
12.4 (8.5-16) (2.8;25) |
8.6 (7.1-10.7) (0.7;24) |
9.3 (6.7-12.9) (0.1;16.3) |
| Neutrophils(x103) | 9.1 (7-11.1 ) (2.8;20.9) |
9.4 (8.3-12.5) (4.5;15.5) |
1.8 (1.4-2.7) (0.03;11.5) |
2.3 (1.5-3) (0.2;4.8) |
1.8 (1.2-2.5) (0.14;7.7) |
1.7 (1.4-2.2) (0.54;4.9) |
| Lymphocytes(x103) | 3.6 (2.9-4.4) (1.5;7.3) |
3.5 (3.1-4.8) (1.9;9.7) |
5.4 (4.5-7) (0.05;24.5) |
7 (5.3-9.9) (1.8;13.3) |
5.5 (4.3-7.1) (0.5:14.5) |
5.5 (3.5-7.1) (0.1;12.1) |
| Monocytes(x103) | 1.6 (1.2-2.1) (0.06;4.1) |
1.6 (1.1-2.1) (0.56;2.5) |
1 (0.7-1.3) (0.01;6.4) |
1.4 (1.1-2.1) (0.2;6) |
0.79 (0.57-1.1) (0.04;3) |
1.3 (0.56-1.5) (0.02;3.4) |
| Eosinophils(x103) | 0.27 (0.2-0.39) (0.04;0.6) |
0.28 (0.17-0.33) (0.05;0.5) |
0.24 (0.16-0.36) (0;2.3) |
0.35 (0.13-0.49) (0.06;4.7) |
0.24 (0.13-0.35) (0.01;1.2) |
0.3 (0.1-0.6) (0.02;2.3) |
| Basophils(x103) | 0.5 (0.23-1.3) (0.04;4.1) |
0.47 (0.15-1.38) (0.08;3.9) |
0.1 (0.07-0.16) (0.02;2.26) |
0.28 (0.13-0.33) (0.04;1.22) |
0.1 (0.06-0.14) (0;0.48) |
0.16 (0.09-0.26) (0.01;0.57) |
| Blood Differential (%) | ||||||
| Neutrophils | 59.3 (52.6-65) (37.3;73.9) |
60 (51.4-66.1) (39.4;76.4) |
21.4 (17.3-27.5) (3.6;52.7) |
18.8 (15.1-25.2) (6.3;41.1) |
20.3 ( 14.9-27.6) (4.1;65.1) |
20 (12.2-25.1) (8.5;40) |
| Lymphocytes | 23.1 (18.2-28.4) (10.7;49.7) |
21.5 (18.5-31.8) (13.1;44.2) |
62.6 (56.3-68) (32.8;86.8) |
57.7 (51.6-65.9) (44.6;79.7) |
65.8 (57.6-72.1) (23.7;86.9) |
60 (50.4-69.9) (44.8;78.9) |
| Monocytes | 10.3 (9.1-12.2) (0.9;22) |
10.2 (9-11.1) (4.5;14.1) |
10.6 (8.8-12.9) (0.7;31.7) |
12.4 (10.3-14.5) (4.1;31.5) |
8.6 (6.6-11.3) (0.8;22.9) |
11.5 (9.4-15.7) (7;23.6) |
| Eosinophils | 1.7 (1.3-2.3) (0.5;4.9) |
1.6 (1.2-1.9) (0.5;2.3) |
2.7 (1.7-3.8) (0.1;25.5) |
2.5 (1.4-5.2) (0.3;28.2) |
2.5 (1.6-4.2) (0.2;15.4) |
3 (1.7-6.9) (0.2;17.4) |
| Basophils | 3.2 (1.6-8.8) (0.5;21.4) |
2.9 (1.4-8.6) (0.6;12.6) |
1.1 (0.9-1.4) (0.3;38.3) |
2 (1.5-2.5) (0.6;5.1) |
1 (0.9-1.3) (0.4;3.3) |
1.7 (1.3-2.3) (0.7;9.7) |
IQR: interquartile range (25th and 75th percentiles).
Min: lowest value for the haematologic parameter; max: highest value for the haematologic parameter.
Maturation Profiles of Infants are Affected by HIV-1 Infection
At birth, leukocyte profiles were not significantly different between EU infants and infected infants exposed to maternal sdNVP (Table 3 and Fig.1). By 6 weeks however, HIV-1 infected infants had significantly higher WBC counts (P=0.002), lymphocyte counts (P=0.017), monocyte percentages (P=0.011), monocyte counts (P<0.001), basophil percentages (P<0.001) and basophil counts (P<0.001) compared to EU infants (Table 3 and Fig.1). At 12 weeks monocyte percentages and monocyte counts were still increased (P=0.001 and P=0.005 respectively) as were basophil percentages and basophil counts (P<0.001 and P=0.043 respectively) relative to EU infants (Table 3 and Fig. 1). In addition RBC counts (P=0.002), HgB levels (P=0.008) and platelet counts (P<0.001) were significantly reduced in HIV-1 infected infants compared to EU infants at 12 weeks (Table 3).
Fig. (1).
Leukocyte maturation profiles of HIV-1 exposed uninfected and HIV-1 infected infants exposed to NVP given to HIV-1 infected mothers to reduce maternal transmission of HIV-1. Data are presented as medians (horizontal bar), 25th and 75th percentiles (boxes), and 10th and 90th percentiles (bars). Significant differences between groups and sample numbers per group are indicated. The South African DISA reference ranges for infants 1 day to 1 week and 1 month to 6 months are indicated by bars appearing next to the control infant data.
Maturation Profiles of Exposed Uninfected Infants are Affected by Exposure to Maternal sdNVP
Having shown that infant leukocyte profile was not significantly affected by exposure to HIV-1 in the absence of HIV infection (EU infants) we wanted to establish whether exposure to maternal sdNVP influences blood counts. We show that haematological profile changes, consistent with maturational development, occurs irrespective of maternal sdNVP exposure (EU no NVP compared to EU NVP groups; Table 2) however, monocyte percentages (P=0.016), monocyte counts (P=0.039) and basophil counts (P=0.033) were significantly elevated at birth amongst EU NVP treated infants versus untreated EU infants (Table 2). By six weeks of age, there were no significant differences in the leukocyte profiles of no NVP and NVP exposed infant groups (Table 2). Infants exposed to maternal triple-drug therapy had significantly reduced RBC counts (P=0.024) compared to maternal sdNVP exposed infants. By 12 weeks only basophil percentages in triple-drug exposed infants remained significantly higher than those of NVP exposed infants (P=0.018) (data not shown).
Gender (male versus female infants) was not found to influence haematological values in the control, EU (unexposed or exposed to combination therapy) or HIV-1 infected (NVP exposed) infants at birth, 6 weeks or 12 weeks (where these comparisons were possible based on sample size). Curiously, at birth EU infant boys (n=49) exposed to maternal sdNVP had significantly higher lymphocyte percentages (P=0.035) but significantly reduced RBC counts (P=0.020), basophil counts (P=0.012) and percentages (P=0.015) compared to EU infant girls (n=64) exposed to maternal sdNVP (data not shown). These data could imply that haematological values of male and female infants may be uniquely affected by short courses of NVP exposure.
At Birth Infant White Blood Cell Counts and Differentials are Influenced by Maternal CD4 Count
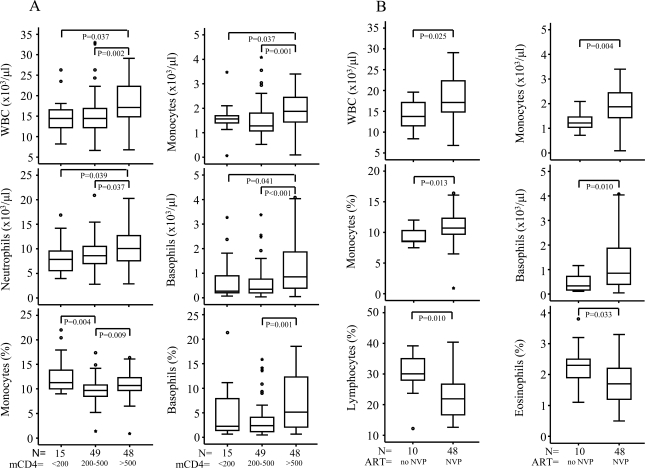
The white blood cell counts and differentials of EU NVP-exposed infants were compared when stratified on the basis of maternal CD4 counts, namely, <200 cells/μl; 200-500 cells/μl (intermediate CD4 counts) and >500 cells/μl (normal - counts range from 500-1500 x 106/l). We found WBC and basophil, neutrophil and monocyte counts to be significantly lower amongst infants whose mothers had low CD4 counts (<200 cells/µl) relative to those with high CD4 counts (>500 cells/μl). Basophil percentages followed a similar pattern and for monocyte percentages those with intermediate CD4 counts (200-500 cells/µl) had the lowest values (Fig. 2A). EU infants not exposed to maternal sdNVP also tended to have lower levels of lymphocyte, monocyte, eosinophil and basophil percentages and lymphocyte, eosinophil and basophil counts when born to mothers with CD4 counts 200-500 cells/µl compared to those born to mothers with higher >500 cells/μl (insufficient sample numbers for <200 cells/μl group; data not shown). We found no significant differences in white blood cell counts and differential of EU infants when separated on the basis of maternal viral load that is, viral loads <50,000 RNA copies/ml or >50,000 RNA copies/ml (high) (data not shown).
Fig. (2).
Leukocyte profiles of newborn infants differentiated according to maternal CD4 counts (<200 cells/µl, 200-500 cells/µl and >500 cells/µl). Mothers of these infants were given sdNVP to reduce MTCT of HIV-1 (A). Significant differences between leukocyte profiles of EU infants at birth born to mothers with CD4 counts >500 cells/µl that had either not taken sdNVP or had taken sdNVP to reduce MTCT of HIV-1 are presented in (B). Data are presented as medians (horizontal bar), 25th and 75th percentiles (boxes), and 10th and 90th percentiles (bars). Significant differences between groups and sample numbers per group are indicated.
Among EU infants born to mothers with CD4 counts >500 cells/μl (Fig. 2B), WBC, monocyte percentages and monocyte and basophil counts were significantly elevated but lymphocyte and eosinophil percentages significantly decreased amongst those infants exposed to the dose of NVP given to the mothers compared to NVP-unexposed EU infants (Fig. 2B). Alterations in numbers or proportions of certain cell types in these EU infants may be a feature of higher maternal CD4 counts, with some of these changes further accentuated in the presence of exposure to maternal sdNVP.
DISCUSSION
Pregnancy is characterized by a change in the balance of the innate and adaptive immune responses, more specifically, changes in cell counts, phenotypes, functions and soluble factors [20]. This altered balance is further impacted on by HIV-1 infection and with the use of antiretroviral drugs to prevent MTCT of HIV-1. While there is no vascular continuity between mother and foetus the transplacental passage of maternal components is known to occur. In fact the transplacental passage of IgG [22], low molecular weight substances e.g. amino acids [23] and inorganic ions [24] as well as inflammatory cytokines (interleukin (IL)-6) [25] and antiretrovirals [26] is well recognised. Neonatal life is a very unique developmental time, where the peripheral pool is stimulated and develops memory (either to foreign- or to self-antigen). Regulation and development of both the innate and adaptive immune responses in an infant therefore requires the presence and interaction of different cell types and several soluble factors e.g. cytokines and chemokines. Considering the fetomaternal interface, the dysregulation of maternal immune responses associated with pregnancy and HIV-1 infection, we aimed to describe alterations in haematological parameters (particularly white blood cell counts and differentials) of infants born to HIV-1 infected mothers, mainly in the context of exposure or not to maternal sdNVP. We further questioned whether maternal immune status might affect the cell profiles of their matched newborns. As some mothers did receive triple drug therapy their infants were also included for study from 6 weeks of age.
As expected there were age-dependant maturational changes (innate to adaptive immunity) demonstrated by the quantitative changes in the peripheral blood of newborns from all infant groups (uninfected, EU, HIV-1 infected). In particular, neutrophils (indicative of innate immune responses) predominated at birth whilst lymphocytes (indicative of a developing adaptive immune system) prevailed by 6 weeks. We found no significant differences between haematological values of control male and female infants or between HIV-1 exposed uninfected male and female infants whose mothers were not given sdNVP or that were given combination therapy. A study conducted on Malawian neonates has similarly reported no haematological variation between male and female neonates (males, n=177; females n=189) [27]. However, sdNVP given to mothers to reduce MTCT appeared to influence male and female blood parameters differentially. This intriguing interaction should be investigated in other cohorts.
Several studies have shown Africans may have different cell count profiles [28-31] and it is important that what may be normal ranges for a particular population group be used as the appropriate frame of reference. We therefore used tables with reference ranges suitable for South African infants. Although neutropenia is more commonly associated with AZT therapy it has been described in infants given NVP [32]. However, our study which only examined effects of maternal HIV-1 and drug exposures up to 12 weeks after birth, failed to demonstrate any association between short-course monotherapy or combination therapy and lower neutrophil counts, unlike other reported studies [4, 8, 10]. Studies conducted in Europe [4] and the United States [8] have indicated that perinatal exposure to antiretrovirals results in significant changes in haematological parameters observed in infants, many of which may persist up to 2 years of age. However, studies differ in the haematological parameters that remain affected for this time period.
With the exception of the basophil counts and percentages which were reduced in EU infants, our study shows no differences in haematological parameters at birth when comparing control infants to EU (no NVP) infants, suggesting that exposure to HIV-1 or other factors in utero reduces basophil numbers at birth, possibly due to altered signals that may affect trafficking of basophils from bone marrow to the periphery. This reduction relative to controls was not maintained at 6 weeks. It is not clear if this would have been maintained had there been no sdNVP given to the infant as PEP following birth, as comparisons between EU infants with and without maternal sdNVP exposure revealed elevations in monocyte percentages and counts and basophil counts in NVP-exposed neonates; these differences were however not maintained at 6 weeks of age. Overall, it appears that short exposure to NVP at birth has transient effects on immune cell parameters. It would be important in future to establish if longer regimens of drug treatments may maintain elevations of various cell types in the peripheral circulation.
Triple drug therapy decreased RBC counts at 6 weeks and increased basophil percentages at 12 weeks relative to sdNVP EU infants. The differences between EU infants and HIV-1 infected infants, assessed in sdNVP exposed infants, was most apparent at 6 weeks of age where WBC counts overall were increased, this being the result of expansions of lymphocytes, monocytes and basophils. The latter two cell type increases were maintained at 12 weeks with accompanying reductions in RBC counts, HgB levels and platelet counts. Therefore HIV-1 infected infants, not unexpectedly, showed more progressive changes over time in haematological parameters and these changes are a likely consequence of chronic immune activation [13].
Perhaps the most intriguing result was that when HIV-1 infected mothers were stratified according to CD4 cell count, lower CD4 cell counts (200-500 cells/μl) were associated with newborn decreases in white blood cell differentials of lymphocyte, monocyte, eosinophil and basophil percentages and lymphocyte, eosinophil and basophil counts. Some of these alterations were even further accentuated by exposure to maternal sdNVP viz. WBC, monocyte and basophil counts and monocyte percentages were increased, while lymphocyte and eosinophil percentages were significantly decreased. These findings raise some interesting questions.
It is notable that uninfected infant mortality and morbidity has been significantly associated with HIV-1 infected mothers with advanced disease (low CD4 counts) [33]. It may be expected that this greater risk to the infant may involve effects of maternal health status on the maturational development of infant blood parameters and also immunological deficiencies influencing such factors as the passively acquired maternal antibodies. Although we observed alterations in infant cell counts by maternal CD4 counts, infants born to mothers with high CD4 counts (>500 cells/ul), with or without NVP exposure, tended to have the most elevations relative to expected norms. The infant leukocyte parameters we measured are quite non-specific and it is interesting that variations by maternal CD4 count were detected at all. We speculate that the data can be explained in the context of an “anergy” that exists in the EU infants born to mothers with low CD4 counts (<200 cells/µl), in that these infants although likely exposed to more HIV-1 because maternal viral loads are generally higher, are unable to respond in a similar fashion to their counterparts born to mothers with higher CD4 counts and more robust immune systems. It is interesting that such an “anergy” may exist in these uninfected infants merely through being born to a mother with advanced disease. It may be that a lack of response to exposure in this way may alter their immune capability to challenge.
On the other hand, increases in certain white blood cell counts and differentials may also have consequences on the immune (innate and adaptive) capability of infants. Findings here suggest that NVP may actually influence responses possibly favouring innate immune capacity over adaptive (reduced proportions of lymphocytes at birth). Furthermore, higher basophil counts are often associated with Th2-related responses [34] and their elevations may be indicative of a more prominent Th2 milieu. Could NVP exert a protective effect beyond its direct role as an antiviral through enhancing innate as opposed to adaptive responses, or through prolonging a Th2 immune milieu? It would be important in future to establish which lymphocyte subsets are altered where proportions of overall lymphocytes are decreased, however if due to reductions in numbers of T cells this might help explain why HIV-specific responses detectable in EU infants are elusive to detection in the context of sdNVP exposure [35], and such responses are reduced in NVP-exposed mothers [36]. A more Th2 oriented environment may also not favour detection of HIV-specific Th1 responses.
A number of studies have raised concerns over the issue of increased frequencies of NVP-induced adverse events (hepatotoxicity and rash) among pregnant HIV-1 infected women [37, 38]. Hepatotoxicity has been more often associated with higher CD4 counts (>250 cells/μl) [39, 40]. Another interpretation of our data may be that more subtle changes of allergy-type responses may be demonstrable in peripheral blood of women with high CD4 cell counts and their matched infants (given their genetic relatedness one might expect similar responses). It is particularly interesting that throughout our analyses basophils emerge as a cell type affected by HIV-1 exposure and infection and NVP and triple drug therapy. Basophils represent only 0.01% to 0.03% circulating leukocytes and characteristically are histamine containing metachromatic granulocytes which when activated exocytose their granule constituents and slowly generate and release cytokines, chemokines and arachidonic acid metabolites. They, along with tissue-based mast cells play an important role in IgE-mediated inflammatory responses. Recent studies in mice have reported basophils to be essential for B-cell memory responses by promoting T helper cell-2 dependent B-cell help [41]. Why NVP might enhance basophil and monocyte counts while decreasing lymphocytes and eosinophil numbers is unclear, but given our previous report of evidence of increased immune activation (elevated plasma soluble activation markers in both mothers and matched infants) through the short exposure to NVP given at the onset of delivery [13] it is likely that trafficking of cells would occur differentially culminating in alterations of cell compositions in the peripheral circulation.
In conclusion, this study has shown that maternal HIV-1 disease status and antiretroviral drug exposure influence infant haematological parameters which may have implications for infant immune response capability. Although these effects may be transient it is not clear how these differences impact (whether at all) on an infant’s ability to respond to HIV-1 challenge or control HIV-1 viraemia or if they influence the induction of protective immune responses by vaccines in early life. Furthermore, this study adds to the evidence purporting the importance of improving maternal health and providing treatment options that reduce maternal viral load and increase CD4 counts.
ACKNOWLEDGEMENTS
This study was supported in part by grants from NICHD 42402 and the Wellcome Trust. CTT is a Wellcome Trust International Senior Research Fellow (076352/Z/05/Z). We wish to thank all participants, the study coordinator, Sarita Lalsab, and the staff of the Perinatal HIV Research Unit, Chris Hani Baragwanath and Coronation Hospitals, for their valuable contribution.
COMPETING INTERESTS STATEMENT
The authors declare that they have no competing financial interests.
REFERENCES
- 1.McDonagh S, Maidji E, Ma W, et al. Viral and bacterial pathogens at the maternal-fetal interface. J Infect Dis. 2004;190:826–34. doi: 10.1086/422330. [DOI] [PubMed] [Google Scholar]
- 2.Clerici M, Saresella M, Colombo F, et al. T-lymphocyte maturation abnormalities in uninfected newborns and children with vertical exposure to HIV. Blood. 2000;96:3866–71. [PubMed] [Google Scholar]
- 3.Nielsen SD, Jeppesen DL, Kolte L, et al. Impaired progenitor cell function in HIV-negative infants of HIV-positive mothers results in decreased thymic output and low CD4 counts. Blood. 2001;98:398–404. doi: 10.1182/blood.v98.2.398. [DOI] [PubMed] [Google Scholar]
- 4.Le Chenadec J, Mayaux MJ, Guihenneuc-Jouyaux C, Blanche S. Perinatal antiretroviral treatment and hematopoiesis in HIV-uninfected infants. AIDS. 2003;17:2053–61. doi: 10.1097/00002030-200309260-00006. [DOI] [PubMed] [Google Scholar]
- 5.Bunders MJ, Bekker V, Scherpbier HJ, et al. Haematological parameters of HIV-1-uninfected infants born to HIV-1-infected mothers. Acta Paediatr. 2005;94:1571–7. doi: 10.1080/08035250510042951. [DOI] [PubMed] [Google Scholar]
- 6.Bunders M, Thorne C, Newell ML. Maternal and infant factors and lymphocyte, CD4 and CD8 cell counts in uninfected children of HIV-1-infected mothers. AIDS. 2005;19:1071–9. doi: 10.1097/01.aids.0000174454.63250.22. [DOI] [PubMed] [Google Scholar]
- 7.El Beitune P, Duarte G. Antiretroviral agents during pregnancy: consequences on hematologic parameters in HIV-exposed, uninfected newborn infant. Eur J Obstet Gynecol Reprod Biol. 2006;128:59–63. doi: 10.1016/j.ejogrb.2006.01.013. [DOI] [PubMed] [Google Scholar]
- 8.Pacheco SE, McIntosh K, Lu M, et al. Effect of perinatal antiretroviral drug exposure on hematologic values in HIV-uninfected children: An analysis of the women and infants transmission study. J Infect Dis. 2006;194:1089–97. doi: 10.1086/507645. [DOI] [PubMed] [Google Scholar]
- 9.Feiterna-Sperling C, Weizsaecker K, Buhrer C, et al. Hematologic effects of maternal antiretroviral therapy and transmission prophylaxis in HIV-1-exposed uninfected newborn infants. J Acquir Immune Defic Syndr. 2007;45:43–51. doi: 10.1097/QAI.0b013e318042d5e3. [DOI] [PubMed] [Google Scholar]
- 10.Bae WH, Wester C, Smeaton LM, et al. Hematologic and hepatic toxicities associated with antenatal and postnatal exposure to maternal highly active antiretroviral therapy among infants. AIDS. 2008;22:1633–40. doi: 10.1097/QAD.0b013e328307a029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hygino J, Lima PG, Filho RG, et al. Altered immunological reactivity in HIV-1-exposed uninfected neonates. Clin Immunol. 2008;127:340–7. doi: 10.1016/j.clim.2008.01.020. [DOI] [PubMed] [Google Scholar]
- 12.Ono E, Nunes dos Santos AM, de Menezes Succi RC, et al. Imbalance of naive and memory T lymphocytes with sustained high cellular activation during the first year of life from uninfected children born to HIV-1-infected mothers on HAART. Braz J Med Biol Res. 2008;41:700–8. doi: 10.1590/s0100-879x2008000800011. [DOI] [PubMed] [Google Scholar]
- 13.Schramm DB, Kuhn L, Gray GE, Tiemessen CT. In vivo effects of HIV-1 exposure in the presence and absence of single-dose nevirapine on cellular plasma activation markers of infants born to HIV-1-seropositive mothers. J Acquir Immune Defic Syndr. 2006;42:545–53. doi: 10.1097/01.qai.0000225009.30698.ce. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fagoaga OR, Nehlsen-Cannarella SL. Maternal modulation of neonatal immune system development. Dev Immunol. 2002;9:9–17. doi: 10.1080/10446670290030972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kuhn L, Meddows-Taylor S, Gray G, Tiemessen C. Human immunodeficiency virus (HIV)-specific cellular immune responses in newborns exposed to HIV in utero. Clin Infect Dis. 2002;34:267–76. doi: 10.1086/338153. [DOI] [PubMed] [Google Scholar]
- 16.Chotpitayasunondh T, Vanprapar N, Simonds RJ, et al. Bangkok Collaborative Perinatal HIV Transmission Study Group. Safety of late in utero exposure to zidovudine in infants born to human immunodeficiency virus-infected mothers: Bangkok. Pediatrics. 2001;107:E5. doi: 10.1542/peds.107.1.e5. [DOI] [PubMed] [Google Scholar]
- 17.Taha TE, Kumwenda N, Kafulafula G, et al. Haematological changes in African children who received short-term prophylaxis with nevirapine and zidovudine at birth. Ann Trop Paediatr. 2004;24:301–9. doi: 10.1179/027249304225019127. [DOI] [PubMed] [Google Scholar]
- 18.Barret B, Tardieu M, Rustin P, et al. Persistent mitochondrial dysfunction in HIV-1-exposed but uninfected infants: clinical screening in a large prospective cohort. AIDS. 2003;17:1769–85. doi: 10.1097/00002030-200308150-00006. [DOI] [PubMed] [Google Scholar]
- 19.European Collaborative Study. Levels and patterns of neutrophil cell counts over the first 8 years of life in children of HIV-1-infected mothers. AIDS. 2004;18:2009–17. doi: 10.1097/00002030-200410210-00005. [DOI] [PubMed] [Google Scholar]
- 20.Luppi P. How immune mechanisms are affected by pregnancy. Vaccine. 2003;21:3352–7. doi: 10.1016/s0264-410x(03)00331-1. [DOI] [PubMed] [Google Scholar]
- 21.Department of Haematology, National Health Laboratory Service (NHLS), Chris Hani Baragwanath and Braamfontein Laboratories Service Handbook 2006-2007. Available from: http://www.health.gpg.gov.za/NHLS/CHB-Braamfontein_Lab_Hand_book.pdf . [[Accessed Oct, 2009]].
- 22.Simister NE. Placental transport of immunoglobulin G. Vaccine. 2003;21:3365–9. doi: 10.1016/s0264-410x(03)00334-7. [DOI] [PubMed] [Google Scholar]
- 23.Cetin I. Amino acid interconversions in the fetal-placental unit: the animal model and human studies in vivo. Pediatr Res. 2001;49:148–54. doi: 10.1203/00006450-200102000-00004. [DOI] [PubMed] [Google Scholar]
- 24.Stulc J. Placental transfer of inorganic ions and water. Physiol Rev. 1997;77:805–36. doi: 10.1152/physrev.1997.77.3.805. [DOI] [PubMed] [Google Scholar]
- 25.Zaretsky MV, Alexander JM, Byrd W, Bawdon RE. Transfer of inflammatory cytokines across the placenta. Obstet Gynecol. 2004;103:546–50. doi: 10.1097/01.AOG.0000114980.40445.83. [DOI] [PubMed] [Google Scholar]
- 26.Roustit M, Jlaiel M, Leclercq P, Stanke-Labesque F. Pharmacokinetics and therapeutic drug monitoring of antiretrovirals in pregnant women. Br J Clin Pharmacol. 2008;66:179–95. doi: 10.1111/j.1365-2125.2008.03220.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mukiibi JM, Mtimavalye LA, Broadhead R, et al. Some haematological parameters in Malawian neonates. East Afr Med J. 1995;72:10–4. [PubMed] [Google Scholar]
- 28.Badenhorst CJ, Fourie J, Steyn K, et al. The haematological profile of urban black Africans aged 15-64 years in the Cape Peninsula. East Afr Med J. 1995;72:19–24. [PubMed] [Google Scholar]
- 29.Lugada ES, Mermin J, Kaharuza F, et al. Population-based hematologic and immunologic reference values for a healthy Ugandan population. Clin Diagn Lab Immunol. 2004;11:29–34. doi: 10.1128/CDLI.11.1.29-34.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tsegaye A, Messele T, Tilahun T, et al. Immunohematological reference ranges for adult Ethiopians. Clin Diag Lab Immunol. 1999;6:410–4. doi: 10.1128/cdli.6.3.410-414.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tugume SB, Piwowar EM, Lutalo T, et al. Hematological reference ranges among healthy Ugandans. Clin Diagn Lab Immunol. 1995;2:233–5. doi: 10.1128/cdli.2.2.233-235.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shetty AK, Coovadia HM, Mirochnick MM, et al. Safety and trough concentrations of nevirapine prophylaxis given daily, twice weekly, or weekly in breast-feeding infants from birth to 6 months. J Acquir Immune Defic Syndr. 2003;34:482–90. doi: 10.1097/00126334-200312150-00006. [DOI] [PubMed] [Google Scholar]
- 33.Kuhn L, Kasonde P, Sinkala M, et al. Does severity of HIV disease in HIV-infected mothers affect mortality and morbidity among their uninfected infants? Clin Infect Dis. 2005;41:1654–61. doi: 10.1086/498029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Min B, Paul WE. Basophils and type 2 immunity. Curr Opin Hematol. 2008;15:59–63. doi: 10.1097/MOH.0b013e3282f13ce8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kuhn L M-TS, Gray G, Schramm DB, Tiemessen CT. HIV-stimulated IL-2 production among exposed-uninfected infants of HIV-infected mothers given nevirapine prophylaxis. 10th Conference on Retroviruses and Opportunistic Infections; Boston MA. 2003. [Google Scholar]
- 36.Shalekoff S, Meddows-Taylor S, Schramm DB, et al. Single-dose nevirapine exposure affects T cell response and cytokine levels in HIV type 1-infected women. AIDS Res Hum Retroviruses. 2009;25:1049–53. doi: 10.1089/aid.2008.0286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Manfredi R, Calza L. Safety issues about nevirapine administration in HIV-infected pregnant women. J Acquir Immune Defic Syndr. 2007;45:365–8. doi: 10.1097/QAI.0b013e318050d879. [DOI] [PubMed] [Google Scholar]
- 38.Phanuphak N, Apornpong T, Teeratakulpisarn S, et al. Nevirapine-associated toxicity in HIV-infected Thai men and women, including pregnant women. HIV Med. 2007;8:357–66. doi: 10.1111/j.1468-1293.2007.00477.x. [DOI] [PubMed] [Google Scholar]
- 39.Jamisse L, Balkus J, Hitti J, et al. Antiretroviral-associated toxicity among HIV-1-seropositive pregnant women in Mozambique receiving nevirapine-based regimens. J Acquir Immune Defic Syndr. 2007;44:371–6. doi: 10.1097/QAI.0b013e318032bbee. [DOI] [PubMed] [Google Scholar]
- 40.Kondo W, Carraro EA, Prandel E, et al. Nevirapine-induced side effects in pregnant women: experience of a Brazilian university hospital. Braz J Infect Dis. 2007;11:544–8. doi: 10.1590/s1413-86702007000600004. [DOI] [PubMed] [Google Scholar]
- 41.Denzel A, Maus UA, Rodriguez Gomez M, et al. Basophils enhance immunological memory responses. Nat Immunol. 2008;9:733–42. doi: 10.1038/ni.1621. [DOI] [PubMed] [Google Scholar]