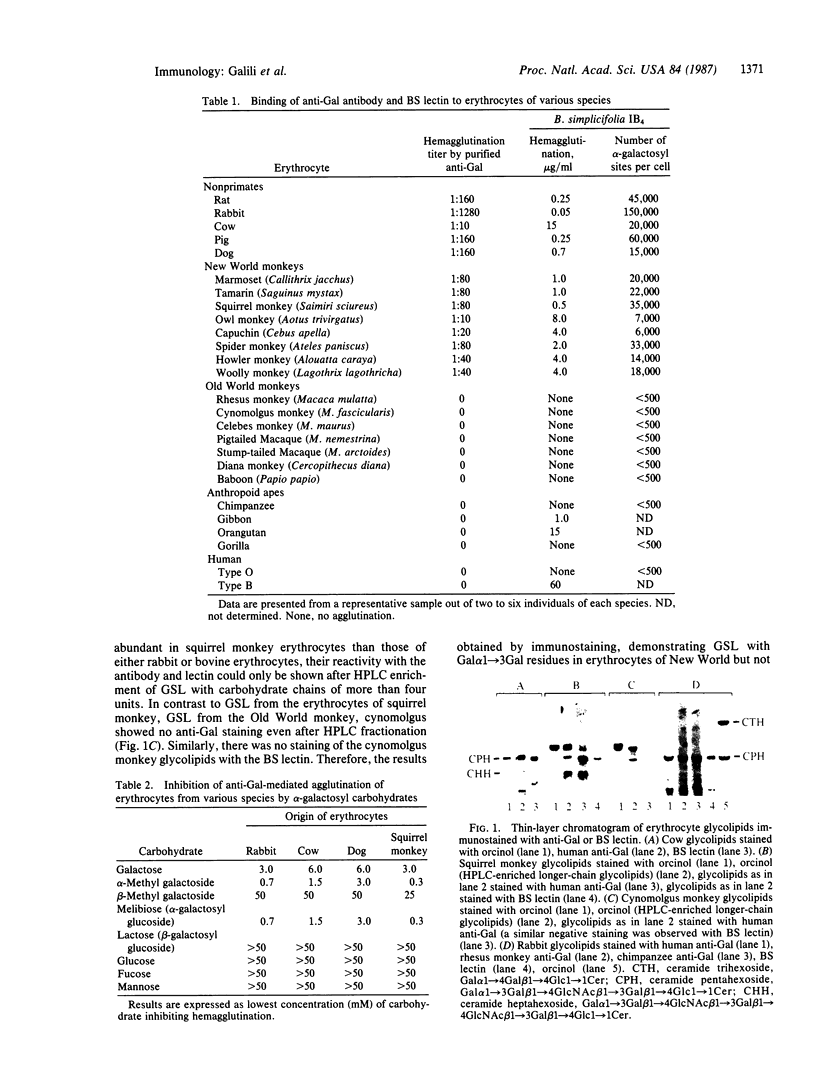
Abstract
Anti-Gal is a natural antibody, which constitutes as much as 1% of circulating IgG in humans and displays a distinct specificity for the structure Gal alpha 1----3Gal. This glycosidic structure has been found on various tissues of many nonprimate mammals. A comparative study of the occurrence of anti-Gal versus the expression of the Gal alpha 1----3Gal epitope was performed in primates, and a distinct evolutionary pattern was observed. Whereas anti-Gal was found to be present in Old World monkeys and apes in titers comparable to those in humans, its corresponding antigenic epitope is abundantly expressed on erythrocytes of New World monkeys. Immunostaining with anti-Gal of glycolipids from New World monkey erythrocytes indicated that the molecules to which anti-Gal binds are similar to those found in rabbit and bovine erythrocytes. These findings indicate that there is an evolutionary reciprocity between New World and Old World primates in the production of the Gal alpha 1----3Gal structure and the antibody that recognizes it. The expression of the Gal alpha 1----3Gal epitope was evolutionarily conserved in New World monkeys, but it was suppressed in ancestral lineages of Old World primates. The suppression of this epitope was accompanied by the production of anti-Gal. The observed in vivo binding of anti-Gal to human normal senescent and some pathologic erythrocytes implies that the Gal alpha 1----3Gal epitope is present in man in a cryptic form.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blanken W. M., Van den Eijnden D. H. Biosynthesis of terminal Gal alpha 1----3Gal beta 1----4GlcNAc-R oligosaccharide sequences on glycoconjugates. Purification and acceptor specificity of a UDP-Gal:N-acetyllactosaminide alpha 1----3-galactosyltransferase from calf thymus. J Biol Chem. 1985 Oct 25;260(24):12927–12934. [PubMed] [Google Scholar]
- Chien J. L., Li S. C., Li Y. T. Isolation and characterization of a heptaglycosylceramide from bovine erythrocyte membranes. J Lipid Res. 1979 Jul;20(5):669–673. [PubMed] [Google Scholar]
- Cummings R. D., Kornfeld S. The distribution of repeating [Gal beta 1,4GlcNAc beta 1,3] sequences in asparagine-linked oligosaccharides of the mouse lymphoma cell lines BW5147 and PHAR 2.1. J Biol Chem. 1984 May 25;259(10):6253–6260. [PubMed] [Google Scholar]
- Eckhardt A. E., Goldstein I. J. Isolation and characterization of a family of alpha-D-galactosyl-containing glycopeptides from Ehrlich ascites tumor cells. Biochemistry. 1983 Nov 8;22(23):5290–5297. doi: 10.1021/bi00292a007. [DOI] [PubMed] [Google Scholar]
- Egge H., Kordowicz M., Peter-Katalinić J., Hanfland P. Immunochemistry of I/i-active oligo- and polyglycosylceramides from rabbit erythrocyte membranes. Characterization of linear, di-, and triantennary neolactoglycosphingolipids. J Biol Chem. 1985 Apr 25;260(8):4927–4935. [PubMed] [Google Scholar]
- Eto T., Ichikawa Y., Nishimura K., Ando S., Yamakawa T. Chemistry of lipid of the posthemyolytic residue or stroma of erythrocytes. XVI. Occurrence of ceramide pentasaccharide in the membrane of erythrocytes and reticulocytes of rabbit. J Biochem. 1968 Aug;64(2):205–213. doi: 10.1093/oxfordjournals.jbchem.a128881. [DOI] [PubMed] [Google Scholar]
- Frank A., Andiman W. A., Miller G. Epstein-Barr virus and nonhuman primates: natural and experimental infection. Adv Cancer Res. 1976;23:171–201. doi: 10.1016/s0065-230x(08)60546-1. [DOI] [PubMed] [Google Scholar]
- Galili U., Clark M. R., Shohet S. B. Excessive binding of natural anti-alpha-galactosyl immunoglobin G to sickle erythrocytes may contribute to extravascular cell destruction. J Clin Invest. 1986 Jan;77(1):27–33. doi: 10.1172/JCI112286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galili U., Flechner I., Knyszynski A., Danon D., Rachmilewitz E. A. The natural anti-alpha-galactosyl IgG on human normal senescent red blood cells. Br J Haematol. 1986 Feb;62(2):317–324. doi: 10.1111/j.1365-2141.1986.tb02935.x. [DOI] [PubMed] [Google Scholar]
- Galili U., Korkesh A., Kahane I., Rachmilewitz E. A. Demonstration of a natural antigalactosyl IgG antibody on thalassemic red blood cells. Blood. 1983 Jun;61(6):1258–1264. [PubMed] [Google Scholar]
- Galili U., Macher B. A., Buehler J., Shohet S. B. Human natural anti-alpha-galactosyl IgG. II. The specific recognition of alpha (1----3)-linked galactose residues. J Exp Med. 1985 Aug 1;162(2):573–582. doi: 10.1084/jem.162.2.573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galili U., Rachmilewitz E. A., Peleg A., Flechner I. A unique natural human IgG antibody with anti-alpha-galactosyl specificity. J Exp Med. 1984 Nov 1;160(5):1519–1531. doi: 10.1084/jem.160.5.1519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanfland P., Kordowicz M., Niermann H., Egge H., Dabrowski U., Peter-Katalinic J., Dabrowski J. Purification and structures of branched blood-group-B-active glycosphingolipids from human erythrocyte membranes. Eur J Biochem. 1984 Dec 17;145(3):531–542. doi: 10.1111/j.1432-1033.1984.tb08589.x. [DOI] [PubMed] [Google Scholar]
- Ito M., Suzuki E., Naiki M., Sendo F., Arai S. Carbohydrates as antigenic determinants of tumor-associated antigens recognized by monoclonal anti-tumor antibodies produced in a syngeneic system. Int J Cancer. 1984 Nov 15;34(5):689–697. doi: 10.1002/ijc.2910340517. [DOI] [PubMed] [Google Scholar]
- Lüderitz O., Simmons D. A., Westphal G. The immunochemistry of Salmonella chemotype VI O-antigens. The structure of oligosaccharides from Salmonella group U (o 43) lipopolysaccharides. Biochem J. 1965 Dec;97(3):820–826. doi: 10.1042/bj0970820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magnani J. L., Brockhaus M., Smith D. F., Ginsburg V., Blaszczyk M., Mitchell K. F., Steplewski Z., Koprowski H. A monosialoganglioside is a monoclonal antibody-defined antigen of colon carcinoma. Science. 1981 Apr 3;212(4490):55–56. doi: 10.1126/science.7209516. [DOI] [PubMed] [Google Scholar]
- Spiro R. G., Bhoyroo V. D. Occurrence of alpha-D-galactosyl residues in the thyroglobulins from several species. Localization in the saccharide chains of the complex carbohydrate units. J Biol Chem. 1984 Aug 10;259(15):9858–9866. [PubMed] [Google Scholar]
- Springer G. F. Blood-group and Forssman antigenic determinants shared between microbes and mammalian cells. Prog Allergy. 1971;15:9–77. [PubMed] [Google Scholar]
- Sung S. S., Sweeley C. C. The structure of canine intestinal trihexosylceramide. Biochim Biophys Acta. 1979 Nov 21;575(2):295–298. doi: 10.1016/0005-2760(79)90031-6. [DOI] [PubMed] [Google Scholar]
- Van Halbeek H., Vliegenthart J. F., Winterwerp H., Blanken W. M., Van den Eijnden D. H. alpha-D-galactosyltransferase activity in calf thymus. A high-resolution 1H-NMR study. Biochem Biophys Res Commun. 1983 Jan 14;110(1):124–131. doi: 10.1016/0006-291x(83)91269-x. [DOI] [PubMed] [Google Scholar]
- Watanabe K., Hakomori S. I., Childs R. A., Feizi T. Characterization of a blood group I-active ganglioside. Structural requirements for I and i specificities. J Biol Chem. 1979 May 10;254(9):3221–3228. [PubMed] [Google Scholar]
- Wood C., Kabat E. A., Murphy L. A., Goldstein I. J. Immunochemical studies of the combining sites of the two isolectins, A4 and B4, isolated from Bandeiraea simplicifolia. Arch Biochem Biophys. 1979 Nov;198(1):1–11. doi: 10.1016/0003-9861(79)90389-8. [DOI] [PubMed] [Google Scholar]