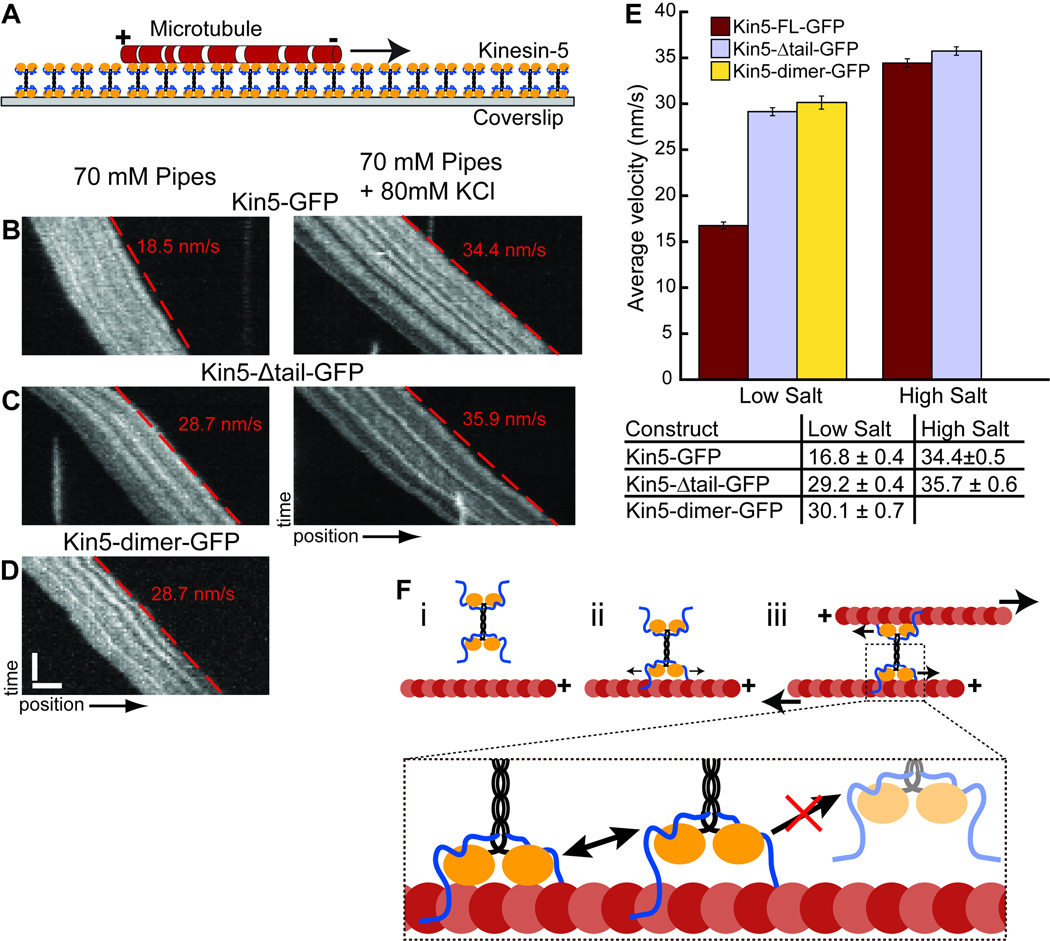
Figure 4. The kinesin-5 C-terminal tail domain reduces motility in low ionic strength.
(A) Schematic of the microtubule surface motility assay. X-rhodamine labeled microtubules bind to kinesin-5 constructs coating a glass surface. (B–D) Examples of Kymographs showing the movement of microtubules driven by Kin5-GFP (B), Kin5-Δtail-GFP (C) and Kin5-dimer-GFP (D) in high salt buffer (left) and low salt buffer (right). The microtubule velocity in each kymograph was determined from the slope of the red dashed line. (E) Average velocities for each construct in two buffer conditions. The population average for Kin5-dimer-GFP in high salt was not determined because the microtubules rapidly detached from the surface. (F) Model for the contribution of the microtubule binding site in the C-terminal tail domain to kinesin-5 motility. (i) A kinesin-5 molecule in solution prior to encountering a microtubule. (ii) The tail domain mediates the initial interaction, allowing 1-D diffusion and increases the probability of crosslinking a second microtubule. (iii) Relative filament sliding driven by kinesin-5’s two pairs of motor domains. The inset highlights how the non-motor tail domains maintain association with the filament and prevent full dissociation of kinesin-5 when the low processivity motor domains unbind. Error bars = sem.