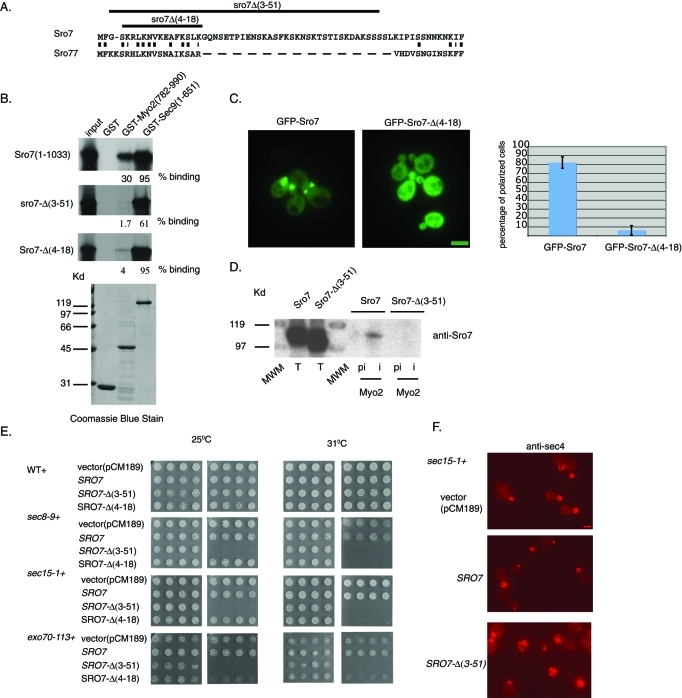
FIGURE 8:
The N-terminal domain of Sro7 is required for the interaction with Myo2. (A) Alignment of the N-terminal region of Sro7 and Sro77 Lgl family members. (B) Deletion of the N-terminal domain of Sro7 affects binding to Myo2. Binding of radiolabeled, in vitro–translated full-length and N-terminal truncations of Sro7 to GST-Myo2IQ and GST-Sec9 on beads. (C) Wild-type cells expressing GFP-Sro7 (CEN) or GFP–Sro7-Δ(4–18) (CEN) were grown to midlog in selective media and then observed by fluorescence microscopy. Quantitation was obtained by scoring >50 cells for the presence of GFP fluorescence at emerging bud sites and bud tips. Bar 1 μm. (D) Wild-type cells expressing Sro7 or Sro7-Δ(3–51) behind a tetracycline-repressible promoter (CEN) were spheroplasted, lysed, and subjected to immunoprecipitation with affinity-purified preimmune and anti-Myo2 antibody. (E) Wild type, sec15-1, sec8-9, and exo70-113 were transformed with a plasmid expressing Sro7, Sro7-Δ(3–51), or Sro7-Δ(4–18) behind a tetracycline-repressible promoter (CEN) or vector only. Four independent transformants were picked into microtiter wells and transferred to selective media in the presence or absence of doxycycline at different temperatures. (F) sec15-1 mutant cells transformed with a plasmid expressing Sro7, Sro7-Δ(3–51), or vector only were grown at 25°C in the presence of doxycycline and then shifted to media without doxycycline for 8 h before fixing and processing for immunofluorescence with monoclonal anti-Sec4 antibody. Scale 1 μm.