Figure 1.
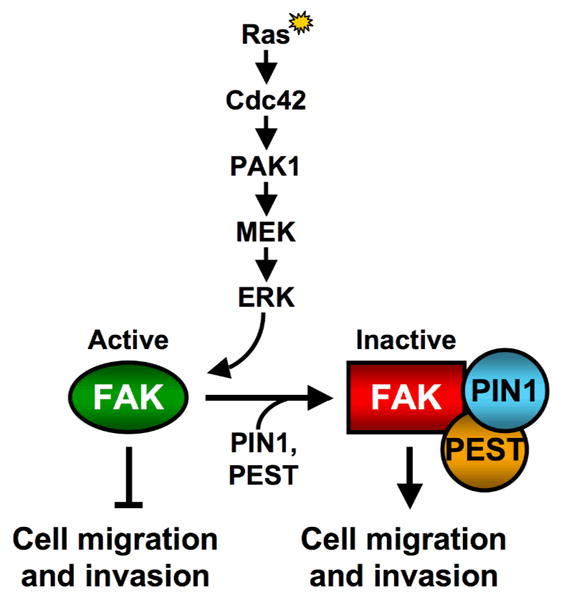
Scheme for the Ras-mediated inactivation of FAK. Activated Ras, signaling through Cdc42, PAK1 and MEK, leads to an ERK-induced phosphorylation of FAK at serine 910. This phosphorylation event triggers the binding of PIN1 to FAK which in turn leads to the recruitment of PTP-PEST to FAK. PTP-PEST then de-phosphorylates tyrosine 397 on FAK, a FAK-autophosphorylation site that is crucial for its signaling functions. Surprisingly, it is the resulting inactivation of FAK that appears to be necessary for Ras-stimulated cell migration and invasion.
