Abstract
Heat shock protein 72 (Hsp72) has been detected within saliva, and its presence may contribute to oral defence. It is currently unknown how physiological stress affects salivary Hsp72 or if salivary Hsp72 concentrations reflect plasma Hsp72 concentrations. We studied the effect of exercise upon salivary Hsp72 expression, and using caffeine administration, investigated the role of sympathetic stimulation upon salivary Hsp72 expression. Six healthy males performed two treadmill running exercise bouts in hot conditions (30°C) separated by 1 week in a randomized cross-over design, one with caffeine supplementation (CAF) the other with placebo (PLA). Plasma and saliva samples were collected prior to, during and post-exercise and assayed for Hsp72 concentration by ELISA. Exercise significantly increased plasma Hsp72, but not salivary Hsp72 concentration. Mean salivary Hsp72 concentration (5.1 ± 0.8 ng/ml) was significantly greater than plasma Hsp72 concentration (1.8 ± 0.1 ng/ml), and concentrations of salivary and plasma Hsp72 were unrelated. Caffeine supplementation and exercise increased the concentration of catecholamines, salivary α-amylase and total protein, whilst the salivary Hsp72:α-amylase ratio was lower in CAF. Salivary Hsp72 was not altered by exercise stress nor caffeine supplementation, and concentrations did not track plasma Hsp72 concentration.
Keywords: Adrenergic, Catecholamines, Cholinergic, Heat shock proteins, Saliva
Introduction
Heat shock protein (Hsp) 72 (also termed HSPA1A (Kampinga et al. 2008)) has been detected in saliva (Fabian et al. 2003), although it is unclear as to its role within this compartment. It has been speculated that salivary Hsp72 may have potential immunological functions in preventing bacterial infection and maintaining periodontal and mucosal health by way of a non-specific continuous immune surveillance (Fabian et al. 2007). The source of salivary Hsp72 is currently unknown, or if a physiological stress such as exercise alters the concentration of salivary Hsp72. Elucidating the effects of exercise stress upon salivary Hsp72 is warranted since exercise stress-induced increases in plasma Hsp72 concentration enhance aspects of the immune system such as neutrophil phagocytosis and chemotaxis (Ortega et al. 2008) and the cytolitic activity of natural killer cells (Horn et al. 2007). As such, exercise-associated increases in salivary Hsp72 may provide additional immuno-enhancement within the oral cavity.
The presence of Hsp72 in saliva is of interest from a methodological perspective since saliva collection is a simple non-invasive method which may be used as an alternative to invasive blood sampling. It is currently unknown if salivary Hsp72 concentrations reflect plasma Hsp72 concentrations. Since plasma Hsp72 is elevated in a number of pathological disorders, it has been proposed that circulating levels may serve as an adjunct diagnostic marker (Wright et al. 2000; Pittet et al. 2002; Abe et al. 2004). As such, the potential use for saliva Hsp72 as a proxy measure of plasma Hsp72 is appealing to both clinicians and researchers. Changes in plasma Hsp72 concentrations have been attributed to alterations in adrenergic activity (Whitham et al. 2006; Johnson et al. 2005), and caffeine, a known stimulator of sympathetic activity, has been shown to increase the concentration of plasma Hsp72 during exercise (Whitham et al. 2006). The role of alterations in adrenergic activity upon salivary Hsp72 is currently unclear, although studies suggest that salivary Hsp72 expression occurs independently of the sympathetically secreted enzyme α-amylase (Fabian et al. 2003, 2004). By providing caffeine during exercise and determining the salivary Hsp72:α-amylase and salivary Hsp72:total protein ratio, the effect of sympathetic stimulation upon salivary Hsp72 expression can be further investigated.
The purposes of this study were (1) to explore the effect of exercise upon salivary Hsp72 expression, (2) to investigate the utility of salivary Hsp72 to track changes in plasma Hsp72 concentration and (3) to examine the role of adrenergic stimulation upon the presence of salivary Hsp72.
Materials and methods
Participants
The study was approved by the ethics committee of The School of Sport, Health and Exercise Sciences of Bangor University. A sample size calculation was performed based on mean and standard deviation data from a previous study (Whitham et al. 2006). Six healthy endurance-trained males gave written informed consent to participate in the study having been fully informed of all procedures and risks involved in the experimental trials. Mean (±SD) demographical data for the participants were age, 21.8 ± 1.9 years; height, 1.79 ± 0.04 m; body mass, 71.4 ± 3.1 kg; maximal oxygen uptake (VO2max), 60.7 ± 2.8 ml/kg/min and habitual caffeine intake, 171 ± 91 mg/day, calculated from their responses to a questionnaire. All participants recruited were non-smokers and free from infection, known diseases, medication or any form of supplementation that may have interfered with the eHsp72, thermoregulatory or exercise responses. In addition, participants were free from known oral or dental disease.
Experimental trials
On two occasions, separated by at least 1 week and at the same time of day to control for circadian fluctuations in saliva flow rate, participants performed 2 × 30 min running bouts in an environmental chamber (Delta Environmental Inc., UK) where ambient conditions were maintained at 30°C and 40% relative humidity with either caffeine (CAF) or placebo (PLA) in a single-blind randomized cross-over design. Two different exercise intensities were used in order to see a progressive increase in plasma Hsp72 against which salivary Hsp72 could be compared. Participants were given a detailed list of common caffeine-containing products to abstain from in the 48-h prior to each visit. They were also instructed to refrain from exercise, alcohol and diuretic intake and consume a standardized meal 24-h prior to testing. Participants began exercise in a euhydrated state as measured by urine specific gravity (no individual value, >1.020 g/ml). After the urine collection, participants were administered either the CAF or PLA solution. In the caffeine trial, participants consumed 6 mg/kg body weight of caffeine (Sigma-Aldrich, W22 240-2) dissolved in 3 ml/kg body weight of grapefruit-flavoured water. The placebo consisted of the same volume of grapefruit-flavoured water but with no caffeine. After 45 min of sitting quietly in the laboratory, nude body mass was assessed, and resting saliva and blood samples were taken (PRE). Subjects then entered the environmental chamber and ran on a motor-driven treadmill at a constant speed equivalent to 50% VO2max for 30 min. Participants were allowed to drink water ad libitum throughout the exercise bout. Upon completion, blood and saliva samples (MID) were taken before the second exercise bout was conducted at 70% VO2max for 30 min. Further blood and saliva samples were taken immediately upon cessation of exercise (POST) and 20 min following this second exercise bout (20 min POST). Core body temperature was monitored throughout using a rectal thermister connected to a digital thermometer (YSI Precision, Yellow Springs, OH, USA), and heart rate was measured by telemetry (Polar Electro, Kempele, Finland).
Blood and saliva sample collection and handling
Blood samples were collected by venepuncture of an antecubital vein using a 23-gauge butterfly needle (Venisystems, Abbott Ireland, Republic of Ireland) into a 4-ml EDTA coated vacutainer (Becton Dickinson & Co., UK). Whole blood was analysed for haemoglobin (Hemocue, UK) and haematocrit (capillary tube method) for determination of plasma volume change for which plasma Hsp72 and catecholamine concentrations were corrected (Dill and Costill 1974). The remaining blood was centrifuged at 1,500×g at 4°C for 10 min with the plasma aspirated into Eppendorf tubes and immediately frozen at −80°C until analysis. Unstimulated saliva samples were collected from participants using a passive drool technique described previously (Oliver et al. 2008). Briefly, participants were seated quietly and were instructed to rinse their mouth with water before swallowing in order to standardise the residual amount of saliva prior to collection. A pre-weighed saliva collection container was placed to the lips, and whilst leaning forwards and with their head down, saliva was allowed to passively collect (without spitting) into the container for approximately 3–6 min depending on pre-determined individual flow rates. By assuming the density of saliva to be 1.00 g/ml, saliva flow rate was calculated by dividing the volume collected by the time of collection. Saliva was sterile filtered to remove bacteria and mucosal cell contamination using Millex-GV 0.22 μm pore size filters (Millipore, USA) and frozen at −80°C.
Plasma and saliva sample analysis
On the day of assay, saliva and plasma samples were thawed and centrifuged prior to analysis of Hsp72 concentrations using a commercially available high-sensitivity sandwich ELISA (EKS-715, Assay Designs Inc., USA) that we have described previously (Fortes and Whitham 2009). Individual participant's plasma and saliva samples for both the caffeine and placebo trials were always assayed on the same plate. Since this was the first study to measure salivary Hsp72 using this assay, we performed spike and recovery and dilution studies in order to confirm the assay's accuracy. To overcome matrix effects, the assay standards were prepared in the assay diluent with the addition of human saliva with a known very low endogenous amount of Hsp72 at a ratio of 1:4. The recovery of spiked and diluted saliva samples was generally satisfactory (110%), providing confidence with regard to the use of this ELISA in determining salivary Hsp72 concentrations. Salivary Hsp72 is reported both as an absolute concentration (nanograms per millilitre) and as a secretion rate (nanograms per minute) by multiplying the concentration by the flow rate. To confirm that caffeine supplementation increased sympathetic activity, EDTA plasma samples were analysed for concentrations of epinephrine and norepinephrine using a commercially available ELISA (CatCombi RE 59242, IBL, Hamburg, Germany). Saliva samples were also assessed for total protein concentration (BCA protein assay kit 23227, Thermo Scientific, USA) and α-amylase activity (Cat number 1–1,902, Salimetrics, USA). All biochemical analyses were performed in duplicate and at room temperature. Intra-assay coefficient of variations was as follows: 3.8% for salivary α-amylase, 2.9% for salivary total protein, 3.2% for plasma epinephrine, 4.3% for plasma norepinephrine, 4.2% for salivary Hsp72 and 2.7% for plasma Hsp72.
Statistical analysis
The data were analysed using two-way fully repeated measures ANOVA with the Greenhouse–Geisser correction applied where appropriate. Significant F tests were followed up using Tukey's honestly significant difference (HSD) post-hoc test. To evaluate the meaningfulness of the differences, eta-squared effect sizes (η2) were also calculated for each variable. Eta-squared denotes the proportion of the total variance attributed to the independent factor and can be interpreted with values of 0.01, 0.06 and >0.14 indicating a small, medium and large effect, respectively (Cohen 1988). All data were analysed using SPSS version 12 software, and all data are reported as means ± SEM.
Results and discussion
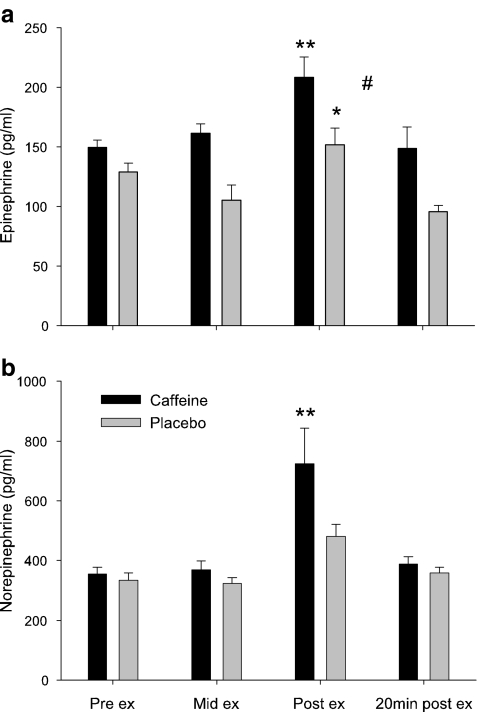
The purposes of this study were to explore the effects of exercise stress upon salivary eHsp72 expression, to investigate the relationship between salivary and plasma eHsp72 concentrations and to gain further insight as to the role of adrenergic activity on salivary Hsp72 expression. Administering caffeine prior to exercise increased both plasma epinephrine (main effect of trial, P < 0.01, η2 = 0.35, Fig. 1a) and norepinephrine concentrations during exercise (main effect of time, P < 0.01, η2 = 0.47, Fig. 1b). As such, we are confident that caffeine supplementation in the quantity administered in the current study (6 ml/kg body mass) was sufficient to enhance sympathetic activity during exercise and provide a useful model with which to investigate the potential sources of salivary Hsp72.
Fig. 1.
Plasma volume change corrected epinephrine (a) and norepinephrine (b) responses to caffeine supplementation (black bars) and placebo (gray bars) during progressive exercise in hot (30°C) conditions. Values are means ± SEM. **P < 0.01, significantly different from pre-exercise, mid-exercise and 20 min post-exercise value; *P < 0.05, significantly different from 20 min post-exercise; #P < 0.05, main effect of trial; n = 6
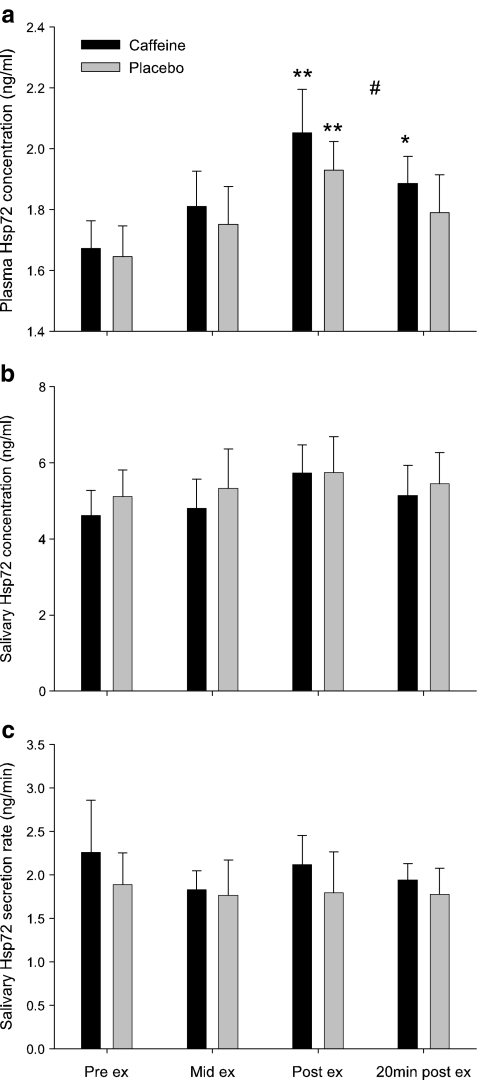
Consistent with previous research (Whitham et al. 2006), exercise resulted in a significant increase in plasma concentration of Hsp72 under both CAF and PLA conditions (main effect of time, P < 0.001, η2 = 0.56) with greater concentrations during CAF than PLA (main effect of trial, P < 0.05, η2 = 0.07, Fig. 2a). The additive effects of exercise and the large increase in core temperature (∼1.9°C) are liable to evoke a significant sympathetic response and Hsp72 release, particularly since Hsp72 concentration during exercise is both duration and intensity dependent (Fehrenbach et al. 2005), related to heat storage (Whitham et al. 2007) and related to sympathetic stimulation of adrenergic receptors (Johnson et al. 2005). To our knowledge, this is the first study to investigate the effect of a physical stressor on salivary Hsp72. Although Hsp72 was detected in all saliva samples, concentrations remained unaffected by exercise alone or exercise with caffeine supplementation (no time × trial interaction, P = 0.815, η2 = 0.02, Fig. 2b). Saliva flow rate remained unchanged with exercise during both trials (no time × trial interaction, P = 0.868, η2 = 0.01, Table 2), as such, no effect of exercise or caffeine administration was observed for salivary Hsp72 secretion rate (P = 0.901, η2 = 0.01; Fig. 2c). Moreover, Pearson's correlational analysis failed to show a significant positive relationship between individual saliva and plasma Hsp72 concentrations (Table 1). The failure of salivary Hsp72 concentration to change during a stressor such as exercise is surprising since it has been suggested that salivary Hsp72 may originate as a passive filtrate from blood plasma (Fabian et al. 2007) and as the current study and others (Walsh et al. 2001; Febbraio et al. 2002; Fehrenbach et al. 2005) have demonstrated a robust increase in plasma Hsp72 concentration following exercise. Additionally, photo-acoustic and chewing-evoked stimulation have both altered salivary Hsp72 expression (Fabian et al. 2003, 2004). It may be argued that the exercise intensity (50 and 70% VO2max) was not great enough to elicit changes in salivary Hsp72 in endurance-trained males. However, as a result of exercising in hot and humid conditions, mean heart rate at the end of the second exercise bout was 181 ± 7 BPM that equated to ∼91% HRmax, and final core temperature was ∼39.0 ± 0.3°C. Thus, the exercise conditions were deemed to be sufficiently stressful that resulted in significant increases in plasma Hsp72 concentration. It may also be argued that the small sample size in the current study could result in inadequate power to detect changes in salivary Hsp72. However, the variability in salivary Hsp72 expression in addition to small eta-squared effect sizes for salivary Hsp72 concentration and secretion rates suggests that the lack of change in these variables with exercise stress is a genuine finding. Indeed, this independence of salivary Hsp72 to plasma Hsp72 is not unique to saliva since exercise also fails to increase the concentration of Hsp72 within the cerebrospinal fluid (Steensberg et al. 2006).
Fig. 2.
Effect of caffeine supplementation (black bars) and placebo (gray bars) upon plasma volume change corrected eHsp72 concentration (a), salivary Hsp72 concentration (b) and salivary Hsp72 secretion rate (c) during progressive exercise in hot (30°C) conditions. Values are means ± SEM. Significantly different from pre-exercise value (**P < 0.01; *P < 0.05). #P < 0.05, main effect of trial; n = 6
Table 2.
Effect of caffeine supplementation (CAF) and placebo (PLA) before, during and after progressive exercise upon saliva flow rate, total protein concentration and salivary α-amylase concentration
| Pre-ex | Mid-ex | Post-ex | 20 min post-ex | |
|---|---|---|---|---|
| Saliva flow rate (ml/min) | ||||
| CAF | 0.48 (0.07) | 0.40 (0.05) | 0.38 (0.06) | 0.41 (0.06) |
| PLA | 0.37 (0.04) | 0.32 (0.07) | 0.31 (0.07) | 0.34 (0.07) |
| Total protein concentration (μg/ml) | ||||
| CAF | 969 (85) | 1,450 (200) | 2,476 (341)**, **** | 1,779 (303)* |
| PLA | 1,175 (128) | 1,060 (48) | 1,561 (202) | 1,730 (383) |
| Salivary α-amylase concentration (U/ml) | ||||
| CAF | 120 (29) | 128 (25)*** | 284 (39)**, **** | 209 (31)**, **** |
| PLA | 115 (27) | 74 (13) | 180 (29)** | 141 (35) |
Values are means (±SEM), n = 6
*P < 0.05; **P < 0.01 (significantly different from pre-exercise value); ***P < 0.05; ****P < 0.01 (CAF significantly greater than PLA)
Table 1.
Within subject correlations between plasma and salivary Hsp72 concentration during progressive exercise in hot conditions (30°C, 40% RH)
| Participant number | ||||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| r | 0.48 | 0.45 | 0.05 | 0.35 | 0.06 | −0.09 |
| P | 0.232 | 0.264 | 0.907 | 0.393 | 0.892 | 0.825 |
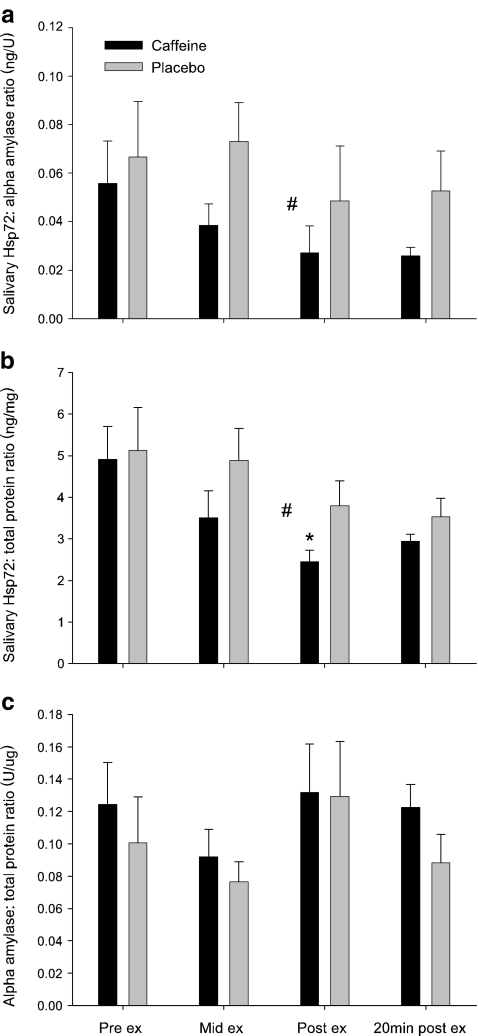
It has been proposed that salivary Hsp72 may originate from one of five sources: (1) salivary glands, (2) mucosal cells, (3) periodontal tissues, (4) mucosal lesions (both of these as exudated blood serum or as direct blood contamination) and (5) bacteria (Fabian et al. 2007). To date, only the first of these has been investigated. Fabian et al. (2003, 2004) suggest that salivary Hsp72 release occurred via a mechanism independent of the traditional secretory route of salivary proteins. Data from the current study is in support of this hypothesis for several reasons. Firstly, salivary Hsp72 concentration and secretion rate were not affected by exercise, whereas the main secretory protein of saliva, α-amylase along with total protein concentration (both of which are released in large quantities after exercise (Walsh et al. 1999)), increased significantly (significant time × trial interaction, P < 0.05, η2 = 0.09 and P < 0.01, η2 = 0.08 for α-amylase and total protein respectively, Table 2). Secondly, the additive effect of caffeine supplementation with exercise failed to augment salivary Hsp72, in contrast to α-amylase and total protein concentration, both of which were increased significantly above placebo (Table 2). Finally, the salivary Hsp72:α-amylase ratio and salivary Hsp72:total protein ratio were significantly lower during CAF than PLA (main effect for trial, P < 0.05, η2 = 0.14 and P < 0.01, η2 = 0.09 respectively, Fig. 3a, b), suggesting that salivary Hsp72 is not released in direct proportion to either α-amylase or total protein. Conversely, caffeine had no effect on the α-amylase:total protein ratio (no time × trial interaction, P = 0.730, η2 = 0.01, Fig. 3c). Taken together, these data reinforce previous findings that salivary Hsp72 is released via an independent mechanism to that of α-amylase, which is released from acinar cells predominantly upon sympathetic stimulation (Nater et al. 2006; Fabian et al. 2007).
Fig. 3.
Effect of caffeine supplementation (black bars) and placebo (gray bars) upon the salivary Hsp72:α-amylase ratio (a), salivary Hsp72:total protein ratio (b) and the salivary α-amylase:total protein ratio (c) during progressive exercise in hot (30°C) conditions. Values are means ± SEM. *P < 0.05, significantly different from pre-exercise value; #P < 0.05, main effect of trial, PLA > CAF; n = 6
Saliva secretion lies predominantly under parasympathetic control, although increased sympathetic input results in considerably increases in protein secretion. For example, previous in vivo research in exercising humans has shown that caffeine-induced sympathetic stimulation increases salivary IgA and α-amylase concentration (Bishop et al. 2006), whilst graded electrical stimulation of sympathetic nerves increases the secretion of peroxidase, IgA, α-amylase and total protein from rodent parotid and submandibular glands (together, these glands account for approximately 85–90% of all secretions into saliva; Asking 1985; Skov et al. 1988; Anderson et al. 1995; Carpenter et al. 2000). Importantly, neuro-endocrine control of saliva secretion is not identical in all salivary glands. For example, in humans, parotid and submandibular gland secretion is regulated primarily by adrenergic innervation, whereas sublingual and minor mucous gland secretion is initiated upon cholinergic (acetylcholine) stimulation (Chicharro et al. 1998; Anderson et al. 1995). Cholinergic stimulation controls the majority of fluid and mucin secretion into saliva (Garrett 1987). Acetylcholine released from parasympathetic nerves innervating the salivary glands acts upon the muscarinic cholinergic receptors, M1 and M3 (Gautam et al. 2004), and subsequent fluid and electrolyte secretion results from an increase in the intra-cellular calcium concentration (Melvin et al. 2005). Hsp72 is present in and released from epithelial cells (Broquet et al. 2003) and also released from a number of haematopoetic cells via exosomes (Clayton et al. 2005; Lancaster and Febbraio 2005). Since exosome release appears to be related to the degree of rise in intracellular calcium concentrations (Savina et al. 2003), this provides a potential mechanism for parasympathetic stimulated continual release of Hsp72 into saliva. Therefore, it is tangible that salivary Hsp72 might originate via parasympathetic innervation of the minor mucous or sublingual glands. Future research should investigate Hsp72 expression within ductal saliva from these glands, or focus on the effect of anticholinergic agents on salivary Hsp72 in human volunteers, or specifically of atropine administration in rodent models since atropine is a competitive antagonist of muscarinic cholinergic receptors.
One of the most striking findings to come from the current study was the greater concentration of Hsp72 within saliva (5.1 ± 0.8 ng/ml) compared to that in plasma (1.8 ± 0.1 ng/ml), an effect confirmed by the fact that salivary Hsp72 concentration was greater than plasma Hsp72 in all participants at every time point. This finding is unlikely to be attributable to differences between ELISAs or in the methodological protocols since both saliva and plasma samples for the same participant were always assayed on the same plate using the same reagents. This suggests that passive exudate from plasma or direct blood contamination does not contribute significantly to salivary Hsp72 expression. It is also unlikely that bacterial-derived Hsp72 contributes to the salivary Hsp72 measured in the current study due to the sterile filtration process and the extremely low cross-reactivity of the ELISA to the bacterial (Escherichia coli) homologue of Hsp72 (DnaK). Saliva acts as a first line of defence against pathogenic threat and contains numerous proteins with antibacterial and antiviral functions such as peroxidases, lysozymes, agglutinins, lactoferrin and immunoglobulins (Tenovuo 2004). Since Hsp72 may prevent bacterial attachment to mucosal surfaces (Boulanger et al. 1995), its chemoattractant ability (Ortega et al. 2008), its involvement in complement activation (Prohaszka et al. 2002) and owing to its roles in cellular protection and wound repair (Kovalchin et al. 2006), the continual presence of Hsp72 within the saliva at such high concentrations might serve to enhance periodontal defence and repair (Fabian et al. 2007). Chewing-evoked stimulation increases saliva flow rate and salivary Hsp72 output (Fabian et al. 2003). Thus, parasympathetic stimulated increases in flow rate (and potentially salivary Hsp72 output) may augment oral defence during food ingestion or dental brushing, when the oral cavity is perhaps at its greatest threat from microbial challenge. Clearly, future studies to investigate the immune functions of salivary Hsp72 are warranted.
In summary, we have demonstrated that Hsp72 concentrations within saliva are significantly greater than but not reflective of plasma concentrations, and that a physical stressor such as exercise does not alter the concentration of salivary Hsp72. Thus, salivary Hsp72 should not be used as a proxy measure of plasma Hsp72. Furthermore, it is likely that the appearance of Hsp72 within the saliva occurs independent of α-amylase, via a non-adrenergic mechanism. We have speculated that salivary Hsp72 may be released via cholinergic stimulation, although further research is required to clarify this.
Acknowledgments
We would like to thank Stephen O'Hara for his help with data collection and Dr. Samuel Oliver for reviewing a draft of this manuscript.
Conflicts of interest The authors declare that there are no conflicts of interest.
References
- Abe M, Manola JB, Oh WK, Parslow DL, George DJ, Austin CL, Kantoff PW. Plasma levels of heat shock protein 70 in patients with prostate cancer: a potential biomarker for prostate cancer. Clin Prostate Cancer. 2004;3:49–53. doi: 10.3816/cgc.2004.n.013. [DOI] [PubMed] [Google Scholar]
- Anderson LC, Garrett JR, Zhang X, Proctor GB, Shori DK. Differential secretion of proteins by rat submandibular acini and granular ducts on graded autonomic nerve stimulations. J Physiol. 1995;485:503–511. doi: 10.1113/jphysiol.1995.sp020746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asking B. Sympathetic stimulation of amylase secretion during a parasympathetic background activity in the rat parotid gland. Acta Physiol Scand. 1985;124:535–542. doi: 10.1111/j.1748-1716.1985.tb00045.x. [DOI] [PubMed] [Google Scholar]
- Bishop NC, Walker GJ, Scanlon GA, Richards S, Rogers E. Salivary IgA responses to prolonged intensive exercise following caffeine ingestion. Med Sci Sports Exerc. 2006;38:513–519. doi: 10.1249/01.mss.0000187412.47477.ee. [DOI] [PubMed] [Google Scholar]
- Boulanger J, Faulds D, Eddy EM, Lingwood CA. Members of the 70 kDa heat shock protein family specifically recognize sulfoglycolipids: role in gamete recognition and mycoplasma-related infertility. J Cell Physiol. 1995;165:7–17. doi: 10.1002/jcp.1041650103. [DOI] [PubMed] [Google Scholar]
- Broquet AH, Thomas G, Masliah J, Trugnan G, Bachelet M. Expression of the molecular chaperone Hsp70 in detergent-resistant microdomains correlates with its membrane delivery and release. J Biol Chem. 2003;278:21601–21606. doi: 10.1074/jbc.M302326200. [DOI] [PubMed] [Google Scholar]
- Carpenter GH, Proctor GB, Anderson LC, Zhang XS, Garrett JR. Immunoglobulin A secretion into saliva during dual sympathetic and parasympathetic nerve stimulation of rat submandibular glands. Exp Physiol. 2000;85:281–286. doi: 10.1017/S0958067000019680. [DOI] [PubMed] [Google Scholar]
- Chicharro JL, Lucia A, Perez M, Vaquero AF, Urena R. Saliva composition and exercise. Sports Med. 1998;26:17–27. doi: 10.2165/00007256-199826010-00002. [DOI] [PubMed] [Google Scholar]
- Clayton A, Turkes A, Navabi H, Mason MD, Tabi Z. Induction of heat shock proteins in B-cell exosomes. J Cell Sci. 2005;118:3631–3638. doi: 10.1242/jcs.02494. [DOI] [PubMed] [Google Scholar]
- Cohen J. Statistical power analysis for the behavioural sciences. 2. Mahwah: Lawrence Erlbaum Associates; 1988. [Google Scholar]
- Dill DB, Costill DL. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J Appl Physiol. 1974;37:247–248. doi: 10.1152/jappl.1974.37.2.247. [DOI] [PubMed] [Google Scholar]
- Fabian TK, Gaspar J, Fejerdy L, Kaan B, Balint M, Csermely P, Fejerdy P. Hsp70 is present in human saliva. Med Sci Monit. 2003;9:BR62–BR65. [PubMed] [Google Scholar]
- Fabian TK, Toth Z, Fejerdy L, Kaan B, Csermely P, Fejerdy P. Photo-acoustic stimulation increases the amount of 70 kDa heat shock protein (Hsp70) in human whole saliva. A pilot study. Int J Psychophysiol. 2004;52:211–216. doi: 10.1016/j.ijpsycho.2003.10.004. [DOI] [PubMed] [Google Scholar]
- Fabian TK, Fejerdy P, Nguyen MT, Soti C, Csermely P. Potential immunological functions of salivary Hsp70 in mucosal and periodontal defense mechanisms. Arch Immunol Ther Exp Warsz. 2007;55:91–98. doi: 10.1007/s00005-007-0012-z. [DOI] [PubMed] [Google Scholar]
- Febbraio MA, Ott P, Nielsen HB, Steensberg A, Keller C, Krustrup P, Secher NH, Pedersen BK. Exercise induces hepatosplanchnic release of heat shock protein 72 in humans. J Physiol. 2002;544:957–962. doi: 10.1113/jphysiol.2002.025148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fehrenbach E, Niess AM, Voelker K, Northoff H, Mooren FC. Exercise intensity and duration affect blood soluble HSP72. Int J Sports Med. 2005;26:552–557. doi: 10.1055/s-2004-830334. [DOI] [PubMed] [Google Scholar]
- Fortes MB, Whitham M. No endogenous circadian rhythm in resting plasma Hsp72 concentration in humans. Cell Stress Chaperones. 2009;14:273–280. doi: 10.1007/s12192-008-0082-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrett JR. The proper role of nerves in salivary secretion: a review. J Dent Res. 1987;66:387–397. doi: 10.1177/00220345870660020201. [DOI] [PubMed] [Google Scholar]
- Gautam D, Heard TS, Cui Y, Miller G, Bloodworth L, Wess J. Cholinergic stimulation of salivary secretion studied with M1 and M3 muscarinic receptor single- and double-knockout mice. Mol Pharmacol. 2004;66:260–267. doi: 10.1124/mol.66.2.260. [DOI] [PubMed] [Google Scholar]
- Horn P, Kalz A, Lim CL, Pyne D, Saunders P, Mackinnon L, Peake J, Suzuki K. Exercise-recruited NK cells display exercise-associated eHSP-70. Exerc Immunol Rev. 2007;13:100–111. [PubMed] [Google Scholar]
- Johnson JD, Campisi J, Sharkey CM, Kennedy SL, Nickerson M, Fleshner M. Adrenergic receptors mediate stress-induced elevations in extracellular Hsp72. J Appl Physiol. 2005;99:1789–1795. doi: 10.1152/japplphysiol.00390.2005. [DOI] [PubMed] [Google Scholar]
- Kampinga HH, Hageman J, Vos MJ, Kubota H, Tanguay RM, Bruford EA, Cheetham ME, Chen B, Hightower LE. Guidelines for the nomenclature of the human heat shock proteins. Cell Stress Chaperones. 2008;14:105–111. doi: 10.1007/s12192-008-0068-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovalchin JT, Wang R, Wagh MS, Azoulay J, Sanders M, Chandawarkar RY. In vivo delivery of heat shock protein 70 accelerates wound healing by up-regulating macrophage-mediated phagocytosis. Wound Repair Regen. 2006;14:129–137. doi: 10.1111/j.1743-6109.2006.00102.x. [DOI] [PubMed] [Google Scholar]
- Lancaster GI, Febbraio MA. Exosome-dependent trafficking of HSP70: a novel secretory pathway for cellular stress proteins. J Biol Chem. 2005;280:23349–23355. doi: 10.1074/jbc.M502017200. [DOI] [PubMed] [Google Scholar]
- Melvin JE, Yule D, Shuttleworth T, Begenisich T. Regulation of fluid and electrolyte secretion in salivary gland acinar cells. Annu Rev Physiol. 2005;67:445–469. doi: 10.1146/annurev.physiol.67.041703.084745. [DOI] [PubMed] [Google Scholar]
- Nater UM, Marca R, Florin L, Moses A, Langhans W, Koller MM, Ehlert U. Stress-induced changes in human salivary alpha-amylase activity—associations with adrenergic activity. Psychoneuroendocrinology. 2006;31:49–58. doi: 10.1016/j.psyneuen.2005.05.010. [DOI] [PubMed] [Google Scholar]
- Oliver SJ, Laing SJ, Wilson S, Bilzon JL, Walsh NP. Saliva indices track hypohydration during 48 h of fluid restriction or combined fluid and energy restriction. Arch Oral Biol. 2008;53:975–980. doi: 10.1016/j.archoralbio.2008.05.002. [DOI] [PubMed] [Google Scholar]
- Ortega E, Hinchado MD, Martin-Cordero L, Asea A. The effect of stress-inducible extracellular Hsp72 on human neutrophil chemotaxis: a role during acute intense exercise. Stress. 2008;12:240–249. doi: 10.1080/10253890802309853. [DOI] [PubMed] [Google Scholar]
- Pittet JF, Lee H, Morabito D, Howard MB, Welch WJ, Mackersie RC. Serum levels of Hsp 72 measured early after trauma correlate with survival. J Trauma. 2002;52:611–617. doi: 10.1097/00005373-200204000-00001. [DOI] [PubMed] [Google Scholar]
- Prohaszka Z, Singh M, Nagy K, Kiss E, Lakos G, Duba J, Fust G. Heat shock protein 70 is a potent activator of the human complement system. Cell Stress Chaperones. 2002;7:17–22. doi: 10.1379/1466-1268(2002)007<0017:HSPIAP>2.0.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savina A, Furlan M, Vidal M, Colombo MI. Exosome release is regulated by a calcium-dependent mechanism in K562 cells. J Biol Chem. 2003;278:20083–20090. doi: 10.1074/jbc.M301642200. [DOI] [PubMed] [Google Scholar]
- Skov OP, Kirkegaard P, Rasmussen T, Magid E, Poulsen SS, Nexo E. Adrenergic effects on secretion of amylase from the rat salivary glands. Digestion. 1988;41:34–38. doi: 10.1159/000199729. [DOI] [PubMed] [Google Scholar]
- Steensberg A, Dalsgaard MK, Secher NH, Pedersen BK. Cerebrospinal fluid IL-6, HSP72, and TNF-alpha in exercising humans. Brain Behav Immun. 2006;20:585–589. doi: 10.1016/j.bbi.2006.03.002. [DOI] [PubMed] [Google Scholar]
- Tenovuo J. Protective functions of saliva. In: Edgar M, O'Mullane D, Dawes C, editors. Saliva and oral health. London: British Dental Journal; 2004. pp. 103–119. [Google Scholar]
- Walsh NP, Blannin AK, Clark AM, Cook L, Robson PJ, Gleeson M. The effects of high-intensity intermittent exercise on saliva IgA, total protein and alpha-amylase. J Sports Sci. 1999;17:129–134. doi: 10.1080/026404199366226. [DOI] [PubMed] [Google Scholar]
- Walsh RC, Koukoulas I, Garnham A, Moseley PL, Hargreaves M, Febbraio MA. Exercise increases serum Hsp72 in humans. Cell Stress Chaperones. 2001;6:386–393. doi: 10.1379/1466-1268(2001)006<0386:EISHIH>2.0.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitham M, Walker GJ, Bishop NC. Effect of caffeine supplementation on the extracellular heat shock protein 72 response to exercise. J Appl Physiol. 2006;101:1222–1227. doi: 10.1152/japplphysiol.00409.2006. [DOI] [PubMed] [Google Scholar]
- Whitham M, Laing SJ, Jackson A, Maassen N, Walsh NP. Effect of exercise with and without a thermal clamp on the plasma heat shock protein 72 response. J Appl Physiol. 2007;103:1251–1256. doi: 10.1152/japplphysiol.00484.2007. [DOI] [PubMed] [Google Scholar]
- Wright BH, Corton JM, El Nahas AM, Wood RF, Pockley AG. Elevated levels of circulating heat shock protein 70 (Hsp70) in peripheral and renal vascular disease. Heart Vessels. 2000;15:18–22. doi: 10.1007/s003800070043. [DOI] [PubMed] [Google Scholar]