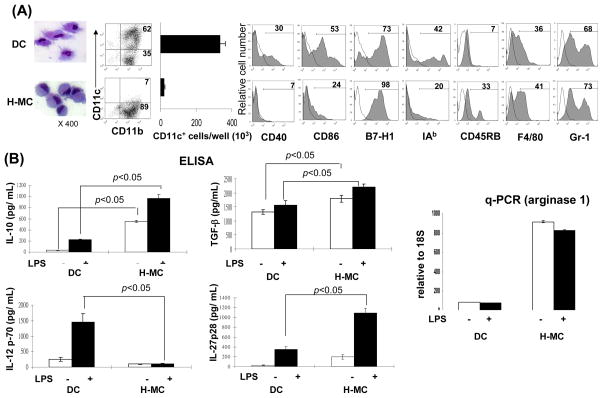
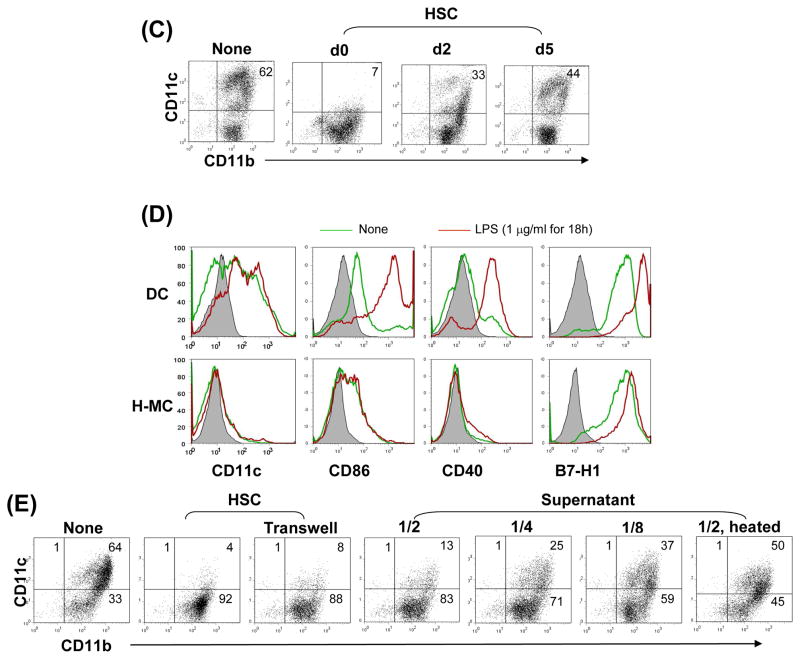
Figure 5. Impact of HSC on BM-derived DC propagation in vitro.
HSC (B6) were added (HSC:BM cells = 1:80) at the beginning of the culture of B6 BM cells (2 × 106/well) in the presence of mouse rGM-CSF (8 ng/ml) for 5 days. The floating cells were harvested, washed, and re-suspended in RPMI 1640 medium (H-MC). The culture in the absence of HSC served as control (DC). (A) Cells were stained with Giemsa for morphology examination. Cells were stained for CD11b, CD11c and indicated key surface molecules, and analyzed by flow cytometry. The absolute numbers of CD11c+ cells/well were calculated (n=3), expressed as mean ± 1SD. The flow histograms show the expression of the indicated key surface molecules. The number is percentage in CD11b+ cell population. (B) The levels of IL-10, IL-12p70, TGF-β and IL-27p28 were measured in culture supernatant by ELISA. Expression of arginase 1 mRNA was determined by q-PCR. For further stimulation, the cells were exposed to LPS (1μg/ml) for the last 18 hours of culture. (C) Time course of HSC effect. HSC were added on day 0, 2 or 5 of BM cell culture. Cells were harvested on day 7 and analyzed for expression of CD11b and CD11c by flow cytometry. (D) H-MC are resistant to maturation. DC or H-MC were exposed to LPS (1μg/ml) for 18 hours. Expression of CD11c and the indicated co-stimulatory molecules was analyzed by flow cytometry, and displayed as histograms gated on CD11b+ cells. (E) Induction of H-MC is mediated by soluble factor (s). BM cells and HSC were cultured in transwell or regular plates. Expression of CD11b and CD11c was analyzed by flow cytometry. Comparable CD11b+CD11c− cells were generated in transwell plate comparable to regular one. This was reexamined by addition of HSC culture supernatant into BM cell culture at various concentrations (1/2, 1/4 or 1/8 of total culture medium volume). The generation of CD11b+CD11c− cells was correlated with concentrations of the added supernatant. To determine the nature of the soluble factor (s), HSC culture supernatant treated at 57°C for 30 minutes failed to induce CD11b+CD11c− cells. The data are representative of three separate experiments.