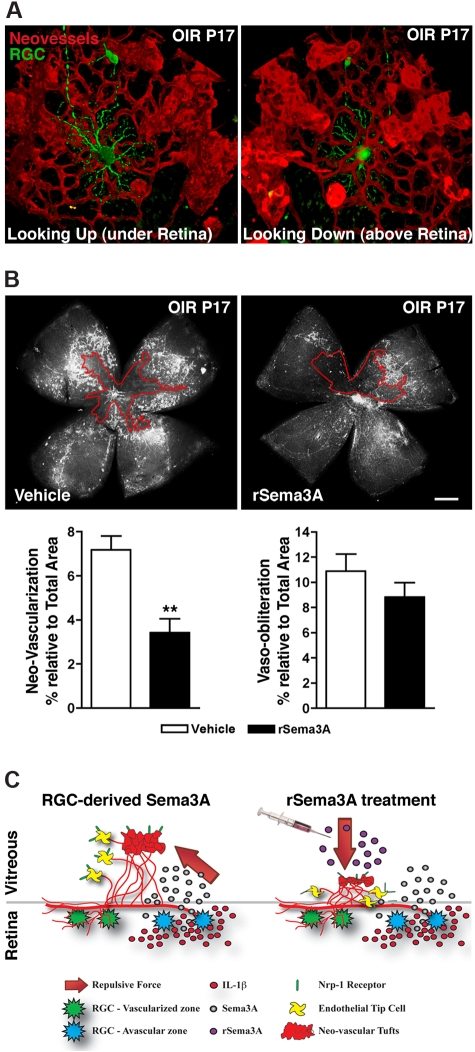
Figure 7.
Intravitreal delivery of rSema3A suppresses pre-retinal neovascularization in OIR. (A) 3D reconstructions of pathologic neo-vessels and RGC-YFP at P17 after OIR. The spatial distribution of retinal neurons and vessels results in the repulsion of neovascular tufts toward the vitreous. (B) Intravitreal injection of rSema3A (100 ng; P14) halved the formation of preretinal vascular tufts at P17. n = 7; **P = .0012 compared with corresponding vehicle. (C) Schematic summary illustrates ischemic neurons in the avascular zones, producing Sema3A secondary to inflammatory stress (IL-1β). Pathologic neovascular tufts are enriched in Nrp-1. RGC-derived Sema3A impedes revascularization and repels neo-vessels away from the avascular neural retina toward the vitreous (left), whereas intravitreal rSema3A (injected) prevents preretinal invasion of pathologic neovessels (right).