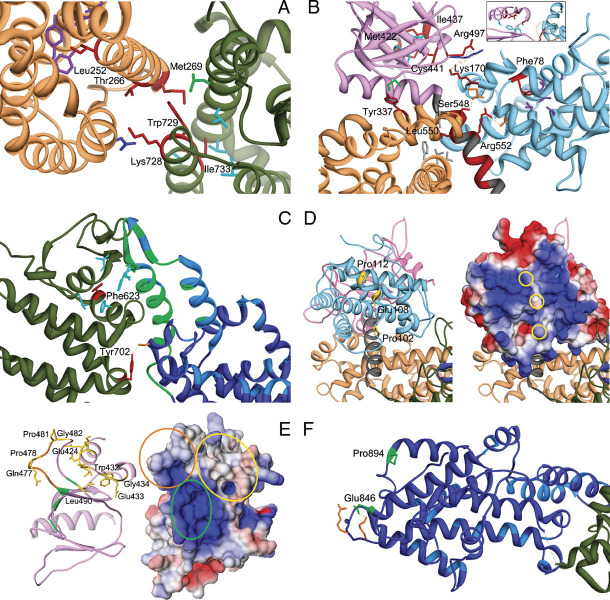
Figure 2.
Detailed analysis of structural perturbations resulting from Noonan syndrome-causing amino acid substitutions. A: Class 1 mutations affecting residues at the distal RAS binding site. The cartoon includes the DH (sandy brown) and REM (dark green) domains only. Affected residues are shown in red. The autoinhibitory binding network includes the hydrophobic interaction between residues Met269 (DH) and Trp729 (REM), both mutated in NS, and between the former and Leu687 (green). Leu690, Val697, Ile718, and Ile736 are hydrophobic residues (cyan) that interact with Ile733 (REM). Mutated residues Thr266 (DH) and Lys728 (REM) face each other. Substitution of Thr266 by lysine would create an electrostatic repulsion with Lys728. The unclassified variant Leu252 contributes to a hydrophobic core with residues Tyr215, Leu219, Ile249, Tyr295, and Tyr298 (purple), whose disruption is expected to perturb considerably the DH domain surface interacting with the REM domain. B: Class 1B mutations. The cartoon includes the HF (sky blue), DH (sandy brown), and PH (magenta) domains and the PH-REM helical linker (gray). Relevant affected residues are shown in red. Met422 and Ile437 (PH) participate in a hydrophobic bonding network with residues Ile425, Phe464, and Leu467 (cyan, see also the inset). Hydrophobic interaction between Tyr337 and Met538 (green) contributes to the binding network stabilizing the interaction between the PH and DH domains. Other interdomain interactions involve Leu550 and residues of the DH and PH domains, Val225, Leu221, Phe226, and Tyr546 (gray), Arg552, and residues of the HF domain, Asp140 and Asp169 (orange), Ser548 and Asp169, and Lys170, and residues of the PH domain, Arg497 and Lys498 (blue). Phe78 participates to the hydrophobic interaction involving residues of the HF domain core located close to the PH domain and PH-REM linker (Leu55, Leu59, Val74, Val133, and Ile137; purple). C: Class 1 mutations affecting the REM domain region (dark green) interacting with the CDC25 domain (blue). The helical hairpin (residues 929–978) implicated in the conformational switch (green) and residues interacting with RAS at the active site (light blue) are shown. Phe623 hydrophobically interact with Ile 601, Leu613, Phe627, Ile956, and Phe958 (cyan). The hydrogen bond between Tyr702 and Ser802 (orange) is also shown. D: Class 2 mutations affecting the HF domain. The left panel shows the HF (light blue), DH (sandy brown), and PH (plum) domains. The HF surface colored according to the electrostatic potential (from red at −3kT/e to blue at +3kT/e) is also shown (right panel). Mutations affect solvent exposed residues (yellow side chains, left panel; yellow circles, right panel) located in a region that has a positive electrostatic potential (right panel), and has been implicated in membrane binding. E: Class 2 mutations affecting the PH domain. The left panel includes the PH domain (plum) only. The PH surface, colored according to the electrostatic potential (from red at −5kT/e to blue at +5kT/e) is also shown (left panel). Affected residues are shown in yellow (side chains, left panel; circles, right panel). Residues that are predicted to bind to PIP2 [Zheng et al., 1997], Lys456, Arg459, Lys472, and Arg489, are shown in green (circled in the right panel), whereas the PA-interacting region (residues 472–483) [Zhao et al., 2007] is shown in orange (circled in the right panel). F: Class 3 mutations. The cartoon includes the REM (dark green) and CDC25 (blue) domains only (residues 567–1049). Affected residues are shown in green. Residues implicated in RAS binding at the catalytic site are shown (light blue). Glu846 and Pro894 are placed distally from the active site and regions implicated in the conformational rearrangement of the CDC25 domain. Glu846 electrostatically interacts with Arg1026 and Lys 1029 (orange).