Abstract
Background
Brain surgery faces important challenges when trying to achieve maximum tumor resection while avoiding post-operative neurological deficits.
Objective
In order for surgeons to have optimal intraoperative information concerning white matter anatomy, we developed a platform that allows the intra-operative real-time querying of tractography datasets during frameless stereotactic neuronavigation.
Methods
Structural magnetic resonance imaging (MRI), functional MRI, and diffusion tensor imaging (DTI) were performed on 5 patients before undergoing lesion resection using neuronavigation. During the procedure, the tracked surgical tool tip position was transferred from the navigation system to the 3D Slicer software package, which used this position to seed the white matter tracts around the tool tip location, rendering a geometric visualization of these tracts on the pre-operative images previously loaded onto the navigation system. The clinical feasibility of this approach was evaluated during five cases of lesion resection. In addition, system performance was evaluated by measuring the latency between surgical tool tracking and visualization of the seeded white matter tracts.
Results
Lesion resection was performed successfully in all five patients. The seeded white matter tracts close to the lesion and other critical structures, as defined by the functional and structural images, were interactively visualized during the intervention to determine their spatial relationships relative to the lesion and critical cortical areas. Latency between tracking and visualization of tracts was less than a second for fiducial radius size of 4–5mm.
Conclusion
Interactive tractography can provide an intuitive way of inspecting critical white matter tracts in the vicinity of the surgical region, allowing the surgeon to have increased intra-operative white matter information to execute the planned surgical resection.
Keywords: intra-operative tractography, white matter, real-time tractography, neuronavigation, DTI
Introduction
Brain surgery faces important challenges when trying to achieve maximum tumor resection while avoiding post-operative neurological deficits 1. Mounting evidence suggests that more extensive surgical resection is associated with an increase in time to tumor progression, a reduction in the incidence of recurrence, decreased risk for seizures, and an improved life expectancy for both low and high grade brain tumors 2–4. Moreover, new neurologic deficits associated with surgery worsen prognosis 5.
Differentiating the tumor from normal brain parenchyma during surgery can be challenging, as visual inspection alone is often insufficient. Tumors located near eloquent cortical areas or critical white matter (WM) fiber tracts pose particular challenges when deciding the extent of resection due to the uncertainty of how the tumors interact with these regions 6. Even if eloquent cortical anatomy has been respected, neurological deficits will occur if damage to critical WM tracts takes place 7. Furthermore, many primary brain tumors grow by infiltration of healthy tissue, meaning that there may be functional WM tracts within tumor boundaries 8.
High resolution structural Magnetic Resonance Imaging (MRI) can demonstrate anatomy and intra-cranial pathology and is routinely integrated into neuronavigation systems to guide surgery. Functional MRI (fMRI), positron emission tomography, and magnetic source imaging can help define functional cortical regions in the brain. However, these procedures are restricted to grey matter, and cannot reveal the spatial location of critical subcortical structures 9. Subcortical stimulation mapping can be performed during tumor resection to identify the motor pathways in deep WM structures, but the technique does not reveal the full three dimensional (3D) extent of the motor tract 10, 11, does not demonstrate the tract until it has been encountered surgically, and is technically very demanding.
Diffusion tensor magnetic resonance imaging (DTI) is presently the only technique that allows measurement of WM fiber orientation in the human brain in vivo. Based on the preferential diffusion of water along WM tracts, diffusion tensor MRI is able to demonstrate tissue microstructure12. This information can be used to generate a detailed 3D representation of the configuration of the WM tracts, using a process called tractography13, which in conjunction with other imaging techniques can show displacement, interruption, or infiltration of the WM tracts by the tumor 14, 15.
To cluster and isolate WM tracts of interest, tractography trajectories, referred to here as “fibers”, are often seeded or selected from points of interest. Manually identified regions of interest (ROI), such as contours or spheres, can be used to select specific relevant fibers that pass through them 16, 17. We recently developed a software tool that can display fibers that pass within a certain variable distance of the tumor, a cortical or subcortical location, an fMRI activation, or other similar segmented structures (see associated manuscript) 18. This tool can also allow the placement of a fiducial of variable size which seeds all the tracts passing through the fiducial volume. By interactively moving the location of the fiducial around the 3D dataset and changing its volume, tracts can be selected “on the fly” in order to display specific tracts of interest. By interactively querying the data, the surgeon can build a mental representation of the relevant WM tracts and their relationship to the lesion and critical anatomic structures.
Visualization of selected critical fiber tracts can be very useful during the intra-operative procedure, and there have been several intents to incorporate this information into the neuronavigation setup to improve decision-making during surgery 19–23. These studies involve pre-operatively selecting and clustering the critical tracts using ROI seeding methods before exporting it to the neuronavigation system for availability during surgery 19. These methods do not include the option of querying specific parts of the tracts intra-operatively or of showing only the tracts that are close to the tool tip location during surgery. This would be especially useful when correlating the spatial location of WM tracts based on intra-operative findings, such as electrocortical stimulation testing. Tractography-integrated neuronavigation has been shown to be a reliable way to map WM connections 24, 25.
This report describes the development of hardware and software tools which integrate tractography datasets into a commercial neuronavigation system and allow interactive real-time querying of the critical WM tracts during surgery. The tracked position of the neurosurgical tools during the intervention is exported from the clinical neuronavigation system to a research hardware station running 3D Slicer which allows the performance of the investigational image-processing. The position of the navigation tool tip is used to seed the WM tracts, providing visualization of the relevant fibers and their spatial configuration in the surgical field with respect to the lesion. The system was tested during frameless stereotactic surgery in five patients undergoing tumor resection, as well as bench tested for latency evaluation.
Materials and Methods
Subjects
Five patients undergoing surgery for brain tumors or cavernous malformation with neuronavigation were selected for an initial feasibility study for the intra-operative real-time querying of critical WM tracts in the surgical field. All subjects were recruited in accordance to the Institutional Review Board policies and gave written informed consent. Patients received pre-operative structural MRI, fMRI, and DTI. As required by clinical considerations and tumor location, fMRI was used to define motor, visual, somato-sensory, and/or language cortices. DTI was acquired for the whole brain in all subjects. Table 1 displays the demographic information and fMRI behavioral paradigms used for the subjects recruited for the study.
Table 1.
Demographic and fMRI scanning information of patients who underwent neurosurgical tumor resection with frameless stereotactic navigation integrated with real-time interactive tractography.
| Subject | Age | Sex | Type of Lesion | Lesion Location |
fMRI behavioral paradigm |
|---|---|---|---|---|---|
| 1 | 38 | M | High grade glial neoplasm with treatment effects |
Left parieto- occipital |
Whole field vision alternating checkerboard |
| 2 | 28 | F | Mixed Grade II glioma |
Right parieto- temporal |
Motor hand clenching and lip pursing and whole field vision |
| 3 | 35 | M | Glioblastoma multiforme |
Left frontal | Language vocalized antonym and noun generation tasks |
| 4 | 31 | F | Grade II astrocytoma |
Left frontal temporal |
Vocalized antonym and noun tasks for both primary and secondary languages, right hand clench, lip pursing |
| 5 | 25 | F | Cavernous angioma |
Right fronto- parietal |
Language antonym generation, lip pursing, left-hand clench |
Image Acquisition
All MR images were acquired with a 3T EXCITE Signa scanner (GE Medical Systems, Milwaukee, WI, USA) with Excite 14.0. Structural images using an 8 channel head coil and ASSET included a high resolution whole brain T1-weighted axial 3D Spatial Gradient Recalled Echo image (TR=7500ms, TE=30ms, matrix=256×256, FOV=25.6cm, flip angle=20°; 176 slices, voxel size=1×1×1mm). Either a high-resolution T2-weighted gradient-echo MR image (TR=7200 ms, TE=70 ms, flip angle=90°, matrix=256×256, 100 slices, voxel size=1×1×1.5mm) or a SPGR gadolinium enhanced image (TR=30ms, TE=5ms, flip angle=45°, matrix=224×224, voxel size = 0.94×0.94×1.4mm) were then acquired to demonstrate surgical pathology and for neuronavigation. Diffusion Weighted Imaging (DWI) for DTI was acquired with a multi-slice single shot echo-planar-imaging sequence (TR=14000ms, TE=80ms, 31 gradient directions with a b-value of 1000 s/mm2, and 1 baseline T2 image, FOV = 25.6cm, matrix=128×128, voxel size=2×2×2.6mm). A quadrature head coil and single-shot gradient-echo echo-planar imaging sequence was used to acquire BOLD functional images for fMRI (TR=2000ms, TE=40ms, flip angle=90°, FOV=25.6cm, dimensions = 128×128, slice gap=0mm, voxel size=2×2×4mm). In each image volume, 27 axial slices were acquired using an ascending interleaved scanning sequence. Behavioral paradigms were selected based on clinical considerations and included visual alternating checkerboard, paced motor tasks for hand, foot, and face/mouth, somato-sensory stimulation, and language tasks.
Image Analysis
The fMRI data acquired was realigned, analyzed, and motion corrected using SPM2 software (Welcome Department of Cognitive Neurology, Institute of Neurology, London, UK).
Most image processing and analysis, including registration, segmentation of critical structures, and tractography, was performed using the functions of the 3D Slicer software package (www.slicer.org), an open source image visualization and processing software developed at the Brigham and Women’s Hospital 26. In tissues such as WM, which have an oriented fibrous structure, water diffusion is anisotropic (varies with direction), and the direction(s) of fastest diffusion parallels the fibrous structure 27. DTI models the diffusion in each voxel in the brain using a tensor, the primary eigenvector of which represents the direction of least restricted diffusion and thus the orientation of the axons in WM 28. Intervoxel connectivity can be modeled by following the primary eigenvector, a process called tractography, which estimates the trajectories of WM tracts. Tractography for the entire brain was performed using the 2nd order Runge-Kutta method implemented in 3D Slicer. A single tensor model described the estimated WM tracts in the brain using a standard streamline tractography method 28 which repeatedly steps in the principal diffusion direction as defined by the tensor at each location. The visualization of the fibers can be color coded based on the fractional anisotropy parameter calculated from the diffusion tensor of each voxel.
Image-based registration between the different image types is required to account for patient motion during acquisition and image acquisition at different sessions. The method brings the different images into anatomical alignment. An affine registration technique, which can compensate for variations in scale, translation, rotation, and shear 29 between acquired images, proved to be a good method for brain alignment. It is based on an algorithm which looks to maximize the Mattes mutual information metric 30 between images, delivering a favorable trade-off between computation time and accuracy 31. This method is available in 3D Slicer using functions from the Insight Toolkit library (ITK, http://www.itk.org). The tumor and other anatomical structures of interest were manually segmented using standard functions in 3D Slicer software. The processed images were then transferred pre-operatively to the commercial neuronavigation system software in order to be displayed during the intervention. The BrainLAB VVCranial pre-surgical workstation running PatXfer 5.2 and iPlan 2.6.1 software (Brainlab AG, Feldkirchen, Germany) enabled image transfer, tumor segmentation, image fusion, and co-localization of multi-modality imaging 32.
Real-Time Interactive Tractography System - Overview
Several components, both in terms of hardware and software, are required to enable real-time interaction with WM tractography using a commercial neuronavigation system during surgery. Figure 1 displays an overview of the system set-up.
Figure 1.
System overview of neuronavigation with intra-operative seeding and visualization of WM tracts. The surgical tool tip position is tracked using BrainLAB VVCranial’s optical tracking technology and sent over a TCP/IP network protocol using VVLink. The BioImage Suite image processing and analysis software then converts the tracking data from the VVLink protocol to the OpenIGTLink protocol, and sends it over a TCP/IP network connection to the 3D Slicer software. The tracked tool tip position is then used to seed the fibers in the tractography dataset, allowing visualization of the relevant fibers in the surgical region of interest.
We connected our research interface software, 3D Slicer 3.3, which contains the algorithms and software modules which allow real-time interaction with white matter fiber bundles, to the VVCranial image guided surgical navigation system. The VVCranial system offers a client-server communication link, called VectorVision Link (VVLink 33), which allows the bidirectional transfer of data from the VVCranial system to external advanced image processing and visualization research software packages. In order to connect from the VVCranial system to 3D Slicer, the data exported using VVLink must be converted into OpenIGTLink, 3D Slicer’s own peer-to-peer network communication protocol architecture 34. This conversion was done using Yale BioImage Suite 35 which acted as a bridging software (Figure 1). With this set-up in place, position data from the tracked surgical instruments proceeding from the VVCranial neuronavigation system were transferred to 3D Slicer, and used to define a fiducial which will seed the tractography dataset, allowing visualization of white matter fibers in the vicinity of the surgical tool.
Real-Time Interactive Tractography System - Components
We used the BrainLAB VectorVision Cranial (VVCranial) image guided surgical navigation system (Brainlab AG, Feldkirchen, Germany) to perform frameless stereotactic neurosurgery. It provides navigation of the pre-surgical plan and can track surgical tool positions using optical technology (Polaris, NDI, Ontario, Canada). Patient registration takes place by either surface laser scanning 36 or fiducial-based registration markers. After successful registration, the VVCranial system tracks the location of the surgical instruments (e.g. pointer, suction device, microscope) during the procedure using an optical reference array consisting of a set of disposable reflective markers. Virtual representations of the tracked surgical instruments are displayed on the pre-operative images to provide frameless stereotactic navigation during the surgical procedure.
To export data from the VVCranial system, BrainLAB provides a proprietary communication protocol called VVLink 33, 37. It is a custom designed TCP/IP client/server architecture that enables data transfer of image data sets, visualizations, and tool positions in real-time between the VVCranial system and other image analysis software packages. VVLink was developed as a joint effort between Yale University, BrainLAB AG, and the Technical University of Munich, and is now available as part of the BrainLAB product line 33. This technology facilitates the communication between the commercial FDA approved device and a research platform, allowing the testing of novel features and algorithms in the clinical setting while isolating the normal and stable functioning of the navigation system during the routine clinical procedure.
3D Slicer receives data using the OpenIGTLink protocol, its own peer-to-peer network communication protocol architecture, which also allows for bi-directional data transmission 34. OpenIGTLink was designed to standardize the exchange of tracking data, images, and device control information among tracking devices, imaging scanners, and systems software with 3D Slicer. Since the VVCranial system does not have an OpenIGTLink compatible layer, bridging software is required to establish communication between the OpenIGTLink and VVLink protocols. The bridge was done through Yale BioImage Suite 34, 35, an integrated image analysis software suite which includes a VVLink interface. The bridging software receives images and tracking data from the VVCranial system using VVLink and converts them into OpenIGTLink messages, which are then sent to the 3D Slicer client over the network (Figure 1).
Once the coordinates of the tracked surgical instruments have been received by 3D Slicer, they are used in a custom developed 3D Slicer navigation software module. This module uses the position data of the tracked tool tip location to specify a 3D fiducial point which seeds the tractography dataset around the fiducial location. The size of the seeded area defined by the fiducial can be modified by the user, allowing a controlled exploration of the tractography dataset in the surgical region of interest. As the surgical tool is moved, so too are the fiducial seeds which update the visualized WM tracts. The 3D Slicer navigation module can display all the relevant pre-operative image information (MRI, fMRI, segmented structures) in order to provide an anatomic and functional reference frame for localization of the visualized WM tracts. The 3D Slicer navigation module also allows the tracked tool position data to be used as a reference position from which to define additional seed fiducials as required. A fiducial seed can be defined at an extended geometric location from the current tool tip position in order to display the WM tracts at a given depth from the tip of the instrument. This allows probing the surgical region “virtually” to visualize WM fibers before the anatomical area is resected.
System Hardware
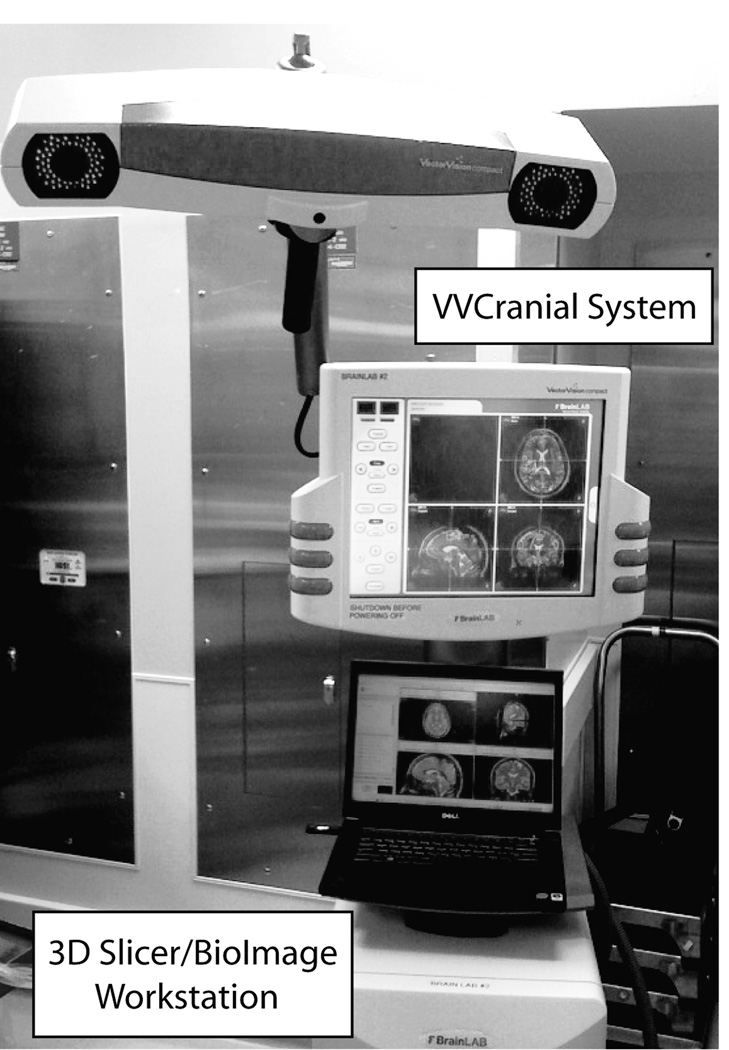
All data analysis and the real-time interactive tractography were performed on a mid-end workstation (Dell Precision T7400 64bit, Intel Dual Xeon processors E5420 at 2.5GHz, 4 Gb of RAM, nVidia Quadro FX570 graphics accelerator with 256 Mb dedicated memory, Linux Fedora 6 operating system), which runs both the BioImage Suite and 3D Slicer software. The VVCranial system has a high end graphic workstation (Intel P4 3.2GHz processors, 1GB of RAM, Windows XP) equipped with two infrared cameras for wireless passive marker optical tracking in the operating room. Both workstations are connected by a dedicated 1GB TCP/IP network link. Figure 2 shows an image of the hardware set-up in the OR, with both the VVCranial system and the workstation (laptop) which runs BioImage Suite and 3D Slicer.
Figure 2.
(a) Hardware set-up for the intra-operative interactive tractography navigation system. The image shows the VVCranial system and the workstation (laptop) required to run intra-operative tractography during a surgical intervention. (b) Set-up in the operating room showing two different displays corresponding to the output from the VVCranial navigation system and the 3D Slicer navigation module.
Evaluation of System Performance
The objective of this research is to develop a system that can allow real-time interactive querying of WM tracts in and around the surgical region of interest during neurosurgery. The real-time nature of the system depends greatly on the latency between tracking and fiber visualization, that is, the time taken between the tracking of the tool tip position by the VVCranial system and the visualization of the fiducial seed and WM tracts in the 3D Slicer software. Computation time for rendering the seeded WM tracts is a function of the size of the fiducial, as the larger the seed fiducial volume, the greater the potential number of fibers that need to be calculated and rendered. Off-line benchmarking was performed to quantify the time delay between tracking and visualization.
To measure the latency of the interactive querying of WM tracts, the entire process was divided into parts and the time taken for each part quantified. Tool tracking by the VVCranial system is done using an optical tracker, which has a refresh rate of 20Hz 33. The update rate at which the VVLink client in BioImage Suite could receive tracking data over the network was measured in 33, and at its slowest was 26.4Hz, implying that the update rate for 3D tool position tracking is limited by the camera system, at a rate of 20Hz (5ms).
BioImage Suite then converts the tracking data into a format compatible with the OpenIGTLink protocol and sends the data to 3D Slicer using the local host as it runs on the same workstation (transfer rates average approximately 50Mb/s). Once received in 3D Slicer, the tracking data is used to define a fiducial point which then seeds the WM tracts in the region, calculating and rendering their location for visualization. This is the process that takes the largest amount of time when compared to the previous steps. We performed experiments to quantify the time required for computation and visualization of seeded WM tracts from the moment in which 3D Slicer receives the tracking data over the OpenIGTLink protocol until the WM tracts are seeded and displayed. The clock time at the moment the tracking data was received by 3D Slicer was compared to another reading of the clock time once visualization of the fiducial and WM tracts was completed. The test was performed for fiducial radius sizes ranging from 1 to 10 mm, in 1mm increments, and for each test the fiducial seed was placed at 20 different locations in a single patient dataset, and the average time computed.
Results
The clinician was able to successfully perform the procedure in all 5 cases with scarcely any added complexity to the workflow. Data from the structural MRI, fMRI, and DTI datasets were visualized in 3D Slicer along with the relevant WM tracts seeded by the fiducial position that corresponded to the surgical tool tip location. The 3D Slicer visualization area was displayed on a separate monitor from the VVCranial navigation system display, as can be seen in Figure 2.
The intra-operative querying of the tractography dataset was performed and visualized on the 3D Slicer navigation module, as can be seen in Figure 3(a). Structural MR images were superimposed with segmented structures such as the brain tumor and eloquent cortical regions as mapped by fMRI. Figure 3(b) shows seeding of the tractography dataset using two sets of fiducials which correspond to the tool tip location and a fixed geometric offset (see Video, Supplemental Digital Content 1, illustrates intra-operative querying of white matter tracts during a case of tumor resection, 1 minute, 47 seconds, 10MB). A video showing real-time intraoperative seeding of white matter tracts is available as supplementary material.
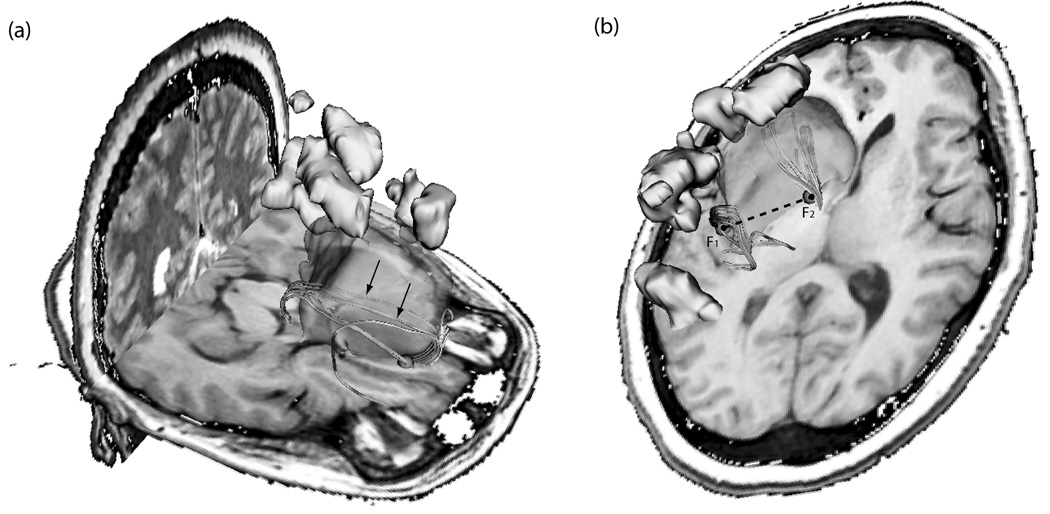
Figure 3.
Images shown in 3D Slicer using the intra-operative tractography navigation software module. The light green volume delineates the tumor location, the orange sphere represents the seed fiducial which is located at the tracked surgical tool tip location, the yellow regions indicate eloquent cortex as shown by language task-associated fMRI and the tubular fibers represent the white matter tracts seeded by the fiducial location. White matter tracts are color coded according to their fractional anisotropy (color scale). (a) White matter fibers being seeded by a fiducial representing tool tip position. Some infiltrating fibers can be seen within the segmented tumor volume (arrows). (b) Intra-operative querying of tractography datasets using the tool offset function. Fiducials F1 and F2 represent the surgical tool tip location and a fixed geometric offset respectively. The navigation module allows for geometric offsets from −80 to 80mm from the tool tip position. The two fiducials can seed the tractography dataset simultaneously
In addition to tracking the tool tip location, it was possible to use the tracked microscope focal point position to seed the tractography dataset. Figure 4 shows a screen shot from the intra-operative microscope view and the 3D Slicer navigation module.
Figure 4.
(a) Intra-operative view from microscope overlaid with segmented tumor anatomy, (b) the 3D Slicer display showing real-time visualization of WM tracts seeded by a fiducial which corresponds to the microscope focal point.
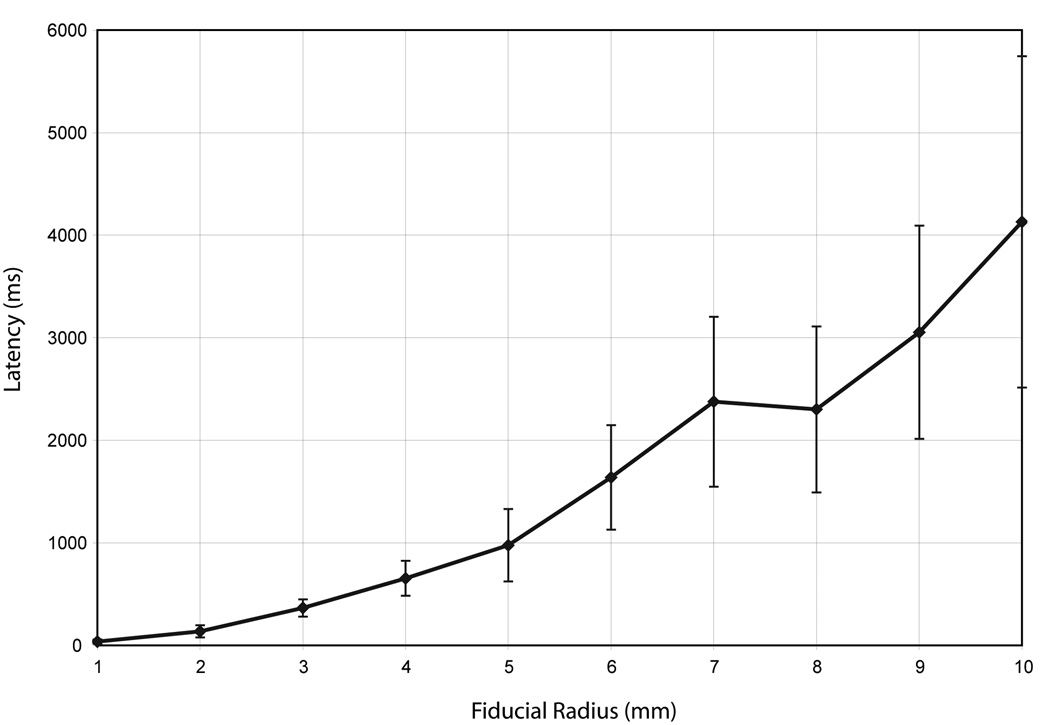
Latency between tracking and visualization increases with fiducial radius as potentially more white matter tracts are included and rendered within the volume of the fiducial. Figure 5 shows the latency recorded from when 3D Slicer receives the fiducial seed position from the BioImage Suite software until the seeded white matter tracts are actually calculated, rendered, and visualized on the screen. Latency varies from an average of 37ms for a 1mm fiducial radius, to a total of 4130ms with a 10mm fiducial radius. In practice a fiducial radius of 4mm is generally selected during the procedure, which is a good trade off between the number of fibers visualized and the latency, which is just under 654ms.
Figure 5.
Graph showing mean latency performance of the system, with error bars showing a standard deviation. The graph indicates the total time taken from when 3D Slicer receives the tracking data indicating fiducial seed location until the seeded white matter tracts are visualized on the screen. The latency increases with fiducial size (except between 7 and 8 mm, where the latency is approximately the same), as the bigger the fiducial, the larger the amount of potential tracts which must be calculated and rendered.
Discussion
Including DTI and tractography into the pre-operative datasets available for procedure planning can help surgeons form a mental map of the localization of critical WM tracts in relation to the tumor area. Presenting this information intra-operatively can make the anatomical and topographical relations between the WM tracts and the tumor readily available during surgery. We demonstrated the implementation of real-time intra-operative querying of white matter tracts by integrating a novel tractography algorithm with a standard neuronavigation system. The connection between the research software 3D Slicer and the VVCranial navigation system was accomplished using network protocols which isolated any adverse effects the research software could have on the commercial system. By tracking surgical instruments, the microscope focal point, or a geometric extension from these positions, a fiducial was defined which could then seed the tractography dataset at the tracked location. This allows visualization of the interaction between the surgical tools and the WM tracts close to the tumor resection site, and their relationship to any intra-operatively acquired information. The system was successfully tested with five patients.
The visualization of white matter tracts derived from DTI can be useful during neurosurgical planning and intervention, especially in patients with brain tumors in functional eloquent areas 38–42. Incorporating preoperative diffusion tensor MR imaging and WM fiber trajectory and location can facilitate maximum tumor resection with minimal neurological deficits 19–22, 43, 44. Functional mapping and image guidance methods have undergone considerable development in the last two decades, helping surgeons devise improved surgical strategies to reduce neurological deficits and increase tumor resection 45, 46. Intraoperative cortical mapping remains an important tool for the definitive demonstration of critical cortical areas 47–50. Using intraoperatively acquired information about critical cortical areas to define which white matter bundles are carrying critical information is one potential advantage of an approach which allows near real-time querying of the data.
Current methods for the intra-operative display of tractography data generally involve pre-operative clustering of certain critical fibers using a ROI approach, representing the selected fibers in a static 3D geometric form and displaying the polygon onto the structural and functional images already included in the clinical navigation system 20. The system presented here allows seeding the tracts using a fiducial defined by the surgical tool tip location, and interactive probing of the relevant fiber tracts, eliminating unnecessary data from the already information-overloaded environment. Tractography-integrated neuronavigation, despite its limitations, has been shown to be a reliable way to demonstrate WM connections intraoperatively 24, 25.
Any method that relies on pre-operatively acquired data can suffer from brain shift induced displacements, which is potentially the largest and most unpredictable source of error in navigation systems. Nimsky et al. 51, in work combining pre-operative and intra-operative MRI acquisition of DTI based tractography datasets, demonstrated that WM tracts can move an average of 2.7±6mm during tumor resection. Hence, there is a role for further intra-operative image acquisition to update the anatomical and tractography data after brain shift and/or tissue resection has taken place. In this sense, our method for querying fibers intra-operatively is compatible with any techniques that can successfully obtain intra-operative acquisition of anatomical and/or DTI datasets during the intervention (for example using intraoperative high field MRI). These newly acquired images can be co-registered and processed in 3D Slicer, and made available for intra-operative querying of the white matter tracts, although depending on the set-up of the operating room, it may be necessary to repeat the patient to image registration in order to take into account the brain shift. Our system can also be used with improved DTI acquisition techniques and tractography algorithms, for example, using two tensor models for resolving fiber crossing 52, combinatorial fiber tracking of the brain 53, or a flow based approach 54.
Intraoperative stimulation mapping remains the gold standard for identifying functional cortical areas 55–57. However, despite improvements in cortical mapping methods, the identification and preservation of eloquent pathways within the white matter remains problematic. Subcortical stimulation testing has been fruitfully used in a few centers but is limited in its ability to delineate the full extent of the tract and does not provide a positive control until the tract of interest has already been encountered 9, 56, 58–63. The method presented in this study could be used in conjunction with electro-cortical and sub-cortical mapping during surgery, where available, to help identify, confirm, and avoid eloquent cortical areas 46, 58, 59, 64, 65 and for cross-modal validation 9, 14, 46, 63. This combined approach of fiber tract visualization using DTI and subcortical mapping of critical tracts has shown to have an impact on surgical procedures by reducing the duration of the mapping and the number of clinically evident seizures 14. In addition, the intra-operative findings from cortical stimulation sites can be used to initiate the delineation of DTI based fiber tracts 65 by providing a seed fiducial position or defining a seed ROI. This can be especially advantageous for identifying critical functional tracts.
Although this study has emphasized the clinical usefulness of the integration of white matter tracts in the intra-operative setting, the use of a gold standard for comparison and validation has been difficult to establish. Some studies have compared the fiber delineation from DTI with the intraoperative findings from subcortical electrical stimulation, 7, 9, 14, 25, 46, 65 and results in rodents show compelling data 66, 67. However, a true validation of DTI based fiber delineation in humans has yet to be reported. Additionally, it is important to consider the accuracy of correspondence between the fiducial position used to seed the WM tracts in the 3D Slicer software and the real location of the surgical tool tip with respect to the brain anatomy. Several factors can induce an error between these two elements in the system, which include (i) the optical tracking error of the surgical tool tip, (ii) the patient to image registration error, (iii) the registration error due to the affine registration between the images from the different modalities, and (iv) brain shift errors during the intervention. Regarding the optical tracking there are two sources of error which can affect accuracy of the tracked tool tip position. The first is due to the inherent error associated to the optical tracking technology, with the VVCranial optical tracker presenting an average error of 0.5mm (with 95% confidence) when tracking the reflective markers on the tools 68. The second type of error refers to the extrapolation of tool tip location from the tracked reflective markers on the handle of the tools. The offset between the reflective markers of the tools and the tip location is a pre-calculated transform obtained through a prior calibration process. Supposing that the tool is relatively rigid, the tool tip extrapolation error is sufficiently small to ignore compared to the inherent optical tracking error. If however tool bending is significant, then there can be a considerable discrepancy between tracked tool position and its real location. The integration of our intra-operative tractography system into the clinical navigation system means that it is subject to these errors, although it does not affect or increase these errors in any way. Regarding patient to image registration, this is performed at the beginning of the procedure with the VVCranial system, and is not affected by the presence of our system. Affine registration provides a method to bring all the images from the different structural and functional modalities into anatomical alignment. The method can compensate for changes between several acquired images only in terms of rotation, translation, shearing, and changes in scale 69, but not for all nonlinear distortions of the images inherent in DTI/fMRI acquisitions. Our system requires the registration between the functional DTI dataset and the anatomical images, and as such, will be affected by the error of this registration process. Finally, intra-operative brain shift, which is not accounted for by the navigation system, can introduce a large and unpredictable error between the tracked location of the tool and its real position with respect to brain anatomy, as shown by intraoperative imaging techniques such as open MRI 70. Other less resource-intensive methods include using an ultrasound probe to measure deformation 71–73, and a stereo vision tracking system which tracks markers located on the cortical surface of the brain 74. Once deformation has been estimated, it can then be used to update the pre-operative brain images using non-rigid techniques, such as biomechanical modeling or elastic registration. As our system relies on the navigation system and the patient to image registration process performed at the beginning of the intervention, it is also subject to the inherent errors due to brain shift.
From a systems perspective, the current method of acquiring data from the VVCranial system requires using a third software system, BioImage Suite, to convert the data from VVLink to OpenIGTLink in order to be imported into the 3D Slicer software. By using this bridging software, we enable several potential points of failure in the system, and hence it is desirable to have a direct connection from the VVCranial directly into 3D Slicer.
Future work is being directed in three areas. The first includes reducing the latency between tool tracking and tractography visualization. This might include exploring ways of accelerating the required tractography computation using very high end workstations or using the processing power available on modern graphic processing units. Secondly, it is currently only possible to visualize seeded white matter tracts generated from whole brain tractography. It would be clinically useful to visualize only some pre-selected major white matter tract systems (like pyramidal tract or optical pathway) in the vicinity of the tool-tip position, and hence we are modifying the current algorithm to allow pre-selection of fibers during pre-operative planning. This would allow showing only those fibers which are considered clinically relevant for the procedure, thus removing superfluous information. Thirdly, we are developing the capability to enable visualization of the seeded white matter fibers through the view of the microscope. This would allow the spatial relationship between the white matter tracts and the brain anatomy to be presented in an intuitive manner closely related to the surgical view, but will require transferring data generated by our research system to a separate commercial system.
Conclusions
Real-time interactive querying of relevant WM tracts during tumor resection has been reliably integrated into a frameless stereotactic navigation and used during five clinical cases. The tracked position of a tool tip, microscope focal point, or any geometric offset can be used to define a fiducial point, which in turn seeds the tractography dataset and renders relevant WM tracts on the pre-operative images used for navigation. This will provide knowledge of the functionally relevant tracts that interact with the tumor, giving the surgeon more detailed and specific information with which to execute the planned surgical resection with minimal neurological deficit.
Supplementary Material
- Authors: Haytham Elhawary, Haiying Liu, Pratik Patel, Isaiah Norton, Laura Rigolo, Xenophon Papademetris, Nobuhiko Hata, Alexandra J. Golby
- Videographers: Haytham Elhawary, Isaiah Norton
- Length: 1 minute, 47 seconds
- Size: 10MB
Acknowledgements
This work was supported by NIH grants 5U41RR019703, P01-CA67165 and R01 EB006494, the Intelligent Surgical Instruments Project of METI (Japan), the Brain Science Foundation and the Klarman Family Foundation. Pratik Patel, third author on this manuscript, is an employee of Brainlab AG (Neurosurgery Senior Application Consultant and Product Manager). Brainlab provided the VVLink software for evaluation purposes in the preliminary phase of this study.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Dimaio SP, Archip N, Hata N, et al. Image-guided neurosurgery at Brigham and Women's Hospital. IEEE Eng Med Biol Mag. 2006 Sep-Oct;25(5):67–73. doi: 10.1109/memb.2006.1705749. [DOI] [PubMed] [Google Scholar]
- 2.Berger MS, Deliganis AV, Dobbins J, Keles GE. The effect of extent of resection on recurrence in patients with low grade cerebral hemisphere gliomas. Cancer. 1994;74(6):1784–1791. doi: 10.1002/1097-0142(19940915)74:6<1784::aid-cncr2820740622>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 3.Claus EB, Horlacher A, Hsu L, et al. Survival rates in patients with low-grade glioma after intraoperative magnetic resonance image guidance. Cancer. 2005;103(6):1227–1233. doi: 10.1002/cncr.20867. [DOI] [PubMed] [Google Scholar]
- 4.Sanai N, Berger M. Glioma extent of resection and its impact on patient outcome. Neurosurgery. 2008 April;62(4):753–766. doi: 10.1227/01.neu.0000318159.21731.cf. 2008. [DOI] [PubMed] [Google Scholar]
- 5.McGirt MJ, Mukherjee D, Chaichana KL, Than KD, Weingart JD, Quinones-Hinojosa A. Association of surgically acquired motor and language deficits on overall survival after resection of glioblastoma multiforme. Neurosurgery. 2009 Sep;65(3):463–469. doi: 10.1227/01.NEU.0000349763.42238.E9. discussion 469–470. [DOI] [PubMed] [Google Scholar]
- 6.Talos I-F, Zou KH, Ohno-Machado L, et al. Supratentorial low-grade glioma resectability: statistical predictive analysis based on anatomic MR features and tumor characteristics. Radiology. 2006 May 1;239(2):506–513. doi: 10.1148/radiol.2392050661. 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kinoshita M, Yamada K, Hashimoto N, et al. Fiber-tracking does not accurately estimate size of fiber bundle in pathological condition: initial neurosurgical experience using neuronavigation and subcortical white matter stimulation. NeuroImage. 2005;25(2):424–429. doi: 10.1016/j.neuroimage.2004.07.076. [DOI] [PubMed] [Google Scholar]
- 8.Ojemann JG, Miller JW, Silbergeld DL. Preserved function in brain invaded by tumor. Neurosurgery. 1996 August;39(2):253–259. doi: 10.1097/00006123-199608000-00003. 1996. [DOI] [PubMed] [Google Scholar]
- 9.Berman JI, Berger MS, Chung SW, Nagarajan SS, Henry RG. Accuracy of diffusion tensor magnetic resonance imaging tractography assessed using intraoperative subcortical stimulation mapping and magnetic source imaging. J Neurosurg. 2007 Sep;107(3):488–494. doi: 10.3171/JNS-07/09/0488. [DOI] [PubMed] [Google Scholar]
- 10.Duffau H, Capelle L, Denvil D, et al. Usefulness of intraoperative electrical subcortical mapping during surgery for low-grade gliomas located within eloquent brain regions: functional results in a consecutive series of 103 patients. Journal of Neurosurgery. 2003;98(4):764–778. doi: 10.3171/jns.2003.98.4.0764. [DOI] [PubMed] [Google Scholar]
- 11.Keles GE, Lundin DA, Lamborn KR, Chang EF, Ojemann G, Berger MS. Intraoperative subcortical stimulation mapping for hemispheric perirolandic gliomas located within or adjacent to the descending motor pathways: evaluation of morbidity and assessment of functional outcome in 294 patients. Journal of Neurosurgery. 2004;100(3):369–375. doi: 10.3171/jns.2004.100.3.0369. [DOI] [PubMed] [Google Scholar]
- 12.Basser PJ, Mattiello J, LeBihan D. Estimation of the effective self-diffusion tensor from the NMR spin echo. J Magn Reson B. 1994 Mar;103(3):247–254. doi: 10.1006/jmrb.1994.1037. [DOI] [PubMed] [Google Scholar]
- 13.Mori S, Crain BJ, Chacko VP, Zijl PCMV. Three-dimensional tracking of axonal projections in the brain by magnetic resonance imaging. Annals of Neurology. 1999;45(2):265–269. doi: 10.1002/1531-8249(199902)45:2<265::aid-ana21>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 14.Bello L, Gambini A, Castellano A, et al. Motor and language DTI fiber tracking combined with intraoperative subcortical mapping for surgical removal of gliomas. NeuroImage. 2008;39(1):369–382. doi: 10.1016/j.neuroimage.2007.08.031. [DOI] [PubMed] [Google Scholar]
- 15.Smits M, Vernooij MW, Wielopolski PA, Vincent AJPE, Houston GC, van der Lugt A. Incorporating functional MR imaging into diffusion tensor tractography in the preoperative assessment of the corticospinal tract in patients with brain tumors. AJNR Am J Neuroradiol. 2007 August 1;28(7):1354–1361. doi: 10.3174/ajnr.A0538. 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hattingen E, Rathert J, Jurcoane A, et al. A standardised evaluation of pre-surgical imaging of the corticospinal tract: where to place the seed ROI. Neurosurg Rev. 2009 May 13; doi: 10.1007/s10143-009-0197-1. [DOI] [PubMed] [Google Scholar]
- 17.Voineskos AN, O'Donnell LJ, Lobaugh NJ, et al. Quantitative examination of a novel clustering method using magnetic resonance diffusion tensor tractography. Neuroimage. 2009 Apr 1;45(2):370–376. doi: 10.1016/j.neuroimage.2008.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Golby AJ, Kindlmann G, Pieper S, Yarmakovich A, Norton I, Kikinis R. Interactive DTI visualization for neurosurgical planning. Neurosurgery, Submitted for review. 2009 doi: 10.1227/NEU.0b013e3182061ebb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chen X, Weigel D, Ganslandt O, Fahlbusch R, Buchfelder M, Nimsky C. Diffusion tensor-based fiber tracking and intraoperative neuronavigation for the resection of a brainstem cavernous angioma. Surg Neurol. 2007 Sep;68(3):285–291. doi: 10.1016/j.surneu.2007.05.005. discussion 291. [DOI] [PubMed] [Google Scholar]
- 20.Nimsky C, Ganslandt O, Fahlbusch R. Implementation of fiber tract navigation. Neurosurgery. 2007 July;61(1 Suppl):306–318. doi: 10.1227/01.neu.0000279224.83998.7d. 2007. [DOI] [PubMed] [Google Scholar]
- 21.Mastronardi L, Bozzao A, D'Andrea G, et al. Use of preoperative and intraoperative magnetic resonance tractography in intracranial tumor surgery. Clin Neurosurg. 2008;55:160–164. [PubMed] [Google Scholar]
- 22.Romano A, Ferrante M, Cipriani V, et al. Role of magnetic resonance tractography in the preoperative planning and intraoperative assessment of patients with intra-axial brain tumours. Radiol Med. 2007 Sep;112(6):906–920. doi: 10.1007/s11547-007-0181-1. [DOI] [PubMed] [Google Scholar]
- 23.Kleiser R, Staempfli P, Valavanis A, Boesiger P, Kollias S. Impact of fMRI-guided advanced DTI fiber tracking techniques on their clinical applications in patients with brain tumors. Neuroradiology. 2009 May 29; doi: 10.1007/s00234-009-0539-2. [DOI] [PubMed] [Google Scholar]
- 24.Kamada K, Todo T, Masutani Y, et al. Combined use of tractography-integrated functional neuronavigation and direct fiber stimulation. Journal of Neurosurgery. 2005;102(4):664–672. doi: 10.3171/jns.2005.102.4.0664. [DOI] [PubMed] [Google Scholar]
- 25.Kamada K, Todo T, Ota T, et al. The motor-evoked potential threshold evaluated by tractography and electrical stimulation. J Neurosurg. 2009 Jan 23; doi: 10.3171/2008.9.JNS08414. [DOI] [PubMed] [Google Scholar]
- 26.Gering DT, Nabavi A, Kikinis R, et al. An integrated visualization system for surgical planning and guidance using image fusion and an open MR. J Magn Reson Imag. 2001;13(6):967–975. doi: 10.1002/jmri.1139. [DOI] [PubMed] [Google Scholar]
- 27.Beaulieu C. The basis of anisotropic water diffusion in the nervous system - a technical review. NMR in Biomedicine. 2002;15(7–8):435–455. doi: 10.1002/nbm.782. [DOI] [PubMed] [Google Scholar]
- 28.Basser PJ, Pajevic S, Pierpaoli C, Duda J, Aldroubi A. In vivo fiber tractography using DT-MRI data. Magnetic Resonance in Medicine. 2000;44(4):625–632. doi: 10.1002/1522-2594(200010)44:4<625::aid-mrm17>3.0.co;2-o. [DOI] [PubMed] [Google Scholar]
- 29.Tao X, Miller J. A method for registering diffusion weighted magnetic resonance images. Med Image Comput Comput Assist Interv. MICCAI. 2006;10:2006. doi: 10.1007/11866763_73. [DOI] [PubMed] [Google Scholar]
- 30.Mattes D, Haynor DR, Vesselle H, Lewellen TK, Eubank W. PET-CT image registration in the chest using free-form deformations. Medical Imaging, IEEE Transactions on. 2003;22(1):120–128. doi: 10.1109/TMI.2003.809072. [DOI] [PubMed] [Google Scholar]
- 31.Wells W, Viola P, Atsumi H, Nakajima S, Kikinis R. Multi-modal volume registration by maximization of mutual information. Med Image Anal. 1996 doi: 10.1016/s1361-8415(01)80004-9. 03. [DOI] [PubMed] [Google Scholar]
- 32.Sure U, Benes L, Riegel T, Schulte DM, Bertalanffy H. Image fusion for skull base neuronavigation. Technical note. Neurol Med Chir (Tokyo) 2002 Oct;42(10):458–461. doi: 10.2176/nmc.42.458. discussion 462. [DOI] [PubMed] [Google Scholar]
- 33.Papademetris X, DeLorenzo C, Flossmann S, et al. From medical image computing to computer-aided intervention: development of a research interface for image-guided navigation. The International Journal of Medical Robotics and Computer Assisted Surgery. 2009;5(2):147–157. doi: 10.1002/rcs.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tokuda J, Fischer GS, Papademetris X, et al. OpenIGTLink: an open network protocol for image-guided therapy environment. Internationl Journal of Medical Robotics and Computer Assisted Interventions. 2009 doi: 10.1002/rcs.274. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Papademetris X, Jackowski M, Rajeevan N, Constable RT, Staib LH. Bioimage suite: An integrated medical image analysis suite. The Insight Journal. 2005 http://hdl.handle.net/1926/37. [PMC free article] [PubMed] [Google Scholar]
- 36.Raabe A, Krishnan R, Wolff R, Hermann E, Zimmermann M, Seifert V. Laser surface scanning for patient registration in intracranial image-guided surgery. Neurosurgery. 2002 Apr;50(4):797–801. doi: 10.1097/00006123-200204000-00021. discussion 802–793. [DOI] [PubMed] [Google Scholar]
- 37.Papademetris X, Vives KP, DiStasio M, et al. Development of a research interface for image guided intervention: initial application to epilepsy neurosurgery. Paper presented at: 3rd IEEE International Symposium on Biomedical Imaging: Nano to Macro. 2006 April 6–9; 2006. [Google Scholar]
- 38.Coenen VA, Krings T, Mayfrank L, et al. Three-dimensional visualization of the pyramidal tract in a neuronavigation system during brain tumor surgery: first experiences and technical note. Neurosurgery. 2001 Jul;49(1):86–92. doi: 10.1097/00006123-200107000-00013. discussion 92–83. [DOI] [PubMed] [Google Scholar]
- 39.Witwer BP, Moftakhar R, Hasan KM, et al. Diffusion-tensor imaging of white matter tracts in patients with cerebral neoplasm. J Neurosurg. 2002 Sep;97(3):568–575. doi: 10.3171/jns.2002.97.3.0568. [DOI] [PubMed] [Google Scholar]
- 40.Hendler T, Pianka P, Sigal M, et al. Delineating gray and white matter involvement in brain lesions: three-dimensional alignment of functional magnetic resonance and diffusion-tensor imaging. J Neurosurg. 2003 Dec;99(6):1018–1027. doi: 10.3171/jns.2003.99.6.1018. [DOI] [PubMed] [Google Scholar]
- 41.Wieshmann UC, Symms MR, Parker GJ, et al. Diffusion tensor imaging demonstrates deviation of fibres in normal appearing white matter adjacent to a brain tumour. J Neurol Neurosurg Psychiatry. 2000 Apr;68(4):501–503. doi: 10.1136/jnnp.68.4.501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Holodny AI, Schwartz TH, Ollenschleger M, Liu WC, Schulder M. Tumor involvement of the corticospinal tract: diffusion magnetic resonance tractography with intraoperative correlation. J Neurosurg. 2001 Dec;95(6):1082. doi: 10.3171/jns.2001.95.6.1082. [DOI] [PubMed] [Google Scholar]
- 43.Gulati S, Berntsen EM, Solheim O, et al. Surgical resection of high-grade gliomas in eloquent regions guided by blood oxygenation level dependent functional magnetic resonance imaging, diffusion tensor tractography, and intraoperative navigated 3D ultrasound. Minim Invasive Neurosurg. 2009 Feb;52(1):17–24. doi: 10.1055/s-0028-1104566. [DOI] [PubMed] [Google Scholar]
- 44.Shinoura N, Yamada R, Kodama T, Suzuki Y, Takahashi M, Yagi K. Preoperative fMRI, tractography and continuous task during awake surgery for maintenance of motor function following surgical resection of metastatic tumor spread to the primary motor area. Minim Invasive Neurosurg. 2005 Apr;48(2):85–90. doi: 10.1055/s-2004-830227. [DOI] [PubMed] [Google Scholar]
- 45.Kurimoto M, Hayashi N, Kamiyama H, et al. Impact of neuronavigation and image-guided extensive resection for adult patients with supratentorial malignant astrocytomas: a single-institution retrospective study. Minim Invasive Neurosurg. 2004 Oct;47(5):278–283. doi: 10.1055/s-2004-830093. [DOI] [PubMed] [Google Scholar]
- 46.Leclercq D, Duffau H, Delmaire C, et al. Comparison of diffusion tensor imaging tractography of language tracts and intraoperative subcortical stimulations. J Neurosurg. 2009 Sep 11; doi: 10.3171/2009.8.JNS09558. [DOI] [PubMed] [Google Scholar]
- 47.Burchiel KJ, Clarke H, Ojemann GA, Dacey RG, Winn HR. Use of stimulation mapping and corticography in the excision of arteriovenous malformations in sensorimotor and language-related neocortex. Neurosurgery. 1989 Mar;24(3):322–327. doi: 10.1227/00006123-198903000-00002. [DOI] [PubMed] [Google Scholar]
- 48.Sartorius CJ. Intraoperative brain mapping during tumor resection from critical areas of the brain. Indiana Med. 1994 Nov-Dec;87(6):476–478. [PubMed] [Google Scholar]
- 49.Rutten GJ, van Rijen PC, van Veelen CW, Ramsey NF. Language area localization with three-dimensional functional magnetic resonance imaging matches intrasulcal electrostimulation in Broca's area. Ann Neurol. 1999 Sep;46(3):405–408. doi: 10.1002/1531-8249(199909)46:3<405::aid-ana17>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- 50.Roux FE, Boulanouar K, Lotterie JA, Mejdoubi M, LeSage JP, Berry I. Language functional magnetic resonance imaging in preoperative assessment of language areas: correlation with direct cortical stimulation. Neurosurgery. 2003 Jun;52(6):1335–1345. doi: 10.1227/01.neu.0000064803.05077.40. discussion 1345–1337. [DOI] [PubMed] [Google Scholar]
- 51.Nimsky C, Ganslandt O, Hastreiter P, et al. Preoperative and intraoperative diffusion tensor imaging-based fiber tracking in glioma surgery. Neurosurgery. 2005;56(1):130–137. doi: 10.1227/01.neu.0000144842.18771.30. discussion 138. [DOI] [PubMed] [Google Scholar]
- 52.Sotiropoulos SN, Bai L, Morgan PS, Auer DP, Constantinescu CS, Tench CR. A regularized two-tensor model fit to low angular resolution diffusion images using basis directions. J Magn Reson Imaging. 2008 Jul;28(1):199–209. doi: 10.1002/jmri.21380. [DOI] [PubMed] [Google Scholar]
- 53.Lifshits S, Tamir A, Assaf Y. Combinatorial fiber-tracking of the human brain. Neuroimage. 2009 Jun 4; doi: 10.1016/j.neuroimage.2009.05.086. [DOI] [PubMed] [Google Scholar]
- 54.Guo W, Chen Y, Zeng Q. A geometric flow-based approach for diffusion tensor image segmentation. Philos Transact A Math Phys Eng Sci. 2008 Jul 13;366(1874):2279–2292. doi: 10.1098/rsta.2008.0042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Berger MS, Ojemann GA. Intraoperative brain mapping techniques in neuro-oncology. Stereotact Funct Neurosurg. 1992;58(1–4):153–161. doi: 10.1159/000098989. [DOI] [PubMed] [Google Scholar]
- 56.Skirboll SS, Ojemann GA, Berger MS, Lettich E, Winn HR. Functional cortex and subcortical white matter located within gliomas. Neurosurgery. 1996 Apr;38(4):678–684. discussion 684–675. [PubMed] [Google Scholar]
- 57.Ojemann G, Ojemann J, Lettich E, Berger M. Cortical language localization in left, dominant hemisphere. An electrical stimulation mapping investigation in 117 patients. J Neurosurg. 1989 Sep;71(3):316–326. doi: 10.3171/jns.1989.71.3.0316. [DOI] [PubMed] [Google Scholar]
- 58.Duffau H. Surgery of low-grade gliomas: towards a 'functional neurooncology'. Curr Opin Oncol. 2009 Nov;21(6):543–549. doi: 10.1097/CCO.0b013e3283305996. [DOI] [PubMed] [Google Scholar]
- 59.Picht T, Wachter D, Mularski S, et al. Functional magnetic resonance imaging and cortical mapping in motor cortex tumor surgery: complementary methods. Zentralbl Neurochir. 2008 Feb;69(1):1–6. doi: 10.1055/s-2007-993138. [DOI] [PubMed] [Google Scholar]
- 60.Duffau H, Thiebaut de Schotten M, Mandonnet E. White matter functional connectivity as an additional landmark for dominant temporal lobectomy. J Neurol Neurosurg Psychiatry. 2008 May;79(5):492–495. doi: 10.1136/jnnp.2007.121004. [DOI] [PubMed] [Google Scholar]
- 61.Duffau H. Contribution of cortical and subcortical electrostimulation in brain glioma surgery: methodological and functional considerations. Neurophysiol Clin. 2007 Dec;37(6):373–382. doi: 10.1016/j.neucli.2007.09.003. [DOI] [PubMed] [Google Scholar]
- 62.Duffau H. Intraoperative cortico-subcortical stimulations in surgery of low-grade gliomas. Expert Rev Neurother. 2005 Jul;5(4):473–485. doi: 10.1586/14737175.5.4.473. [DOI] [PubMed] [Google Scholar]
- 63.Berman JI, Berger MS, Mukherjee P, Henry RG. Diffusion-tensor imaging-guided tracking of fibers of the pyramidal tract combined with intraoperative cortical stimulation mapping in patients with gliomas. J Neurosurg. 2004 Jul;101(1):66–72. doi: 10.3171/jns.2004.101.1.0066. [DOI] [PubMed] [Google Scholar]
- 64.Duffau H, Lopes M, Arthuis F, et al. Contribution of intraoperative electrical stimulations in surgery of low grade gliomas: a comparative study between two series without (1985–96) and with (1996–2003) functional mapping in the same institution. J Neurol Neurosurg Psychiatry. 2005 Jun;76(6):845–851. doi: 10.1136/jnnp.2004.048520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Henry RG, Berman JI, Nagarajan SS, Mukherjee P, Berger MS. Subcortical pathways serving cortical language sites: initial experience with diffusion tensor imaging fiber tracking combined with intraoperative language mapping. Neuroimage. 2004 Feb;21(2):616–622. doi: 10.1016/j.neuroimage.2003.09.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Xue R, van Zijl PC, Crain BJ, Solaiyappan M, Mori S. In vivo three-dimensional reconstruction of rat brain axonal projections by diffusion tensor imaging. Magn Reson Med. 1999 Dec;42(6):1123–1127. doi: 10.1002/(sici)1522-2594(199912)42:6<1123::aid-mrm17>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 67.Mori S, Itoh R, Zhang J, et al. Diffusion tensor imaging of the developing mouse brain. Magn Reson Med. 2001 Jul;46(1):18–23. doi: 10.1002/mrm.1155. [DOI] [PubMed] [Google Scholar]
- 68.Wiles AD, Thompson DG, Frantz DD. Accuracy assessment and interpretation for optical tracking systems; Paper presented at: Medical Imaging 2004: Visualization, Image-Guided Procedures, and Display; San Diego, CA, USA. 2004. [Google Scholar]
- 69.Jenkinson M, Smith S. A global optimisation method for robust affine registration of brain images. Medical Image Analysis. 2001 Jun;5(2):143–156. doi: 10.1016/s1361-8415(01)00036-6. [DOI] [PubMed] [Google Scholar]
- 70.Nabavi A, Black PM, Gering DT, et al. Serial Intraoperative Magnetic Resonance Imaging of Brain Shift: Neurosurgery. 2001 April;48(4):787–798. doi: 10.1097/00006123-200104000-00019. 2001. [DOI] [PubMed] [Google Scholar]
- 71.White PJ, Whalen S, Tang SC, Clement GT, Jolesz F, Golby AJ. An intraoperative brain shift monitor using shear mode transcranial ultrasound: preliminary results. J Ultrasound Med. 2009 Feb;28(2):191–203. doi: 10.7863/jum.2009.28.2.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ji S, Wu Z, Hartov A, Roberts DW, Paulsen KD. Mutual-information-based image to patient re-registration using intraoperative ultrasound in image-guided neurosurgery. Med Phys. 2008 Oct;35(10):4612–4624. doi: 10.1118/1.2977728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Reinertsen I, Lindseth F, Unsgaard G, Collins DL. Clinical validation of vessel-based registration for correction of brain-shift. Med Image Anal. 2007 Dec;11(6):673–684. doi: 10.1016/j.media.2007.06.008. [DOI] [PubMed] [Google Scholar]
- 74.DeLorenzo C, Papademetris X, Staib LH, Vives KP, Spencer DD, Duncan JS. Nonrigid intraoperative cortical surface tracking using game theory; Paper presented at: IEEE 11th International Conference on Computer Vision.2007. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
- Authors: Haytham Elhawary, Haiying Liu, Pratik Patel, Isaiah Norton, Laura Rigolo, Xenophon Papademetris, Nobuhiko Hata, Alexandra J. Golby
- Videographers: Haytham Elhawary, Isaiah Norton
- Length: 1 minute, 47 seconds
- Size: 10MB