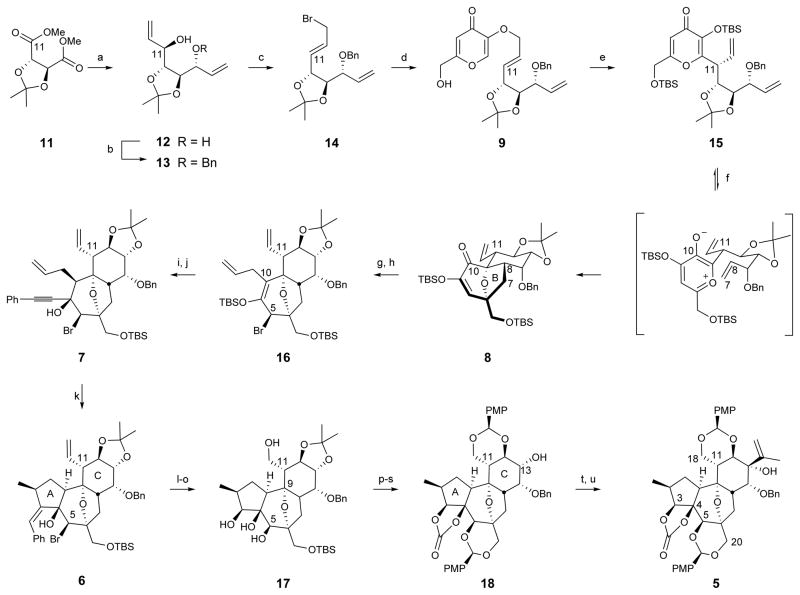
Figure 2. Synthesis of the complete daphnane skeleton (5).
Conditions: (a) DIBAL, toluene, −78 °C, then divinylzinc, −78 °C to r.t. (6:1 ratio of diastereomers), 80%; (b) NaH, BnBr, DMF, 0 °C to r.t., 86%; (c) PPh3, CBr4, CH2Cl2, 0 °C to r.t., 86%; (d) 10, i-PrOH, 80 °C, 78%; (e) neat, 122 °C, then TBS-Cl, imidazole, CH2Cl2, r.t. (10:1 ratio of diastereomers), 74%; (f) 1,2-dichlorobenzene, 250 °C, microwave, 91%; (g) Mg, I2, allyl bromide, Et2O, 0 °C, 93%; (h) SOBr2, pyridine, Et2O, −40 °C, 82%; (i) TBAF, AcOH, THF, 0 °C, 84%; (j) PhCCH, MeLi · LiBr, THF, −78 °C to r.t., 92%; (k) Pd2(dba)3·CHCl3 (1 mol %), PMHS (10 eq), HCOOH (3 eq), PhCH3, r.t., 80%; (l) O3, CH2Cl2/MeOH, −78 °C, then thiourea, 89%; (m) NaBH4, CeCl3·7H2O, CH2Cl2/MeOH, −78 °C to r.t., 79%; (n) Zn, NH4Cl, EtOH, 55°C, 89%; (o) VO(acac)2, t-BuOOH, CH2Cl2, r.t., 84%; (p) TBS-Cl, imidazole, CH2Cl2, r.t.; (q) triphosgene, pyridine, CH2Cl2, r.t., 86% (two steps); (r) TBAF, THF, 0 °C to r.t.; (s) (PMP)CH(OMe)2, p-TsOH, CH2Cl2, r.t., 93% (two steps); (t) Dess-Martin periodinane, NaHCO3, CH2Cl2, r.t.; (u) isopropenyl lithium, CeCl3, THF, −78 °C, 49% (two steps).