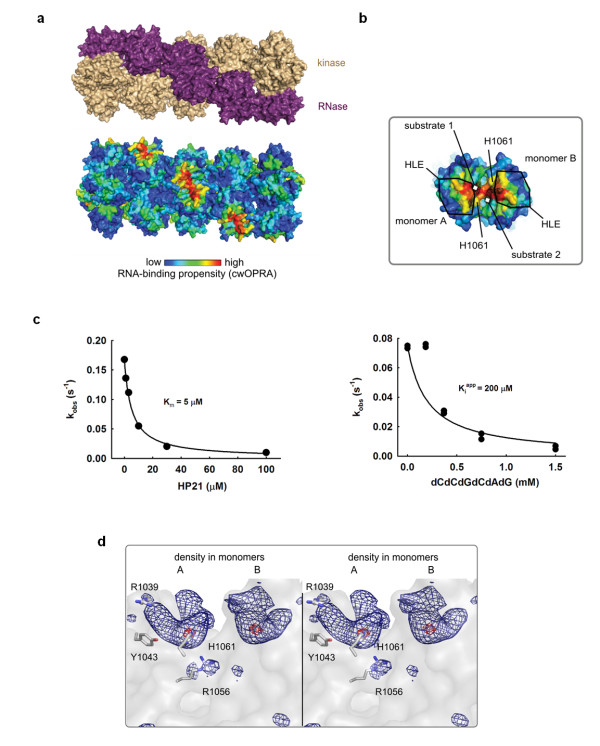
Figure 1.
Oligonucleotide binding to Ire1 RNase. (a) Ire1 oligomer (PDB ID 3fbv) colored by domains (top) and RNA-binding propensity (bottom). (b) Close view of symmetric back-to-back RNase dimer from the oligomer in (a) with electron density for dCdCdGdCdAdG from the C222 crystal structure superimposed. Electron density colocalizes with the area of the strongest RNA binding propensity and is in direct contact with conserved histidine H1061 and the helix-loop element (HLE). (c) Binding of an RNA stem-loop HP21 and of dCdCdGdCdAdG to Ire1KR32 measured via inhibition of 32P-5'-HP21 cleavage by unlabeled HP21 (Km = 5 ± 1 μM) or dCdCdGdCdAdG (Kiapp = 200 ± 50 μM). Reactions contained 20 mM 4-(2-hydroxyethyl)-1-piperazine-ethanesulfonic acid (HEPES) (pH 7.4), 70 mM NaCl, 5% glycerol, 2 mM Mg(CH3COO)2, 2 mM ADP, 4 mM dithiothreitol, 3 μM total Ire1KR32 and ≤ 1 pM 32P-5'-HP21, and were conducted at 30°C. Measurements of HP21 binding at 1 μM and 10 μM of total Ire1KR32 are shown in Additional file 1, Figure S1. (d), Wall-eyed stereoview of non-crystallographic symmetry (NCS)-averaged Fo-Fc electron density map at the RNase active site. Contour levels are 7σ (dark blue) and 13σ (red). Side chain positions are from the crystal structure 3fbv.