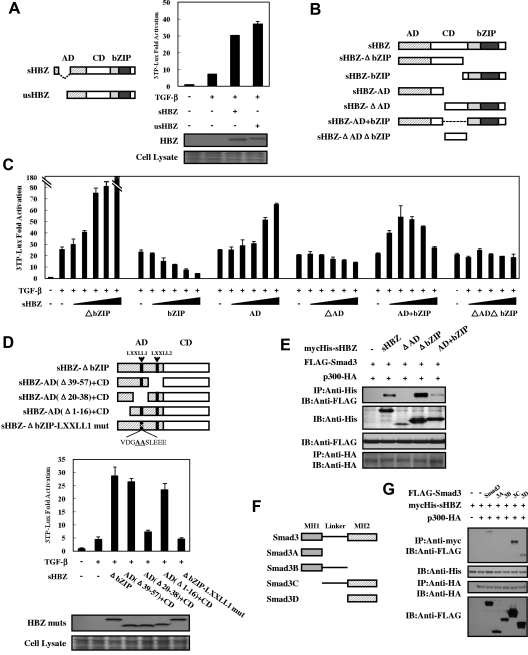
Figure 4.
Domains of HBZ responsible for the activation of TGF-β signaling. (A) Comparison of the effect of sHBZ and usHBZ on TGF-β activation. (Left panel) Schematic diagram of sHBZ and usHBZ. (Right panel) HepG2 cells were cotransfected with 3TP-Lux (0.5 μg), phRL-TK (2 ng), pcDNA3.1-mycHis-sHBZ, or pcDNA3.1-mycHis-usHBZ (200 ng). Luciferase activity was measured 24 hours after 10 ng/mL TGF-β stimulation. mycHis tagged sHBZ and usHBZ were detected by Western blot (middle panel). CBB staining was shown as the loading control (bottom panel). (B) Schematic diagram of sHBZ and its mutants used in this study. Characteristic domains of sHBZ are indicated as follows: AD, CD, and basic leucine zipper domain (bZIP). (C) Analysis of sHBZ deletion mutants for their effect on TGF-β–mediated signaling. In 12-well plates, HepG2 cells were cotransfected with 3TP-Lux (0.5 μg), phRL-TK (2 ng), and pME18Sneo-sHBZ mutants (0, 5, 20, 100, 200, and 500 ng). Luciferase activity was measured 24 hours after stimulation by TGF-β (10 ng/mL). (D) The N-terminal LXXLL1 motif of HBZ is important in enhancing TGF-β–induced luciferase expression. (Top panel) Schema of sHBZ-ΔbZIP and its deletion mutants. The locations of the LXXLL motifs are indicated. The mutated residues in the LXXLL1 motif are in bold and underlined. (Middle panel) In 24-well plates, HepG2 cells were cotransfected with 3TP-Lux (0.25 μg), phRL-TK (1 ng), and mycHis-sHBZ-ΔbZIP or its mutants (1 μg). At 24 hours after transfection, the cells were stimulated with or without 10 ng/mL TGF-β. Cell lysates were subjected to luciferase assay 24 hours after stimulation. sHBZ-ΔbZIP and its mutants were detected by Western blot. CBB staining was shown as the loading control (bottom panel). (E) Determination of the region of HBZ responsible for the interaction with Smad3. COS7 cells were transfected with the indicated mycHis-sHBZ mutants along with the FLAG-Smad3 and HA-p300 vectors. Cell lysates were subjected to immunoprecipitation using anti–c-Myc followed by immunoblotting using anti-FLAG. (F) Schematic drawing of Smad3 and its deletion mutants. The locations of the MH1 domain, MH2 domain, and the linker domain are indicated. (G) Mapping the region of the Smad3 protein necessary for interaction with sHBZ. COS7 cells were transfected with HA-p300, mycHis-sHBZ, and full-length or mutant FLAG-Smad3. At 48 hours after transfection, total cell lysates were subjected to immunoprecipitation using anti–c-Myc followed by IB using anti-FLAG.