Figure 2.
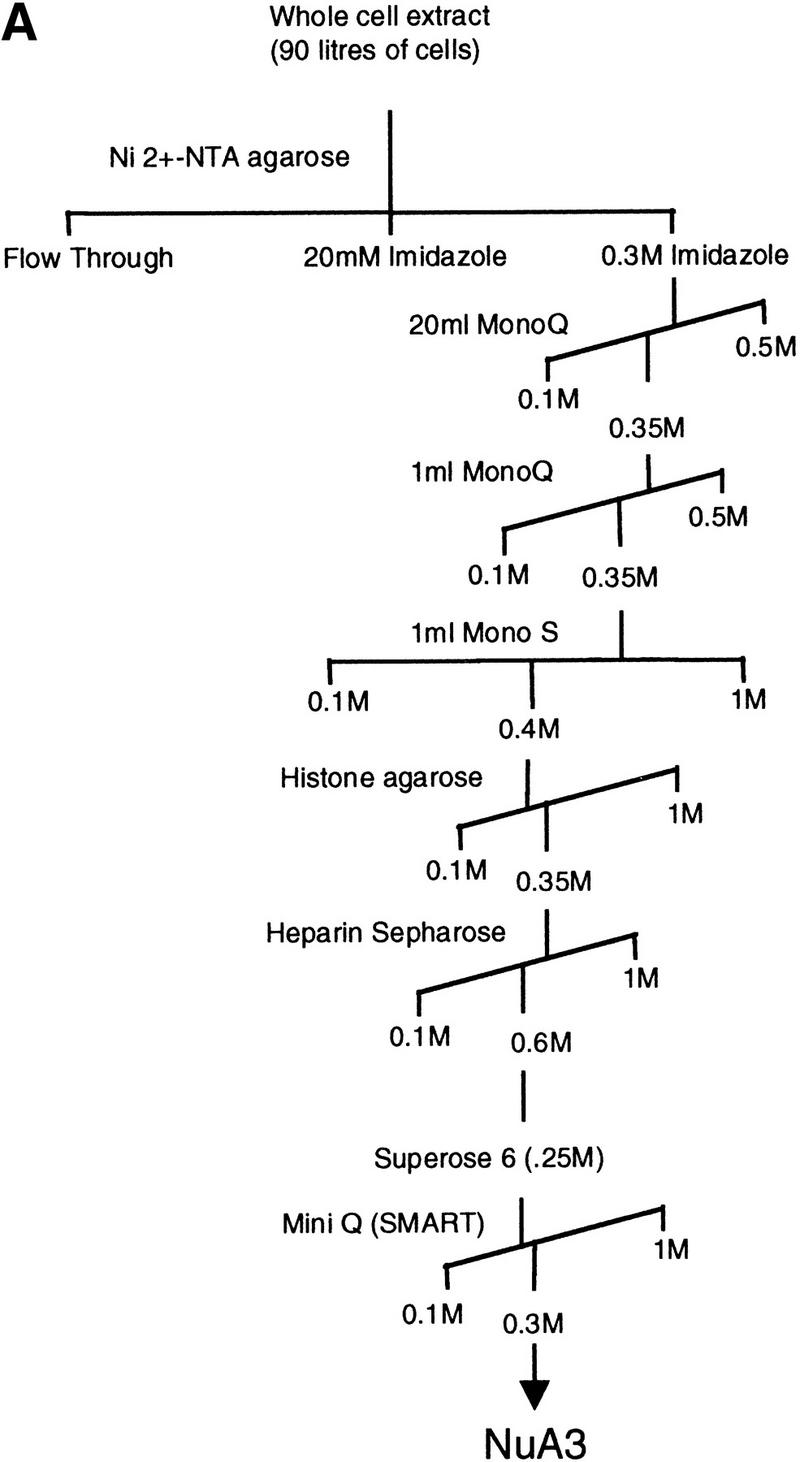
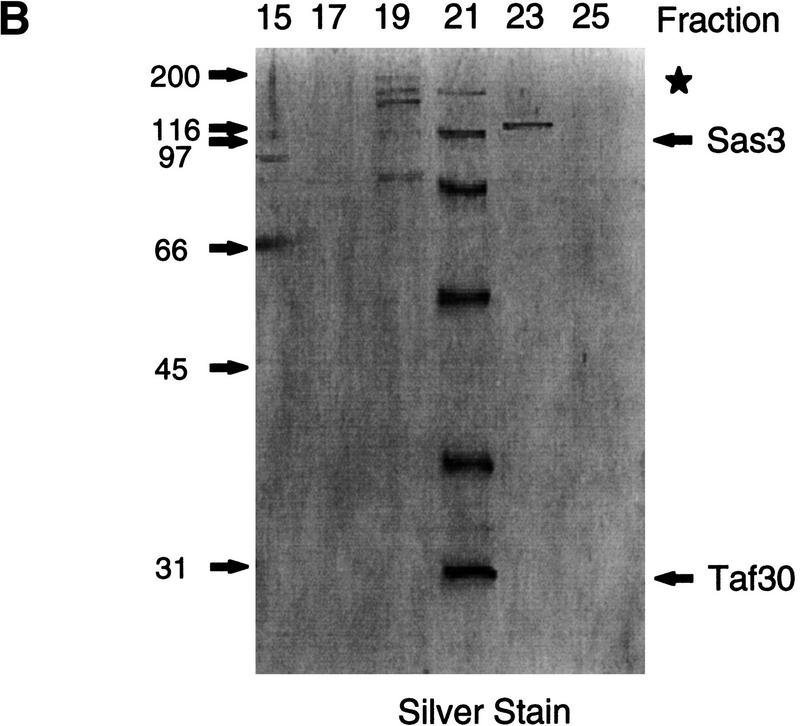
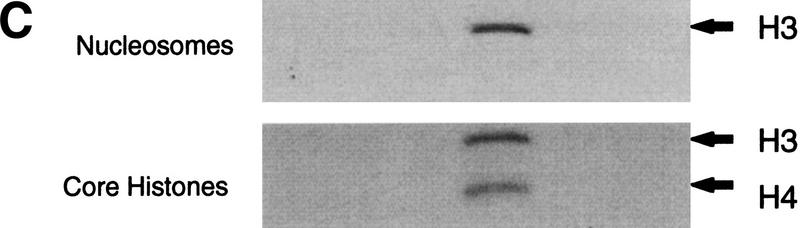
Purification scheme of the yeast NuA3 complex. Whole-cell extract from 90 liters of yeast strain CY396 were prepared for the biochemical purification of the NuA3 complex. (A) Purification scheme for the 0.4–0.5 MD nucleosomal HAT complex, NuA3 (see Materials and Methods for details). (B) Silver-stained gel from the final MiniQ column. (Arrows) Bands in the peak fraction (fraction 21) that yielded peptide sequences to Sas3 and TAF30. The identity of the other bands in fraction 21 is unconfirmed. (Star) Contaminating band that is also present in fraction 19. (C) Fluorogram of HAT assays on nucleosomes and core histones using 0.25 μl of MiniQ fractions show that the specificity of NuA3 is primarily histone H3 on nucleosomes and H3 and weakly H4 on core histones. The peak HAT activity is coincident with the protein peak. (Arrows) Positions of histones H3 and H4.