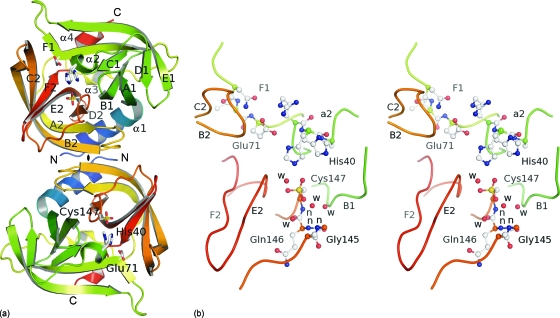
Fig. 3.
Crystal structure of EV-93 3Cpro. (a) Ribbon representation of the two molecules present in the asymmetric unit with the noncrystallographic 2-fold axis (♦) perpendicular to the plane. The protease folds into two antiparallel β barrels (in green and orange tones from the N to the C terminus), forming the chymotrypsin-like fold. The catalytic triad is highlighted as stick representations. (b) Active site of EV-93 3Cpro. Key residues are highlighted as ball-and-stick representations (stereo view). Main-chain amides forming the oxyanion hole are indicated by “n.”