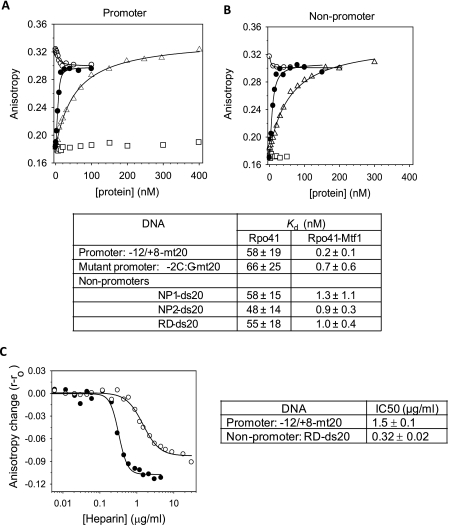
FIGURE 2.
Equilibrium binding of promoter and non-promoter DNAs by fluorescence anisotropy. The TMR-labeled -12/8-mt20 promoter (5 nm) in A and TMR-labeled RD-ds20 non-promoter (5 nm) in B was titrated with increasing Rpo41 (triangle), Mtf1 (square), Mtf1 after saturation with Rpo41 (blank circle), or with 1:1.2 ratio mixture of Rpo41 and Mtf1 (filled circle). Fluorescence anisotropy was measured after excitation at 555 nm and emission at 580 nm. Titration data were fit to Equation 1 (solid lines) to derive the Kd values listed in the table below with standard deviation errors from multiple independent measurements. C, heparin competition was monitored by fluorescence anisotropy of 5 nm TMR-labeled RD-ds20 (filled circle) or −12/+8-mt20 (blank circle) saturated with 5 nm Rpo41+Mtf1 (1:1.2) in the standard binding buffer at 20 °C. The adjacent table shows the heparin IC50 values.