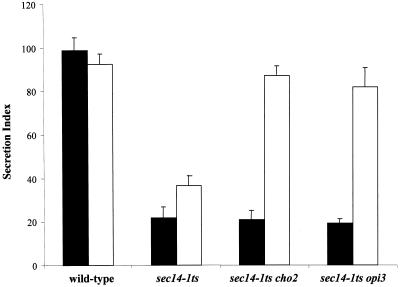
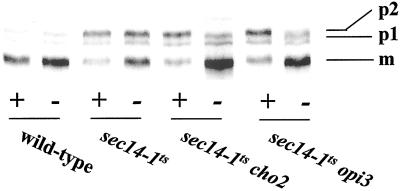
Figure 4.
(A) Efficiency of invertase secretion at 37°C for PtdEtn-methylation pathway mutants grown in I+C+ (black bars) and I+C− media (open bars). Secretion index was calculated as (extracellular invertase/total invertase × 100%) (Salama et al., 1990), and the values presented represent the averages of triplicate determinations from at least three independent experiments. The secretion indices of wild-type strain and sec14-1ts strain represent the secretory efficiency under Sec14p-proficient and Sec14p-deficient conditions, respectively. Strains used were CTY182 (wild type), CTY1-1A (sec14-1ts), CTY1374 (sec14-1ts cho2), CTY1375 (sec14-1ts opi3). (B) Trafficking of CPY through the secretory pathway to vacuole in I+C+ and I+C− media. Appropriate strains were grown at 26°C to early logarithmic phase in the indicated media (I+C+ media are indicated by “+” and I+C− media was indicated by “−” below the lane), shifted to 37°C for 2 h, and pulse-radiolabeled with 35S-amino acids at 37°C for 30 min. Radiolabeled CPY species were recovered, resolved, and quantitated as described (Fang et al., 1996). The p1 (ER), p2 (Golgi), and mature vacuolar (m) forms of CPY are indicated at right. Strains used included CTY182 (wild type), CTY1-1A (sec14-1ts), CTY1374 (sec14-1ts cho2), and CTY1375 (sec14-1ts opi3).