Abstract
During development, primordial germ cells (PGCs) migrate from the sites of their specification towards the region in which the future gonad develops. This cell migration requires polarization of PGCs and their responsiveness to external guidance cues. In zebrafish, the directed migration and polarization of PGCs are regulated independently, by the chemokine Cxcl12a and the Rho GTPase Rac1, respectively. However, the upstream signals controlling Rac activity in this context have not yet been identified. By investigating the role of G proteins in PGC migration, we found that signaling mediated by G protein subunits Gβγ is required to regulate cell polarization. PGCs that are defective for Gβγ signaling failed to polarize, and developed multiple protrusions in random locations, resembling the defects observed in PGCs with decreased Rac activity. These defects render PGCs incapable of migrating actively and responding to directional cues. FRET-based assays showed that PGCs require Gβγ signaling for polarized Rac activation and actin organization at the leading front, as well as for maintaining overall Rac levels in these cells. Conversely, overexpression of Gβγ in PGCs increases Rac activity. Our results indicate that during PGC migration in vivo, Gβγ signaling regulates Rac activity to control cell polarity, which is required for the responsiveness to chemokine signaling.
Keywords: Gβγ signaling, Cell migration, Primordial germ cells, Zebrafish
INTRODUCTION
Directed cell migration is involved in diverse processes during embryogenesis, immune response and disease (Friedl and Gilmour, 2009; Raz and Mahabaleshwar, 2009). Elucidating the underlying mechanisms is thus highly relevant for understanding both normal development and pathological conditions. In many species, primordial germ cells (PGCs) migrate a long distance from the sites of their specification to the region in which the future gonad develops (Richardson and Lehmann, 2010; Tarbashevich and Raz, 2010). They, thus, represent a powerful model for studying directed cell migration in the context of the developing organism.
Like other cell types, PGCs rely on both intrinsic cell motility and external guidance cues to migrate efficiently and precisely to their targets. In zebrafish, the chemokine Cxcl12a/Sdf1a guides PGC migration by activating its cognate G protein-coupled receptor Cxcr4b, which is expressed in PGCs (Doitsidou et al., 2002; Knaut et al., 2003; Boldajipour et al., 2008). However, PGCs lacking Cxcl12a or Cxcr4b are able to polarize and migrate actively, albeit non-directionally (Reichman-Fried et al., 2004; Kardash et al., 2010). Recent studies demonstrated that PGC polarization involves restricted Rac1 activation and the formation of actin-rich structures called ‘actin brushes’ at the leading edge (Kardash et al., 2010). Interestingly, the as yet unidentified signals that regulate the activation of Rac1 act independently of chemokine signaling (Kardash et al., 2010).
Investigating the function of G proteins in PGC migration, we found that the G protein subunits Gβγ are crucial for the efficient polarization of PGCs and their migration. Inhibition of Gβγ signaling resulted in a loss of polarity and the ectopic distributions of PGCs. Live-cell imaging indicated that, unlike PGCs with reduced chemokine signaling, PGCs with compromised Gβγ signaling failed to become polarized and to migrate actively, suggesting that Gβγ is involved in a chemokine-independent pathway. We further show that this phenotype stems from a lack of persistent Rac activation and a consequent failure to accumulate polymerized actin at the leading edge. Our data thus provide the first evidence that Gβγ signaling functions upstream of Rac to control the polarity and motility of PGCs in vivo, which is crucial for the response of PGCs to chemokine signaling.
MATERIALS AND METHODS
Zebrafish strains
AB*, Tuebingen, transgenic Tol-kop-EGFP-F-nos-3′UTR (Blaser et al., 2005) and medusaNY054 (Valentin et al., 2007) strains of zebrafish were used.
Cloning of zebrafish genes and expression of the Gβ isoforms
Zebrafish Gβ isoforms and Gαt1 (rod-type transducin α subunit) were cloned by RT-PCR. Accession numbers of gnb genes and the homology to their human counterparts are provided in supplementary material Table S1. Primers used for determining the expression of Gβ isoforms by RT-PCR are shown in supplementary material Table S2.
RNA expression constructs
For preferential expression in PGCs, open reading frames of genes of interest were fused to nos1-3′UTR, which has been shown to direct degradation and inhibit translation of the mRNA in the soma, whereas protecting the mRNA from degradation and ensuring protein translation in PGCs (Koprunner et al., 2001). Other constructs used in this study include: GFP-nos1-3′UTR (Koprunner et al., 2001); cxcl12a/pSP64T (Doitsidou et al., 2002); vasa-DsRed-nos1-3′UTR (Blaser et al., 2006); lifeact-eGFP-nos1-3′UTR, cytosolic Rac-FRET-nos1-3′UTR and dnRac-nos1-3′UTR (Kardash et al., 2010); and PHAKT-GFP-nos1-3′UTR (Dumstrei et al., 2004).
Time-lapse imaging, FRET imaging and analysis
Time-lapse imaging was performed as previously described (Dumstrei et al., 2004; Lin et al., 2005), using a Leica DMI 6000 or Zeiss LSM510 confocal microscope. FRET imaging was performed on PGCs expressing a cytosolic Rac biosensor (Kardash et al., 2010), using a Leica DMI 6000 microscope with a Dual-View image splitter and a 40×/NA 1.25 oil objective. YFP/CFP ratio images were generated and analyzed using the Metamorph and Image J software as described previously (Kardash et al., 2010).
In vivo chemoattractant assay
The previously described method (Doitsidou et al., 2002) was used, except that the cxcl12a mutant line medusaNY054 served as the host embryos (Valentin et al., 2007).
Statistical analysis
Data were compiled from at least two independent experiments and are presented as the mean±s.e.m. Unpaired two-tailed Student’s t-tests with unequal variance were used for statistical analyses. The number of embryos analyzed in each experiment is indicated in the figures.
RESULTS AND DISCUSSION
Inhibition of Gβγ signaling interferes with PGC migration
In investigating the role of Gβγ signaling in PGC migration, we initially targeted Gβ isoforms as they form functional complexes with Gγ proteins (Hamm, 1998; Smrcka, 2008; Dupre et al., 2009). Among the five Gβ isoforms (total nine members) identified in zebrafish (supplementary material Table S1), only genes encoding Gβ1 and Gβ4 (gnb1a, gnb1b, gnb4a, gnb4b) are uniformly expressed during gastrulation, the stage during which individual PGC migration takes place (supplementary material Fig. S1).
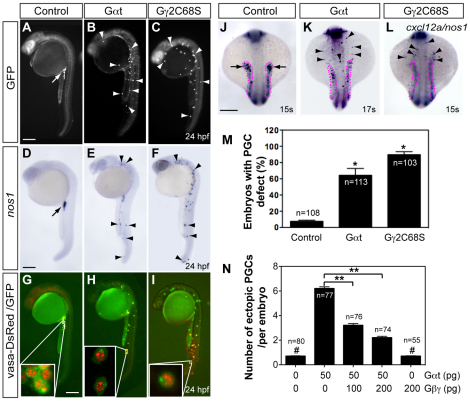
To inhibit the function of the Gβ1 and Gβ4 proteins, we first employed antisense morpholino oligonucleotides (MOs), which efficiently blocked the protein translation of their encoding GFP-tagged constructs (supplementary material Fig. S2). However, embryos injected with these MOs retained substantial amounts of endogenous Gβ (most likely the maternally deposited proteins, supplementary material Fig. S3), and PGC migration was not affected in these morphants (not shown). As an alternative strategy to interfere with Gβγ signaling, we expressed a Gβγ scavenger Gαt preferentially in PGCs. By sequestering free Gβγ, this protein blocks the activation of its downstream effectors (Federman et al., 1992). Additionally, we expressed a mutant Gγ2 (Gγ2C68S), which was recently shown to interfere with the membrane localization of Gβ and its function in PGCs (Mulligan et al., 2010). Whereas control PGCs expressing GFP were clustered in the region of the presumptive gonad at 24 hpf (Fig. 1A), PGCs expressing either Gαt or Gγ2C68S were randomly distributed throughout the embryo (Fig. 1B,C,M). Importantly, these ectopic PGCs exhibited appropriate expression of a PGC marker, nos1 (Koprunner et al., 2001) (Fig. 1D-F), and also perinuclear granule-specific localization of the Vasa-DsRed protein (Blaser et al., 2006) (Fig. 1G-I), suggesting that PGCs defective for Gβγ signaling are properly differentiated. Additionally, interference with Gβγ signaling in the PGCs did not affect the expression pattern of the cxcl12a transcript (Fig. 1J-L), indicating that the aberrant PGC distribution was not due to impaired Cxcl12a expression. Finally, co-expression of Gβ1γ2 in PGCs reduced the number of ectopic PGCs induced by Gαt (Fig. 1N) or Gγ2C68S (not shown) in a dose-dependent manner. Collectively, these results demonstrate that Gβγ-mediated signaling is essential for proper PGC migration.
Fig. 1.
Gβγ signaling is required for the migration, but not differentiation, of PGCs. Embryos injected with RNAs encoding GFP-nos1-3′UTR, either alone (control, 100 pg) or together with Gαt-nos1-3′UTR (50 pg) or Gγ2C68S-nos1-3′UTR (40 pg). Normally localized (arrow) and ectopic (arrowheads) PGCs are indicated. (A-I) Lateral views of embryos showing PGCs labeled with GFP (A-C), expressing nos1 detected by in situ hybridization (D-F) or labeled with vasa-DsRed and GFP (insets show high-magnification images) (G-I). (J-L) The expression of cxcl12a (outlined with magenta dots) and nos1 by in situ hybridization. Dorsal views. Scale bars: 200 μm. (M) The percentage of embryos with PGC migration defects, with the latter defined as more than three PGCs (detected by nos1 expression) per embryo present outside the presumptive gonad region at 24 hpf (Dumstrei et al., 2004). *P<0.01 versus control. (N) Total number of ectopic PGCs per embryo. **P<0.01; #P>0.5 versus control. Data are mean+s.e.m.
Gβγ signaling is essential for PGC polarization and motility
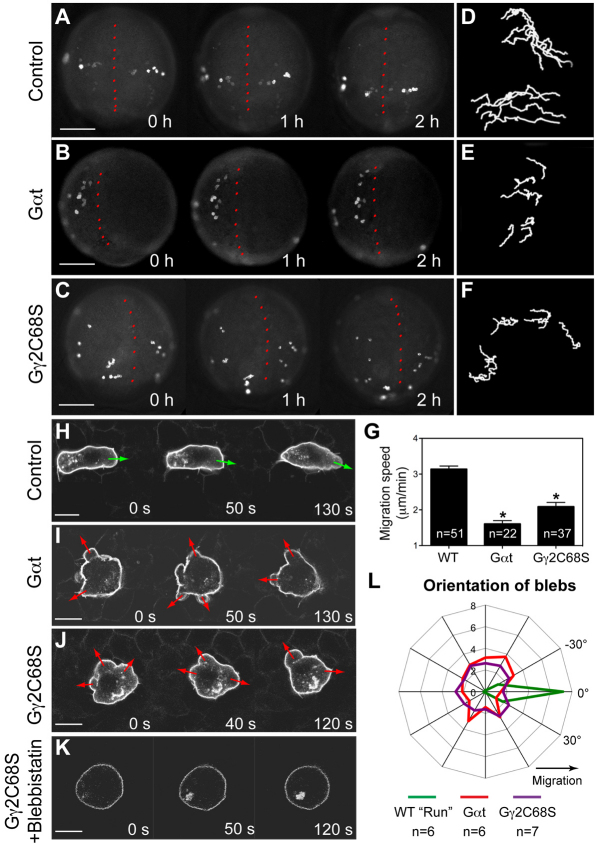
To determine the cellular basis for the migration phenotype of PGCs in which Gβγ signaling was disrupted, we monitored PGC migratory behaviors during their directional migration in response to Cxcl12a (8-10 hpf). Whereas control PGCs migrated actively towards their target (Fig. 2A; supplementary material Movie 1), Gαt- or Gγ2C68S-expressing PGCs migrated randomly without obvious direction (Fig. 2B,C; supplementary material Movie 1), as manifested by the shortened, less directed migration tracks when compared with controls (compare Fig. 2E-F to 2D). Moreover, the migration speed was significantly reduced in PGCs defective for Gβγ signaling (Fig. 2G). We next followed the effects of interfering with Gβγ signaling on individual cell morphology and behavior, using confocal time-lapse imaging. As previously described, migrating zebrafish PGCs alternate between two migratory modes: ‘run’ and ‘tumbling’ (Reichman-Fried et al., 2004). During the run period, PGCs migrate actively, constantly changing their positions relative to the neighboring somatic cells, while forming protrusion in the form of ‘blebs’ at the leading edge (Fig. 2H; supplementary material Movie 2). During the tumbling phase, by contrast, PGCs do not migrate, and they lose their polarity and extend blebs from multiple sites around the circumference of the cell (Reichman-Fried et al., 2004). Strikingly, we found that PGCs expressing either Gαt or Gγ2C68S failed to exhibit the migratory behaviors typical for the run phase. Rather, they constantly displayed tumbling-like behavior, forming multiple small bleb-like protrusions in random locations around the cell perimeter, with limited migration relative to the surrounding somatic cells (Fig. 2I,J; supplementary material Movie 2). Analyses of the distribution of blebs around the cell perimeter showed that during the run phase, 93% of the blebs in control PGCs formed in the direction of migration (±45°), whereas those in Gαt- or Gγ2C68S-expressing PGCs showed no such directional bias (Fig. 2L). These results indicate that Gβγ signaling is required for PGC polarization and motility, which appears to be required for Cxcl12a-guided directional PGC migration.
Fig. 2.
Gβγ signaling regulates the motility and polarity of PGC migration. (A-G) Epifluorescence time-lapse experiments performed on Tol-kop-EGFP-F-nos1-3′UTR embryos, with PGCs expressing membrane-bound GFP (memGFP) at low magnification (5× objective) at 8-10 hpf (supplementary material Movie 1). (A-C) Snapshots from time-lapse movies. Dorsal view; red dotted line indicates the notochord. (D-F) Representative tracks delineate PGC migration routes. (G) Average migration speed of PGCs. *P<0.01 versus control. Data are mean+s.e.m. (H-L) Confocal time-lapse imaging of PGCs expressing memGFP, taken at 8-10 hpf (supplementary material Movie 2). (H-K) Snapshots from movies of the control PGCs during the run phase (H) (green arrows indicate protrusion at the leading edge); Gαt- or Gγ2C68S-expressing PGCs (I,J) (red arrows indicate bleb-like protrusions); PGCs expressing Gγ2C68S in embryos treated with blebbistatin (K). Scale bars: 200 μm in A-C; 10 μm in H-K. (L) Orientation of blebs relative to the direction of PGC migration, as analyzed from four independent movies per group (10-second intervals, blebs grouped into 15° sectors).
The position of a bleb in PGCs is correlated with Ca2+-dependent myosin activity (Blaser et al., 2006). Consistent with this observation, Ca2+ elevations determined by using the Ca2+-sensitive fluorescence dye Oregon Green488 (Blaser et al., 2006) were detected in the multiple bleb-like protrusions that formed in PGCs expressing either Gαt or Gγ2C68S, and the Ca2+ levels were comparable with those in control embryos (supplementary material Fig. S4). Furthermore, treating embryos with the myosin II inhibitor blebbistatin (65 μM) completely eliminated the formation of blebs in PGCs expressing Gαt or Gγ2C68S (Fig. 2K and not shown). Thus, Gβγ inhibition did not affect the elevation in Ca2+ levels and the consequent activation of myosin, but rather abrogated the ability of the cell to confine myosin activation to the site within the cell where the leading edge should form.
Previous work showed that the absence of chemokine signaling does not abrogate the PGC polarization (at either the morphological or molecular level) or motility, although the migration becomes non-directional and less persistent (Doitsidou et al., 2002; Reichman-Fried et al., 2004; Kardash et al., 2010). Based on the lack of polarization in cells in which Gβγ function is disrupted, we suggest that Gβγ-mediated signaling is relevant for cell polarization and motility. Our data imply that, whereas the chemokine receptor Cxcr4b controls the directionality of PGC migration, other receptors or molecules such as receptor-independent activators of G protein signaling (AGS) (Cismowski, 2006) could control Gβγ activity, PGC polarization and motility. Notwithstanding our findings, we recognize that Cxcr4b could potentially modulate Gβγ activity and thereby contribute to maintaining PGC polarization during directed migration.
Gβγ signaling regulates actin cytoskeleton dynamics through Rac
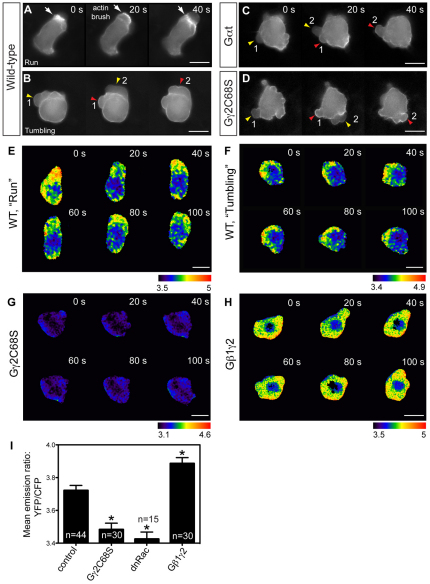
Previous work indicates that the polarity and motility of zebrafish PGCs are regulated by the persistent activation of Rac at the leading edge of migrating cells, and a resulting accumulation of F-actin in actin brush (Kardash et al., 2010). Interestingly, the behaviors of PGCs in which Rac signaling is inhibited are similar to those we observed in PGCs with defective Gβγ signaling, suggesting that Gβγ might regulate Rac activity. To test this notion, we first examined the effects of Gβγ inhibition on actin cytoskeleton dynamics in PGCs. As reported previously (Kardash et al., 2010), actin brushes were present at the leading edge of PGCs during the run, but not the tumbling, period (Fig. 3A,B; supplementary material Movie 3). Consistent with the lack of a run period in PGCs expressing either Gαt or Gγ2C68S (Fig. 2I,J), actin brushes failed to form in these cells (Fig. 3C,D; supplementary material Movie 3). Rather, transient actin accumulation was observed during periods of bleb retraction, similar to the features displayed by WT tumbling PGCs (Fig. 3B-D). These results indicate that Gβγ signaling regulates the formation of actin brushes and the distribution of blebs, but not the formation of blebs.
Fig. 3.
Gβγ signaling regulates actin cytoskeleton dynamics through RhoGTPase Rac. (A-D) Snapshots from epifluorescence time-lapse movies taken of embryos at 8-10 hpf, with actin cytoskeleton dynamics revealed using Lifeact-GFP labeling, in wild-type PGCs and PGCs expressing Gαt or Gγ2C68S (supplementary material Movie 3). White arrows indicate actin brushes; arrowheads indicate blebs. Numbers follow the same bleb. Actin labeling is not obvious in newly formed blebs (yellow arrowheads), but becomes prominent when blebs begin to shrink (red arrowheads). (E-I) Rac activity of PGCs expressing a cytosolic RacFRET biosensor, as determined by time-lapse analysis at 8-9 hpf. (E-H) Pseudocolored images of PGCs showing the ratio of emission from yellow fluorescent protein (YFP) to emission from cyan fluorescent protein (CFP). Wild-type PGCs during the run or tumbling phase (E,F; supplementary material Movie 4); PGCs expressing Gγ2C68S or wild-type Gβ1γ2 (G,H; supplementary material Movie 5). Scale bars: 10 μm. (I) Average Rac activity (mean YFP/CFP emission ratio for the whole cell) in control PGCs and in PGCs expressing Gγ2C68S, dnRac or Gβ1γ2. Total number of PGCs analyzed is indicated. *P<0.01 versus control. Data are mean+s.e.m.
Next, we determined whether Gβγ-mediated regulation of actin brush formation involves the control of Rac activity in PGCs. As previously reported (Kardash et al., 2010), in wild-type embryos, Rac was persistently activated at the leading edge of the actively migrating PGCs, but only transiently activated in tumbling PGCs (Fig. 3E,F; supplementary material Movie 4). Nevertheless, average overall Rac activity was comparable in wild-type PGCs at the run and tumbling periods (not shown). Notably, disruption of Gβγ signaling not only diminished the persistence of Rac activation, but also decreased the overall level of Rac activity in PGCs (Fig. 3G,I; supplementary material Movie 5), similar to the results obtained when a dominant-negative form of Rac was expressed in PGCs (Kardash et al., 2010) (Fig. 3I). Conversely, overexpression of Gβ1γ2 in PGCs significantly increased Rac activity (Fig. 3H,I; supplementary material Movie 5). Taken together, these findings indicate that Gβγ signaling is required for Rac activation in PGCs, and that it promotes actin brush formation to control cell polarization.
It would be important to investigate further how Gβγ regulates Rac activity in PGCs and how the Gβγ function itself is regulated. Gβγ can stimulate Rac activity by activating PI3Ks (Andrews et al., 2007; Wang, 2009) or Rac-specific RhoGEFs, for example, PLEKHG2 and p-Rex1 (Welch et al., 2002; Ueda et al., 2008). However, we showed that blocking Gβγ signaling did not affect PI3K activation in PGCs (supplementary material Fig. S5). Thus, it will be worth testing the roles of those RhoGEFs in PGC migration.
Gβγ signaling is essential for the response of PGCs to the chemokine Cxcl12a
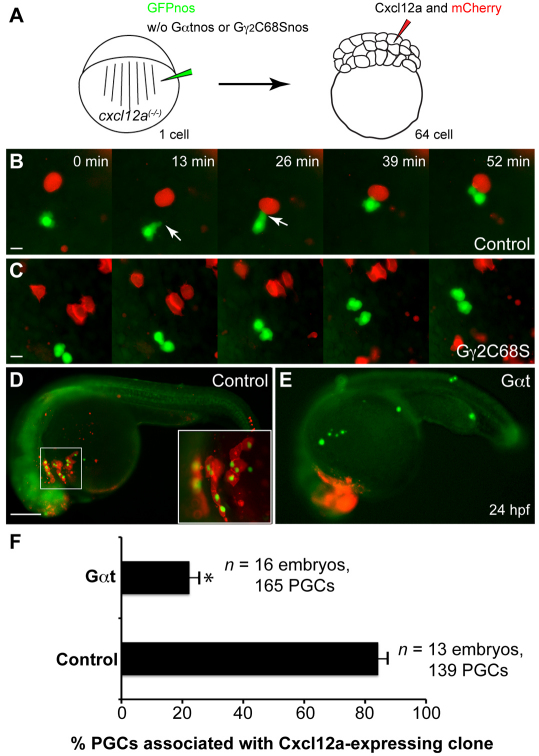
PGC migration follows the dynamic changes in cxcl12a expression during development (Doitsidou et al., 2002). Thus, it is possible that the decreased motility of PGCs defective for Gβγ signaling impairs their ability to reach the cxcl12a-expressing domains. To examine this possibility, we performed an in vivo mosaic chemoattractant assay in which Cxcl12a was expressed in cells near PGCs and assessed how PGCs respond to this strong source of Cxcl12a. As expected, control PGCs were attracted to the ectopically located Cxcl12a-expressing cells (Doitsidou et al., 2002), forming polarized protrusions in the direction of the Cxcl12a source as they migrated (Fig. 4B; supplementary material Movie 6). By contrast, PGCs expressing Gγ2C68S or Gαt extended multiple protrusions and failed to polarize and to migrate towards the nearby Cxcl12a-expressing cells (Fig. 4C; supplementary material Movie 6). Accordingly, 84±3% of control PGC (139 PGCs, 13 embryos) colocalized with Cxcl12a/mCherry-expressing cells at 24 hpf, whereas only 22±3% of Gαt-expressing PGCs (165 PGCs, 16 embryos) did so (Fig. 4D-F and not shown; supplementary material Fig. S6). These findings indicate that Gβγ signaling is essential for PGCs to polarize, and thus for their chemotactic response towards Cxcl12a.
Fig. 4.
Gγ2C68S- and Gαt-expressing PGCs fail to respond to Cxcl12a. (A) Schematic illustration outlining the chemoattractant assay. cxcl12a(–/–) embryos at the one-cell stage were injected with gfp-nos mRNA (to label PGCs) alone (control) or together with mRNAs encoding either Gγ2C68S-nos1-3′UTR or Gαt-nos1-3′UTR. At the 64-cell stage, a single cell at the animal pole was co-injected with RNAs encoding Cxcl12a and mCherry (tracer). (B,C) Snapshots from time-lapse movies showing the positions of PGCs (green) and Cxcl12a-expressing cells (red) in control embryos (B, supplementary material Movie 6) or in embryos injected with Gγ2C68S-nos1-3′UTR RNA (C, supplementary material Movie 6) at 8-9 hpf. White arrows indicate protrusions. (D,E) Representative embryos at 24 hpf showing localization of control or Gαt-expressing PGCs (green) and the Cxcl12a-expressing cells (red). Inset indicates high-magnification image of the boxed area. Scale bars: 10 μm in B,C; 200 μm in D,E. (F) Quantitative analysis of PGC colocalization with Cxcl12a-expressing cells at 24 hpf. *P<0.01 versus control. Data are mean+s.e.m.
Overall, our data reveal a previously unknown role of Gβγ signaling in regulating PGC polarization, via the control of Rac activity. Future work is required to dissect the molecular mechanisms by which Gβγ activate Rac in this context, and to determine whether this signaling cascade is conserved in other migrating cell types and in PGCs of other organisms.
Supplementary Material
Acknowledgments
We thank Lila Solnica-Krezel, Christina Speirs and Diane Sepich for critical comments.
Footnotes
Funding
This study is supported by grants from the National Institutes of Health (NIH) [K99R00RR024119 to F.L. and R01GM094255 to S.C.]; by the American Heart Foundation [AHA10GRNT3620015 to S.C.]; by the Deutsche Forschungsgemeinschaft and by the Max-Planck Society (E.R. and E.K.). Deposited in PMC for release after 12 months.
Competing interests statement
The authors declare no competing financial interests.
Supplementary material
Supplementary material available online at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.073924/-/DC1
References
- Andrews S., Stephens L. R., Hawkins P. T. (2007). PI3K class IB pathway in neutrophils. Science STKE 2007, cm3 [DOI] [PubMed] [Google Scholar]
- Blaser H., Eisenbeiss S., Neumann M., Reichman-Fried M., Thisse B., Thisse C., Raz E. (2005). Transition from non-motile behaviour to directed migration during early PGC development in zebrafish. J. Cell Sci. 118, 4027–4038 [DOI] [PubMed] [Google Scholar]
- Blaser H., Reichman-Fried M., Castanon I., Dumstrei K., Marlow F. L., Kawakami K., Solnica-Krezel L., Heisenberg C.-P., Raz E. (2006). Migration of zebrafish primordial germ cells: a role for myosin contraction and cytoplasmic flow. Dev. Cell 11, 613–627 [DOI] [PubMed] [Google Scholar]
- Boldajipour B., Mahabaleshwar H., Kardash E., Reichman-Fried M., Blaser H., Minina S., Wilson D., Xu Q., Raz E. (2008). Control of chemokine guided cell migration by ligand sequestration. Cell 132, 463–473 [DOI] [PubMed] [Google Scholar]
- Cismowski M. J. (2006). Non-receptor activators of heterotrimeric G-protein signaling (AGS proteins). Semin. Cell Dev. Biol. 17, 334–344 [DOI] [PubMed] [Google Scholar]
- Doitsidou M., Reichman-Fried M., Stebler J., Koprunner M., Dorries J., Meyer D., Esguerra C. V., Leung T., Raz E. (2002). Guidance of primordial germ cell migration by the chemokine SDF-1. Cell 111, 647–659 [DOI] [PubMed] [Google Scholar]
- Dumstrei K., Mennecke R., Raz E. (2004). Signaling pathways controlling primordial germ cell migration in zebrafish. J. Cell Sci. 117, 4787–4795 [DOI] [PubMed] [Google Scholar]
- Dupre D. J., Robitaille M., Rebois R. V., Hebert T. E. (2009). The role of Gβγ subunits in the organization, assembly, and function of GPCR signaling complexes. Annu. Rev. Pharmacol. Toxicol. 49, 31–56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Federman A. D., Conklin B. R., Schrader K. A., Reed R. R., Bourne H. R. (1992). Hormonal stimulation of adenylyl cyclase through Gi-protein βγ subunits. Nature 356, 159–161 [DOI] [PubMed] [Google Scholar]
- Friedl P., Gilmour D. (2009). Collective cell migration in morphogenesis, regeneration and cancer. Nat. Rev. Mol. Cell Biol. 10, 445–457 [DOI] [PubMed] [Google Scholar]
- Hamm H. E. (1998). The many faces of G protein signaling. J. Biol. Chem. 273, 669–672 [DOI] [PubMed] [Google Scholar]
- Kardash E., Reichman-Fried M., Maitre J. L., Boldajipour B., Papusheva E., Messerschmidt E. M., Heisenberg C. P., Raz E. (2010). A role for Rho GTPases and cell-cell adhesion in single-cell motility in vivo. Nat. Cell Biol. 12, 47–53 [DOI] [PubMed] [Google Scholar]
- Knaut H., Werz C., Geisler R., Nusslein-Volhard C. (2003). A zebrafish homologue of the chemokine receptor Cxcr4 is a germ-cell guidance receptor. Nature 421, 279–282 [DOI] [PubMed] [Google Scholar]
- Koprunner M., Thisse C., Thisse B., Raz E. (2001). A zebrafish nanos-related gene is essential for the development of primordial germ cells. Genes Dev. 15, 2877–2885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin F., Sepich D. S., Chen S., Topczewski J., Yin C., Solnica-Krezel L., Hamm H. (2005). Essential roles of Gα12/13 signaling in distinct cell behaviors driving zebrafish convergence and extension gastrulation movements. J. Cell Biol. 169, 777–787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulligan T., Blaser H., Raz E., Farber S. A. (2010). Prenylation-deficient G protein gamma subunits disrupt GPCR signaling in the zebrafish. Cell Signal. 22, 221–233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raz E., Mahabaleshwar H. (2009). Chemokine signaling in embryonic cell migration: a fisheye view. Development 136, 1223–1229 [DOI] [PubMed] [Google Scholar]
- Reichman-Fried M., Minina S., Raz E. (2004). Autonomous modes of behavior in primordial germ cell migration. Dev. Cell 6, 589–596 [DOI] [PubMed] [Google Scholar]
- Richardson B. E., Lehmann R. (2010). Mechanisms guiding primordial germ cell migration: strategies from different organisms. Nat. Rev. Mol. Cell Biol. 11, 37–49 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smrcka A. V. (2008). G protein βγ subunits: central mediators of G protein-coupled receptor signaling. Cell Mol. Life Sci. 65, 2191–2214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarbashevich K., Raz E. (2010). The nuts and bolts of germ-cell migration. Curr. Opin. Cell Biol. 22, 715–721 [DOI] [PubMed] [Google Scholar]
- Ueda H., Nagae R., Kozawa M., Morishita R., Kimura S., Nagase T., Ohara O., Yoshida S., Asano T. (2008). Heterotrimeric G protein βγ subunits stimulate FLJ00018, a guanine nucleotide exchange factor for Rac1 and Cdc42. J. Biol. Chem. 283, 1946–1953 [DOI] [PubMed] [Google Scholar]
- Valentin G., Haas P., Gilmour D. (2007). The chemokine SDF1a coordinates tissue migration through the spatially restricted activation of Cxcr7 and Cxcr4b. Curr. Biol. 17, 1026–1031 [DOI] [PubMed] [Google Scholar]
- Wang F. (2009). The signaling mechanisms underlying cell polarity and chemotaxis. Cold Spring Harb. Perspect. Biol. 1, a002980–a002980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welch H. C., Coadwell W. J., Ellson C. D., Ferguson G. J., Andrews S. R., Erdjument-Bromage H., Tempst P., Hawkins P. T., Stephens L. R. (2002). P-Rex1, a PtdIns(3,4,5)P3- and Gβγ-regulated guanine-nucleotide exchange factor for Rac. Cell 108, 809–821 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.