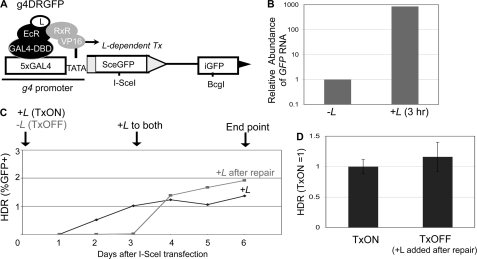
FIGURE 4.
The frequency of HDR is not affected by DSB transcription context. A, schematic of g4DRGFP. The SceGFP cassette is shown downstream of GAL4 recognition sites and a minimal TATA promoter (g4). HDR of an I-SceI-induced DSB using iGFP as the template restores a GFP+ cassette, as shown in Fig. 3A. Shown are the regulator proteins EcR-GAL4-DBD and RxR-VP16, which require the presence of a soluble ligand (L) bound to EcR to induce transcription. B, addition of L leads to rapid induction of mRNA expression from the g4 promoter. The g4DRGFP reporter was integrated into two cell lines that stably express the EcR-GAL4-DBD and RxR-VP16 proteins (HEK293-A7 and U2OS-R1). Using the g4DRGFP HEK293-A7 cell line, RNA was isolated after a 3-h incubation in media containing +L or untreated (−L). Samples were reverse transcribed prior to quantitative PCR. Shown is the relative abundance of GFP RNA, based on 2ΔΔCt values normalized to actin primers and relative to the untreated condition (−L = 1). C, time course to establish treatment conditions for TxON and TxOFF samples. The g4DRGFP HEK293-A7 cell line was transfected in the presence (TxON) or absence (TxOFF) of L for 3 days, at which time L was added to all the samples for an additional 3 days of culture (6 days total). Shown is the frequency of GFP+ cells from these samples each day post-transfection. D, DSB transcription context has no effect on the frequency of HDR. An expression vector for I-SceI was transfected in the g4DRGFP U2OS-R1 cell line under the TxON and TxOFF conditions described in C. Shown is the frequency of HDR for both Tx conditions from this experiment, relative to the TxON condition (TxON = 1).