Abstract
Derived sabercats had craniomandibular morphologies that in many respects were highly different from those of extant felids, and this has often been interpreted functionally as adaptations for predation at extreme gape angles with hypertrophied upper canines. It is unknown how much of this was a result of intraspecific postnatal ontogeny, since juveniles of sabercats are rare and no quantitative study has been made of craniomandibular ontogeny. Postnatal ontogenetic craniomandibular shape changes in two morphologically derived sabercats, Smilodon fatalis and S. populator, were analysed using geometric morphometrics and compared to three species of extant pantherines, the jaguar, tiger, and Sunda clouded leopard. Ontogenetic shape changes in Smilodon usually involved the same areas of the cranium and mandible as in extant pantherines, and large-scale modularization was similar, suggesting that such may have been the case for all felids, since it followed the same trends previously observed in other mammals. However, in other respects Smilodon differed from extant pantherines. Their crania underwent much greater and more localised ontogenetic shape changes than did the mandibles, whereas crania and mandibles of extant pantherines underwent smaller, fewer and less localised shape changes. Ontogenetic shape changes in the two species of Smilodon are largely similar, but differences are also present, notably those which may be tied to the presence of larger upper canines in S. populator. Several of the specialized cranial characters differentiating adult Smilodon from extant felids in a functional context, which are usually regarded as evolutionary adaptations for achieving high gape angles, are ontogenetic, and in several instances ontogeny appears to recapitulate phylogeny to some extent. No such ontogenetic evolutionary adaptive changes were found in the extant pantherines. Evolution in morphologically derived sabercats involved greater cranial ontogenetic changes than among extant felids, resulting in greatly modified adult craniomandibular morphologies.
Introduction
Felids are some of the most anatomically specialized of all mammals for vertebrate predation, and the extinct sabertoothed felids (Felidae: Machairodontinae) included some of the most craniodentally specialized of all mammalian carnivores. Among the most morphologically derived were Smilodon spp., in particular the two great Pleistocene species Smilodon fatalis and S. populator from North and north-western South America, and eastern South America, respectively [1], [2]. Recently, S. populator has also been documented from Venezuela [3] and as far south as Chilean Patagonia [4]. Some [5], [6] have argued that they represent a single species, to which the name S. populator [7] would then apply, but this view has not gained ground, and there are several character differences between them pertaining, for instance, to cranial form and canine size and shape [1], [2]. A recent phylogenetic study found a further difference in that the P4 protocone is more reduced in S. populator [8].
Smilodon were the quintessential “sabertoothed tigers”, and craniodentally, they differed markedly from all extant felids, having, among others, greatly elongate and lateromedially flattened upper canines and greatly reduced (incisiform) lower canines; a tall, compact cranial shape; an enormously developed mastoid process and a greatly reduced paroccipital process, implying great enhancement of the cranial flexor musculature; shorter and more massive zygomatic arches; prognatheous incisors; lowered glenoid joint; and a mandible with a rectangular and straight horizontal ramus, verticalized mandibular symphysis, greatly reduced coronoid process, laterally rotated lower carnassials, and a deflected retroarticular process [2], [9], [10]–[17]. Their bite forces relative to body size were lower than among extant large felids [18]–[21], and their mandibular morphology indicated significant differences in predatory behaviour from extant felids [10], [21], [22]. The tall skull shape was likely an adaptation for re-orientation of the major mandibular adductor muscles to facilitate more efficient biting even at high gape angles and to partially compensate for the reduction in mandibular adductor muscle size [9], [10], [19]–[21]. The prognatheous incisors are thought to have facilitated carcass dismembering with very large upper canines, and perhaps as an auxiliary anchor point during predatory biting [2], [10], [11].
Extant adult felids are hyper-carnivorous and, as such, undergo a dramatic change in diet from suckling to a mechanically demanding ecology as vertebrate predators, necessitating high bite forces [23]. This transition in behaviour and functional morphology could be accompanied by large changes in craniomandibular morphology, but skull ontogeny in felids has received relatively little attention, and most studies have focused on dental eruption and subsequent wear, and its implication for age determination [24]–[31]. Other studies of postnatal morphology and size changes in lynxes (Lynx spp.) [32], [33]; margay (Leopardus wiedii) [34]; caracal (Caracal caracal) [35]; jaguar (Panthera onca) [36]; tiger (Panthera tigris) [37]; and lion (Panthera leo) [38] have been primarily descriptive. To date, the only species in which craniomandibular ontogeny has been specifically studied is the puma (Puma concolor) using both qualitative [39] and quantitative [40] approaches. Another study involved adult size and morphology changes [41], since growth in felids proceeds for years beyond sexual maturity, as in many other mammal species [42]–[44]. A recent study involved allometric cranial proportions in extant lions to address the nature of the Ice Age pantherine Panthera atrox, but did not address ontogeny as such [45].
Since some sabercats were even more craniomandibularly and dentally specialized than extant felids, studies of craniomandibular ontogeny could potentially shed light onto how sabercat adults came to be so morphologically different from modern felids. Unfortunately, postnatal ontogenetic changes in sabercat craniomandibular morphologies are even more poorly known than those of extant felids, and juveniles are known for only a few species, for instance Homotherium serum [46], [47] and Smilodon fatalis [48]; additionally, previous authors have focused on descriptive comparisons between juveniles and adults. In the voluminous collections of the Late Pleistocene fauna from the La Brea tar seeps, juvenile Smilodon fatalis from several dental ages are known [48], and juveniles appear not to have been rare, since Miller [49], in a study of 918 skulls, estimated that 16.6% represented juveniles; 23.2% were young adults; 17.2% were adults; and 8.5% were old individuals; 34.5% were indeterminable, but were mostly adult specimens who could not be classified as young adults, adults, or old. Most juvenile skulls are incomplete, however, owing to poor ossification and the movement of the tar. Other than Merriam & Stock [48] and Tejada-Flores & Shaw [50], the S. fatalis juveniles have received relatively little literature exposure.
Smilodon populator was even larger [51] and in some respects more specialized than S. fatalis, for instance in having an even larger C1 [1], [2], but only fragmentary and incomplete juvenile skulls have been reported of this species [52]. However, an exquisitely preserved juvenile specimen is present at the Naturhistoriska riksmuseet in Stockholm, (Fig. 1,2; Fig. S1), and other than a meticulous fitting together of a few skull bones around the nasals, owing to their not being co-ossified yet, the specimen is in excellent and near undistorted condition. The deciduous dentition is fully erupted and there are wear facets on the carnassials (dP3; dP4), in particular the dP4. The permanent dentition has not yet erupted, but the permanent carnassials (P4; M1) can be seen inside their alveoli. The left dI3 has fallen out indicating that the permanent incisors may soon commence replacing the deciduous incisors, but no permanent incisors are visible. The dC1 have unworn crenulations along their posterior carinae. The dC1 is relatively smaller and more incisiform than the C1.
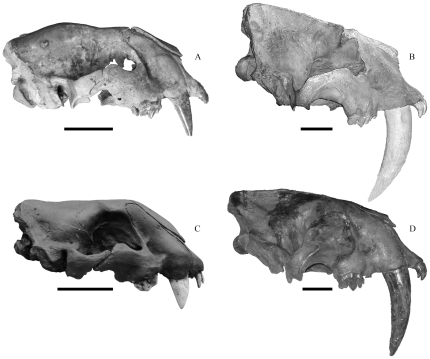
Figure 1. A comparison of crania in juvenile and adult Smilodon spp. scaled to the same condylobasal length.
A, juvenile S. populator (NRM); B, adult S. populator (BM, cast); C, juvenile S. fatalis (PC coll.; from Dinocasts); D, adult S. fatalis (LACMHC2001-173). Scale bars equal 5 cm.
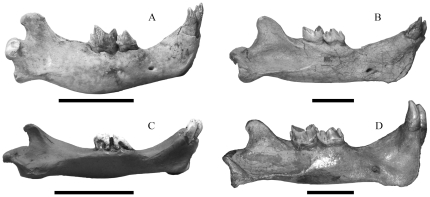
Figure 2. A comparison of mandibles in juvenile and adult Smilodon spp. scaled to the same overall length.
A, juvenile S. populator (NRM); B, adult S. populator (CN52); C, juvenile S. fatalis (PC coll.; from Dinocasts); D, adult S. fatalis (LACMHC3E-350). The juvenile S. fatalis mandible is an example of a gracile mandible type whereas the adult mandible is of a more robust type, but variation in juveniles and adults in the La Brea material is substantial. Scale bars equal 5 cm.
In this paper, I explore the postnatal ontogenetic changes in Smilodon relative to some extant pantherine felids using geometric morphometric analyses of crania and mandibles, in order to document the nature and magnitude of craniomandibular shape changes during ontogeny in Smilodon; the similarities and differences, if any, in ontogenetic shape changes between S. fatalis and S. populator; and the nature and magnitude of ontogenetic craniomandibular shape changes in the comparative sample of extant pantherines. This will allow a detailed comparison of postnatal ontogenetic craniomandibular shape chances in derived sabertoothed felids relative to extant large felids, and could potentially throw light onto sabercat evolution and to which extant ontogenetic shape changes may mimic evolutionary relationships.
Materials and Methods
Data
The database for analysis consisted of the juvenile Smilodon populator from Naturhistoriska riksmuseet (NRM) in Stockholm, and 6 crania and 8 mandibles from adult specimens from the Zoological Museum in Copenhagen (CN), the Museum National d'Histoire Naturelle in Paris (MNHN), and the Natural History Museum in London (NHM); three juvenile crania and four juvenile mandibles and 8 adult specimens of S. fatalis from the Hancock collection at Los Angeles County Museum (LACMHC), the MNHN in Paris, as well as the author's private collection of casts (PC coll.; a juvenile from Dinocasts, and BC-018T, cast of LACMHC2001-249 from Bone Clones) (Fig. 1,2). The sexes of the included Smilodon specimens were unknown, but Smilodon fatalis appears to show little, if any, sexual dimorphism [1], [31], which is in contrast to most extant felids, where size-dimorphism is often present, and occasionally also morphological dimorphism [37], [53]–[59].
As such, the sexes were also mixed in the comparative samples of extant large felids. For comparison were used three pantherines, the Sunda clouded leopard (Neofelis diardi; 4 juveniles [1♂; 3♀], and 25 adults [14♂; 11♀]), the jaguar (Panthera onca; 4 juveniles [2♂; 2♀], and 50 adults [30♂; 20♀]), and the tiger (P. tigris; 4 juveniles [2♂; 2♀], and 50 adults [26♂; 24♀]) (Fig. 3,4). The specimens were from the CN in Copenhagen, NHM in London, MNHN in Paris, the National Museum of Natural History (Naturalis) in Leiden (RMNH), and the author's private collection. More juveniles of Smilodon fatalis and the extant large felids were available, but were not included since only juveniles which corresponded in dental ontogenetic stage to the juvenile of S. populator were included to ensure comparable ages for analyses of craniomandibular ontogenetic changes.
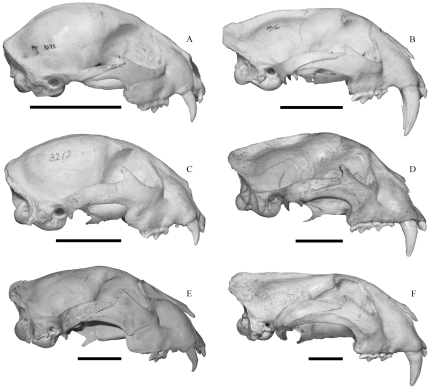
Figure 3. A comparison of crania in juvenile and adult extant felids scaled to the same condylobasal length.
A, Neofelis diardi juvenile (RMNH3518); B, Neofelis diardi adult (RMNH 71/41); C, Panthera onca juvenile (RMNH3212); D, Panthera onca adult (CN842); E, Panthera tigris juvenile (CN5654); F, Panthera tigris adult (RMNH “n”). Scale bars equal 5 cm.
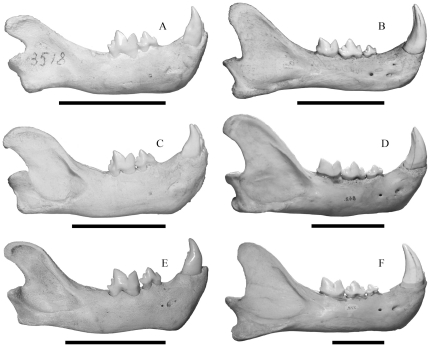
Figure 4. A comparison of mandibles in juvenile and adult extant felids scaled to the same overall length.
A, Neofelis diardi juvenile (RMNH3518); B, Neofelis diardi adult (RMNH “a”); C, Panthera onca juvenile (RMNH3212); D, Panthera onca adult (CN842); E, Panthera tigris juvenile (CN1523); F, Panthera tigris adult (CN772). Scale bars equal 5 cm.
In Smilodon fatalis [48], [50] and Homotherium serum [46] the postcanine dental formula in juveniles is dP3, dP4 (and P3, P4, and M1 in adults) and dP3 and dP4 (P4, M1 in adults; Homotherium have a reduced P3, and this tooth is frequently absent in Smilodon). In juveniles, the functional carnassials are dP3 and dP4, respectively, and the adult carnassials are represented by a small, molariform dP4, whereas the dM1 does not occur. The deciduous and permanent dentition formula is identical in Smilodon populator to S. fatalis. The deciduous and permanent dental formulae for the sabercats are also similar to those of extant felids such as leopards [25], lions [27], tigers [37], snow leopards [60], lynxes [29], and other felids [26], except that Smilodon and Homotherium lack the dP2 and P2.
In the Smilodon populator juvenile and, accordingly, the other juveniles as well, the entire deciduous dentition is fully erupted, and there is no sign of any of the permanent dentition, not even the incisors, which are the first to erupt in sabercats [46], [48], [50] and extant felids [25]–[27], [29], [37], [60]. Tejada-Flores & Shaw [50] divided a growth series of Smilodon fatalis from La Brea into five stages for the premaxillae; ten stages for the maxillae; and seven stages for the dentaries. The differences were due to the material often being unassociated. In stage I, all deciduous teeth in premaxilla, maxilla, and mandible were fully erupted, except the dC1. Premaxilla and maxilla stages II–IV were not coincident with the same stages in the (un-associated) mandibles, because of the long eruption time of dC1. In stage V, all permanent incisors had erupted and the permanent carnassials were in occlusion, and this is too advanced an ontogenetic stage relative to the juveniles used in this analysis. In S. fatalis, the dC1 is fully erupted when P4 is almost erupted, and the C1 begins to emerge when the P4 is fully erupted and the dP3 is about to be shed. This is, as noted, a considerably later ontogenetic stage than the juveniles used in the current analysis. It would appear that the Smilodon juveniles in the current analysis correspond to stages II–III.
Age-wise the Smilodon juveniles would appear to correspond to Rawn-Schatzingers [46] late stage III (all deciduous incisors and dP3 and dP3 fully erupted with well developed roots and dP4 partly erupted) to early stage IV (all deciduous teeth fully erupted with well-developed roots; wear facets are obvious with concomitant loss of serrations; and deciduous incisors are beginning to be replaced). However, replacement of deciduous incisors has not yet occurred in the juveniles of the current study. Rawn-Schatzinger [46] estimated the age of stage III at 3–4 months and the age of stage IV at 5–12 months, implying that the Smilodon juveniles could have been around 4–5 months old. Extant lions with fully erupted deciduous dentition are around 4–5 months old as well, and the first parts of the permanent dentition (incisors) begin erupting at around 7–9 months of age [27], which is similar in tigers [37].
Geometric morphometrics
To analyze craniomandibular ontogenetic shape changes, digital shape analysis was performed using the Thin Plate Splines (TPS) approach. The TPS function decomposed by its partial warps is a 2D model for analysing shape deformations of structures compared to a predefined reference shape configuration [61]–[63], and may be regarded as a modern model-equivalent of the Cartesian transformation grids for studying evolutionary shape changes originally proposed by Sir D'Arcy Wentworth Thompson (1860–1948) in 1917 [64]. The reference configuration is non-arbitrary and non-local, defining the point of tangency between shape space and approximating tangent space in the computation of the thin plate splines; by default, it is oriented by its principal components axis. It is computed by the generalized orthogonal least squares Procrustes superimposition procedure [62], [65], [66] as a mean reference shape of the included specimens, and, accordingly, it has no morphological, ontogenetic, or phylogenetic significance.
Bending energy is a function of the distance between individual landmarks of the reference configuration and any given specimen being analysed, and it increases progressively with increased localization, i.e. changes affecting only a limited area [62], [63], [67], [68]. The above implies that morphological changes, which may superficially appear to be localised to a particular region and, accordingly, in evolutionary terms uncoupled from the evolution of other structures, are interpreted as localised only if it is required; that is, when adjacent landmarks display contrasting displacements [62], [63], [68], [69]. Accordingly, minimising the amount of spatially localised information leads to more parsimonious interpretation, in that characters are interpreted as evolving independently only when the data require it.
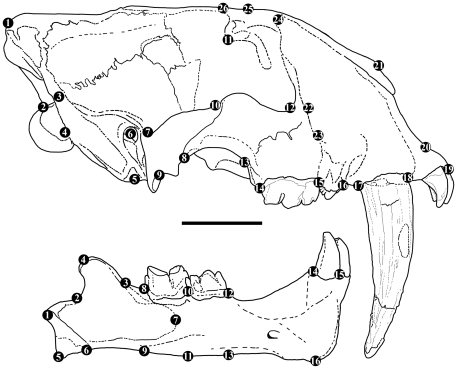
Twenty-six landmarks were digitized onto each skull and 16 landmarks were digitized onto each mandible in the program tpsDig [70] (Fig. 5). Comparative multivariate analyses on Relative Warp scores, incorporating all included specimens [62], [63], were conducted in tpsRelw [71]. At an α = 0, as used in this study, a Relative Warp analysis is a Principal Component Analysis of shape changes based on the covariance matrix of Partial Warp scores [62], [63], [72]–[74]. The Relative Warps are orthogonal and uncorrelated, and account for virtually all of the variation in the sample.
Figure 5. Illustration of a skull of Smilodon fatalis (LACMHC2001–3) showing landmarks used in the analysis.
Landmarks on the cranium are: 1, top of cranium at the junction of sagittal and nuchal crests; 2, top of occipital condyle; 3, dorsal extent of the mastoid musculature; 4, apex of paroccipital process; 5, apex of mastoid process; 6, centre of external auditory meatus; 7, posterior base of zygomatic arch; 8, ventral junction of jugal-squamosal suture; 9, centre of mandibular condyle; 10, base of postorbital process (jugal portion); 11, apex of postorbital process (frontal portion); 12, centre of orbital aperture; 13, junction of jugal-maxilla suture; 14, posterior, and 15, anterior edge of P4 (dP4 in juveniles); 15, posterior, and 16, anterior edge of P3 (dP3 in juveniles); 17, posterior, and 18, anterior edge of C1 (dC1 in juveniles); 19, anterior edge of premaxilla at incisor alveolus; 20, ventral edge of external narial aperture; 21, apex of nasal; 22, dorsal, and 23, ventral edge of infraorbital foramen; 24, dorsal edge of maxilla-frontal suture; 25, dorsal edge of centre of frontal postorbital process; 26, dorsal edge of beginning of temporal fossa. Landmarks on the mandible are: 1, apex of mandibular cotyle; 2, posterior, and 3, anterior base of coronoid process; 4, apex of coronoid process; 5, posterior, and 6, anterior edge of retroarticular process; 7, anterior extent of mandibular (M. temporalis) adductor musculature; 8, posterior, and 10, anterior edge of M1 (in the included juveniles, M1 is un-erupted but the alveolar orifice and scar can easily be made out); 10, posterior, and 12, anterior edge of P4 (dP4 in juveniles); 14, posterior, and 15, anterior edge of C1 (dC1 in juveniles) at the alveolar border; 16, ventral edge of mandibular symphysis; and the depth of the horizontal mandibular ramus posterior to M1 (8, 9), at the M1/P4 junction (10, 11); and anterior to P4 (12, 13). Scale bar equals 5 cm.
To analyse net ontogenetic shape changes in juveniles to adults in Smilodon and extant pantherine felids multiple specimens within each category (juveniles and adults separately for each taxon) were averaged and ontogenetic shape change analyses from juveniles to adults were conducted in tpsSpline [75]. This approach is often used to study evolutionary shape changes in a selected ingroup of species relative to an outgroup [63], [69]. To facilitate additional comparisons of juvenile to adult shape changes other than Cartesian deformation grids and vector analysis of landmark displacements, the bending energy and the Procrustes Distance, d, were computed for each species-pair of juvenile and adult average shape configurations. The bending energy is related to the degree of localisation of landmarks, as noted above, and the Procrustes Distance is an often used metric in pair-wise comparisons of the amount of difference between biological shapes [62], [76].
Results
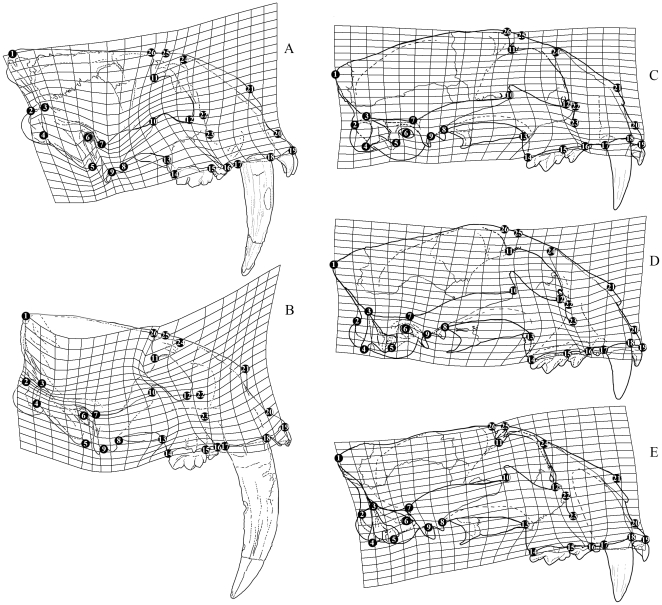
Juvenile cranial and mandibular shapes are substantially different from the shapes in adult specimens in each species (Fig. 1, 2, 3, 4) and they occupy different portions of shape space in Relative Warps analysis (Fig. S2,3). Net ontogenetic shape changes in the crania of Smilodon are much greater than in extant pantherines, as indicated by vector analysis and the nature of the warp grids of net shape changes (Fig. 6); this is corroborated by much larger Procrustes Distances between juveniles and adults in Smilodon compared to extant pantherines. Shape changes are also more localized in Smilodon as indicated by much greater bending energies than were computed for extant pantherines. Procrustes Distances of net shape changes between juveniles and adults are 0.1326 in S. fatalis and 0.1550 in S. populator compared to 0.0687 in Neofelis diardi; 0.0833 in Panthera onca; and 0.0704 in P. tigris. Bending energies are 2.0297 in S. fatalis and 3.3846 in S. populator compared to 1.4804 in Neofelis diardi; 0.9185 in Panthera onca; and 1.0249 in P. tigris.
Figure 6. Comparison of cartesian deformation grids illustrating ontogenetic net shape changes in crania of Smilodon spp. and extant pantherines.
A, Smilodon fatalis; B, S. populator; C, Neofelis diardi; D, Panthera onca; and D, P. tigris.
Most ontogenetic changes are similar in Smilodon fatalis and S. populator, but there qre also differences. Both Smilodon species show similar overall trends in cranial ontogenetic modularization in that the posterior part of the cranium appears to undergo different shape changes from the anterior part of the cranium (Fig. 6A,B). The facial part of the cranium (landmarks 18–21; 24–26) undergoes a posterodorsal displacement in both species, most notably in S. populator, thus causing the palatal region of the skull to become elevated relative to the basicranial region. The mandibular cotyle also becomes more ventrally displaced in the adults (ventral displacement of landmark 9; most strongly in S. populator). These two morphological differences from other felids have previously been regarded as key evolutionary adaptations for gaping at high angles to facilitate biting with hypertrophied upper canines [2], [9], [13], [14], and it is here demonstrated that they are ontogenetic changes. It is noteworthy that these shape changes are most strongly expressed in S. populator, which on average has larger C1 than S. fatalis.
In both Smilodon species, the already very large mastoid process of the juveniles grows even larger by anteroventral displacement of landmark 5 and posterodorsal displacement of landmark 3, also causing dorsal displacement of the occipital condyle (landmark 2). Consequently, the distance between the anterior tip of the mastoid process and the mandibular cotyle (landmark 9) becomes reduced. In both species, the zygomatic arches expand in anteroposterior length and become more massive in dorsoventral height by anterodorsal displacement of landmark 10 (base of jugal postorbital process) and concomitant anteroventral displacement of landmark 13. In S. fatalis, the ventral junction of the jugal-squamosal suture (landmark 8) becomes slightly posteriorly displaced, whereas this is not the case in S. populator. There are marked differences in dental proportions from juveniles to adults and replacing the small dP4 with the much larger permanent carnassial causes a large posterior displacement of landmark 14 and a large anterior displacement of landmark 15. The width of the C1 also increases relative to the dC1 by posterior displacement of landmark 17 and anterior displacement of landmark 18. Concomitantly, the P3 becomes reduced relative to the functional deciduous carnassial in juveniles.
The dorsal outline of the cranium is straighter and the posterior part of the sagittal crest is more elevated in S. populator compared to S. fatalis [1], and this is also an ontogenetic difference in that the sagittal-occipital junction (landmark 1) in S. populator becomes markedly dorsally displaced whereas it becomes posteriorly and slightly ventrally displaced in S. fatalis. In both species, the adults are more prognatheous than the juveniles, as indicated by anterior (S. fatalis) and anterodorsal (S. populator) displacement of landmark 19. The narial aperture becomes enlarged by dorsal displacement of landmark 21 relative to landmark 20.
In comparison, net ontogenetic cranial shape changes in extant pantherines are substantially less (Fig. 6C–E). In Neofelis diardi, the mastoid process grows larger (anterior displacement of landmark 5), and the paroccipital process becomes more posteriorly oriented (posterior displacement of landmark 4); this is different from Panthera spp., where the mastoid does not enlarge and the paroccipital faces ventrally, and are traits which Neofelis spp. shares with sabercats [13], [14]. Similar changes occur with the zygomatic arches and dentition in extant pantherines as with Smilodon, as noted above. Adult extant pantherines also become slightly more prognatheous relative to juveniles (anterior displacement of landmark 19), and although the size of the narial aperture does increase compared to the juveniles, it does so much less than in Smilodon. The size of the infraorbital foramen increases slightly in Panthera spp., whereas no such changes occur in Neofelis diardi or Smilodon.
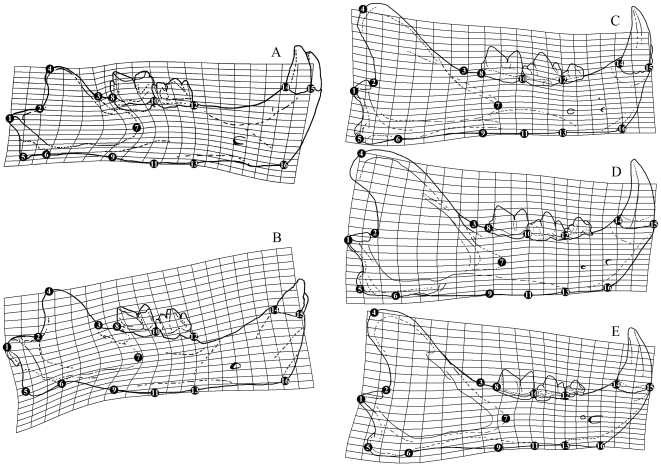
In contrast, net ontogenetic changes in mandibular shape are notably less in all species (Fig. 7), and the great differences in Procrustes Distance and bending energy between Smilodon and the extant pantherines observed in the cranium are not present in the mandible. Procrustes Distances between juveniles and adults are 0.0947 in S. fatalis and 0.1021 in S. populator compared to 0.0776 in Neofelis diardi; 0.1162 in Panthera onca; and 0.1289 in P. tigris. Bending energies are 0.4029 in S. fatalis and 0.1591 in S. populator compared to 0.0669 in Neofelis diardi; 0.0882 in Panthera onca; and 0.1234 in P. tigris. The low bending energies imply much less localized shape changes than are present in cranial ontogeny, as confirmed by the warp grids (Fig. 7) and landmark vector analysis.
Figure 7. Comparison of cartesian deformation grids illustrating ontogenetic net shape changes in mandibles of Smilodon spp. and extant pantherines.
A, Smilodon fatalis; B, S. populator; C, Neofelis diardi; D, Panthera onca; and D, P. tigris.
The mandible also shows modularization in that the posterior part (coronoid process, mandibular condyle, retroarticular process) appears to change independently of the mid-part of the horizontal ramus (dentition portion); and the symphyseal area also undergoes separate ontogenetic shape changes. In Smilodon, the symphysis expands dorsoventrally, but not by a large amount, and the mid-part of the horizontal ramus expands slightly dorsoventrally (Fig. 7A,B). Eruption of the permanent carnassial causes anterior displacement of landmark 10 and posterior displacement of landmark 8. The greatest ontogenetic difference is a localized shape change, anterior expansion of the coronoid fossa and attachment for the mandibular adductors, as indicated by a great anterior displacement of landmark 7. The low coronoid of juveniles remains low in adults, and even becomes slightly lower in S. fatalis by ventral displacement of landmark 4, whereas no change occurs in S. populator. The mandibular condyle becomes further posteriorly offset by a posterior displacement of landmark 1.
Unlike Smilodon, where cranial ontogenetic shape changes are much larger than mandibular shape changes, the mandible appears to ontogenetically change equally subtly in extant pantherines as does the cranium (Fig. 7C–E). As in Smilodon, the mandible is modularized, and the posterior part undergoes shape changes which appear detached from changes affecting the mid part of the horizontal ramus and the symphyseal region. The most marked shape changes are expansion of the coronoid process by a dorsal (Neofelis diardi) or dorsal and slightly anterior (Panthera spp.) displacement of landmark 1 and a slight deepening of the posterior coronoid fossa. In contrast to Smilodon, the attachment for the mandibular adductors does not increase (i.e., no anterior displacement of landmark 7); the mid-part of the horizontal ramus becomes slightly dorsoventrally constricted rather than expanded, and the symphyseal area does not undergo dorsoventral expansion. In this region is a localized shape change, an expansion of the alveolar width of the C1, as evidenced by slight posterior displacement of landmark 14 (all species) and slight anterior displacement of landmark 15 (Panthera only). The permanent carnassial causes anterior displacement of landmark 10 and posterior displacement of landmark 8, although less so than in Smilodon.
Discussion
The greatly different cranial morphology of adult Smilodon compared to extant felids was brought about by a combination of a juvenile cranial morphology that differed from those of extant pantherine juveniles, as well as postnatal ontogenetic shape changes which exceeded those observed in extant pantherines both in nature and magnitude. In contrast, the greatly different mandibular morphology of adult Smilodon compared to extant felids was also present in juveniles and ontogenetic changes were markedly less, and were comparable in magnitude to those observed in extant pantherines. Despite pantherines having a different adult cranial morphology than Smilodon and despite their undergoing less radical shape changes during postnatal ontogeny, many of the same areas in the cranium underwent similar postnatal ontogenetic shape changes. Mandibular shape changes were rather subtle and despite differences between Smilodon and pantherines, those differences were less than could be surmised from their greatly different adult morphologies.
This would imply that felids, in general, may undergo similar craniomandibular ontogenetic shape changes, and that differences in adult morphology are brought about by a combination of differences in juvenile morphology and the magnitude of ontogenetic shape changes rather than radically different shape changes affecting different craniomandibular areas. This is in agreement with the findings of Goswami [77] who demonstrated similar patterns of cranial landmark integrations among felids, including Smilodon. Some areas, however, underwent changes in Smilodon but not Panthera, such as mastoid size, and it is noteworthy that the Sunda clouded leopard, a taxon which has a number of craniomandibular and dental features in common with basal sabercats [13], [14], also underwent ontogenetic changes in this area, albeit less markedly. The above suggests that the ontogenetic pattern established for Smilodon could be characteristic of all derived sabercats and this may be tested by analyses of the few species where juveniles are known, notably the homotherines Amphimachairodus giganteus and Homotherium serum.
Smilodon and extant pantherines show modularization of postnatal ontogenetic shape changes along the same lines that have previously been documented for other mammals. Modularization of the alveolar (including dentition) portion of the mandible from the rest of the mandibular corpus appears to be an ancient and basic mammalian pattern [78], [79], and has been documented in, for instance, mice [80], [81], and a variety of primates, such as lorises [82], cercopithecines [83], great apes [84], and humans [85]. In general, the structures associated with the mandibular adductors (coronoid process; posterior part of horizontal ramus) appear to form a rather well integrated unit in a variety of mammals. As such, it appears reasonable to extend those inferences to felids as well, such as Smilodon and other sabercats, as well as extant felids, which is corroborated by the findings of this study. However, in primates, the ontogenetic mandibular integration of modules is not necessarily accompanied by similarities in cranial integration, and different parts of the cranium even in closely related species may have different patterns of cranial character integration, in particular in the region of the facial skeleton and degree of prognatism [85]–[90]. This indicates that among great apes (including humans), the mandible has undergone less evolutionary ontogenetic changes than the cranium [85]. This is also the case in Smilodon but not extant pantherines.
It has long been realized that the widespread notion in late 19th–early 20th century biological sciences that ontogeny recapitulates phylogeny is, at best, oversimplified and in many instances simply incorrect [91]–[94] in the sense presented by Ernst Haeckel (1834–1919) in his dually famous and infamous Biogenetic Law, first presented in full in his 1868 contribution, Natürliche Schöpfungsgeschichte [95]. However, it has been demonstrated that vertebrate embyos do bear a strong, if in some instances superficial resemblance to one another in early ontogeny, increasing their morphological differentiation through later ontogny, and post-natal juveniles in many groups of closely related mammals often bear a closer resemblance to each other structurally than do the adults [92], [93], [96]. In several mammalian groups, characters that appear early in phylogeny also often appear at an earlier stage during ontogeny [92], [93]; a famous example is the fossiliferously well documented equid evolution and the correlates with studies of postnatal ontogny [97]–[99].
Specialized, Plio-Pleistocene sabertoothed felids are substantially different from basal Miocene species in terms of craniomandibular and to a lesser extent dental morphology [2], [12]–[17], [19], [20], [100], [101]. The Smilodon juveniles do not in most respects bear much resemblance to adults of basal sabercats such as Machairodus, Promegantereon [102], Paramachairodus, Dinofelis, or Metailurus, and, as such, their ontogeny does not reflect phylogenetic shape changes among adult sabercats, but their overall, more elongate cranial shape is clearly plesiomorphic in that this was also the case among adults of basal sabercats but not adult Smilodon. In other respects, Smilodon juveniles are clearly morphologically derived, as the adults, for instance in having a very low coronoid process, a very large mastoid process, and large, blade-like upper canines and very short, incisiform lower canines. However, it is clear that in some respects, postnatal ontogeny in Smilodon does resemble, if not exactly recapitulate, phylogeny (see phylogeny in [8]).
The palatal part of the skull is far more upturned in derived sabercats than in basal sabercats, and this is an ontogenetic change in Smilodon accomplished by posterodorsal displacement of anterior landmarks 18–21 and 24–26 relative to the basicranial landmarks. The mandibular cotyle is far more ventrally displaced in Smilodon adults than in basal sabercats, and this also takes place during postnatal ontogeny by ventral displacement of landmark 9. Both of these morphological differences from extant felids have previously been regarded as key evolutionary adaptations for gaping at high angles to facilitate biting with hypertrophied upper canines [2], [9], [13], but it has hitherto been unknown if they constituted ontogenetic changes. Basal sabercats are less prognatheous than derived sabercats, most notably the homotherines (e.g., Homotherium, Xenosmilus), but also Smilodon, and landmark 19 becomes anteriorly (S. fatalis) or anterodorsally (S. populator) displaced during postnatal ontogeny in Smilodon. Enlargement of the narial aperture also occurs ontogenetically, and in basal sabercats the narial aperture is relatively smaller than in derived species, including Smilodon. The mandibular condyle (mandible landmark 1) is more posteriorly displaced in derived sabercats, and is another instance of Smilodon ontogeny recapitulating phylogeny; this trait is believed to be another adaptation for achieving high gape angles [9], [20].
Efficient biting and high bite forces are key adaptations for predation among carnivores, and unsurprisingly, in the puma, the most prevalent craniomandibular ontogenetic changes are those affecting portions of the cranium and mandible which are directly associated with the more demanding feeding ecology experienced when gradually substituting suckling for large vertebrate predation. Accordingly, during ontogeny puma skulls become taller with well developed occipital and sagittal crests, and relative postorbital width decreases, thus creating more space for the m. temporalis; zygomatic width increases; the muzzle become more sturdy; and the coronoid process increases [39], [40]. The findings of these authors for the puma are corroborated by the results for the pantherines of the current study, and, as such, the suggestion of Peigné & Bonis [103] that juveniles of leopard, serval, and caracal have more backwards-oriented coronoid processes than adults, but that coronoid process height does not change ontogenetically is not corroborated by the present study or by the findings of Giannini et al. [40]. This similarity of ontogenetic patterns among felids is to be expected, since felids are strictly carnivorous, and, accordingly, Goswami [77] found no correlation of cranial landmark integration in felids, including Smilodon, whereas this was present in Caniformia, Arctoidea and Musteloidea (including Mustelidae), which have a wider range of diets and, thus, presumably have undergone more differentiated evolutionary adaptations to accompany their dietary diversity.
Postnatal ontogenetic shape changes in Smilodon appear also to have increased biting efficiency and power, for instance in raising the palatal region to facilitate greater clearance between the upper and lower canines. The skull becomes taller and the zygomatic arches expand in anteroposterior direction (more space for the m. temporalis), but the arches also become dorsoventrally taller, thus creating a larger insertion area for the m. masseter, which would provide the arches with greater mechanical resistance to the action of the masseters. The attachment for the mandibular adductors on the mandibular horizontal ramus expands anteriorly; the coronoid process becomes anteroposteriorly expanded; the mandibular horizontal ramus becomes more sturdy, presumably so as to become more resistant to forces from biting [22]; the already large mastoid process in juveniles enlarges even further; and the occipital condyle is lifted, thus providing more leverage for the important head depressing action during predation [2], [10], [12], [20]. These ontogenetic shape changes would appear all to be tied to the mechanically more demanding actions of vertebrate predation than suckling, as they imply increased biting power. The upper canine also grows much longer but also more anteroposteriorly sturdy. However, in contrast to extant felids, the coronoid process does not get taller, presumably since a low coronoid process was an important adaptation for achieving very high gape angles [2], [9], [10], [12]–[16], [18], [20].
Morphological variation in the voluminous La Brea material of Smilodon fatalis adults is substantial [31], [47], [104], and this was part of the reasons for Berta [6] synonymizing the Pleistocene North and South American Smilodon into one species, S. populator. Kurtén & Werdelin [1] provided a comprehensive analysis showing that consistent differences were present, and that this indicated two species, S. fatalis from North and north-western (Pacific coast) South America, whereas S. populator occupied the remainder of the South American continent. To their species differences may be added several ontogenetic differences, such as greater palatal elevation and greater mandibular cotyle depression in S. populator; and posterior displacement of the ventral junction of the jugal-squamosal suture in S. fatalis. The straight dorsal cranial profile in S. populator was a character upon which Kurtén & Werdelin [1] placed emphasis as species-distinction from S. fatalis, and the current study expands on this finding, demonstrating that it is a postnatal ontogenetic shape difference between the two species. Accordingly, craniomandibular ontogeny also indicates differences between the eastern South American and the North American Pleistocene material of Smilodon, corroborative of two species.
Supporting Information
Juvenile specimen of Smilodon populator from Naturhistoriska riksmuseet in Stockholm.
(DOC)
Relative Warps analysis of juvenile and adult cranial shape in Smilodon spp. and extant pantherines.
(DOC)
Relative Warps analysis of juvenile and adult mandible shape in Smilodon spp. and extant pantherines.
(DOC)
Acknowledgments
I am indebted to the staffs at the Naturhistoriska riksmuseet in Stockholm; the Zoological Museum in Copenhagen; the Museum National d'Histoire Naturelle in Paris; the Natural History Museum in London; and the National Museum of Natural History (Naturalis) in Leiden. Dr. John Harris and Mr. Christopher Shaw kindly provided additional information and data of Smilodon fatalis from the voluminous Hancock collection at Los Angeles County Museum. I am grateful to academic editor Dr. Anjali Goswami and two anonymous referees for providing insightful criticism of an earlier draft of this manuscript.
Footnotes
Competing Interests: The author has declared that no competing interests exist.
Funding: Funding received from Aalborg University. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Kurtén B, Werdelin L. Relationships between North and South American Smilodon. J Vert Paleontol. 1990;10:158–169. [Google Scholar]
- 2.Turner A, Antón M. The big cats and their fossil relatives. An illustrated guide to their evolution and natural history. New York: Columbia Univ Press; 1997. [Google Scholar]
- 3.Rincón AD. A first record of the Pleistocene saber-toothed cat Smilodon populator Lund, 1842 (Carnivora: Felidae: Machairodontinae) from Venezuela. Ameghiniana. 2006;43:499–501. [Google Scholar]
- 4.Prieto A, Labarca R, Sierpe V. New evidence of the sabertooth cat Smilodon (Carnivora: Machairodontinae) in the late Pleistocene of southern Chilean Patagonia. Rev Chil Hist Nat. 2010;83:299–307. [Google Scholar]
- 5.Berta A. The status of Smilodon in North and South America. Contr Sci Nat Hist Mus Los Angeles County. 1985;379:1–15. [Google Scholar]
- 6.Berta A. The sabercat Smilodon gracilis from Florida and a discussion of its relationships (Mammalia, Felidae, Smilodontini). Bull Florida State Mus Biol Sci. 1987;31:1–63. [Google Scholar]
- 7.Lund PW. Blik paa Brasiliens Dyreverden för sidste Jordomvæltning. Fjerde Afhandling: Fortsættelse af Pattedyrene. Det Kongelige Danske Videnskabernes Selskabs Naturvidenskabelige og Mathematiske Afhandlinger, 4 (9de Deel) 1842:137–208. [Google Scholar]
- 8.Christiansen P. Phylogeny of the sabertoothed felids (Carnivora: Felidae: Machairodontinae). Cladistics. in press doi: 10.1111/cla.12008. [DOI] [PubMed] [Google Scholar]
- 9.Emerson SB, Radinsky LB. Functional analysis of sabertooth cranial morphology. Paleobiol. 1980;6:295–312. [Google Scholar]
- 10.Akersten WA. Canine function in Smilodon (Mammalia; Felidae; Machairodontinae). Contr Sci Nat Hist Mus Los Angeles County. 1985;356:1–22. [Google Scholar]
- 11.Biknevicius AR, Van Valkenburgh B, Walker J. Incisor size and shape: Implications for feeding behavior in saber-toothed “cats”. J Vert Palaeontol. 1996;16:510–521. [Google Scholar]
- 12.Antón M, Salesa MJ, Pastor JF, Sánchez IM, Fraile S, et al. Implications of the mastoid anatomy of larger extant felids for the evolution and predatory behaviour of sabretoothed cats (Mammalia, Carnivora, Felidae). Zool J Linn Soc. 2004;140:207–221. [Google Scholar]
- 13.Christiansen P. Sabertooth characters in the clouded leopard (Neofelis nebulosa Griffith, 1821). J Morphol. 2006;267:1186–1198. doi: 10.1002/jmor.10468. [DOI] [PubMed] [Google Scholar]
- 14.Christiansen P. Evolutionary convergence of primitive sabertooth craniomandibular morphology: the clouded leopard (Neofelis nebulosa) and Paramachairodus ogygia compared. J Mamm Evol. 2008;15:155–179. [Google Scholar]
- 15.Slater GJ, Van Valkenburgh B. Long in tooth: evolution of sabertooth cat cranial shape. Paleobiol. 2008;34:403–419. [Google Scholar]
- 16.Prevosti FJ, Turazzini GF, Chemisquy AM. Cranial morphology in sabertooth cats: Allometry, function and phylogeny. Ameghiniana. 2010;47:239–256. [Google Scholar]
- 17.Werdelin L, O'Brien SJ, Johnson WE, Yamaguchi N. Phylogeny and evolution of cats (Felidae). In: Macdonald DW, Loveridge AJ, editors. Biology and Conservation of Wild Felids. Oxford Univ Press; 2010. pp. 60–82. [Google Scholar]
- 18.Wroe S, McHenry C, Tomason JJ. Bite club: Comparative biteforce in big biting mammals and the prediction of predatory behaviour in fossil taxa. Proc Royal Soc Lond (Ser B) 2005;272:619–625. doi: 10.1098/rspb.2004.2986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Christiansen P. Comparative biteforces and canine bending strengths in feline and sabretooth felids: implications for predatory ecology. Zool J Linn Soc. 2007;151:423–437. [Google Scholar]
- 20.Christiansen P. A dynamic model for the evolution of sabrecat predatory bite mechanics. Zool J Linn Soc. 2011;162:220–242. [Google Scholar]
- 21.McHenry CH, Wroe S, Clausen PD, Moreno K, Cunningham E. Supermodeled sabercat, predatory behavior in Smilodon fatalis revealed by high-resolution 3D computer simulation. Proc Natl Acad Sci. 2007;104:16010–16015. doi: 10.1073/pnas.0706086104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Therrien F. Feeding behaviour and bite force of sabretoothed predators. Zool J Linn Soc. 2005;145:393–426. [Google Scholar]
- 23.Christiansen P, Wroe SM. Bite forces and evolutionary adaptations to feeding ecology in carnivores. Ecology. 2007;88:347–358. doi: 10.1890/0012-9658(2007)88[347:bfaeat]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 24.Volf J. Exigenes alimentaires et dentition des jeunes de trois especes de felides. Mammalia. 1972;36:683–686. [Google Scholar]
- 25.Ewer RE. The carnivores. New York: Cornell Univ Press; 1973. [Google Scholar]
- 26.Slaughter BH, Pine RH, Pine NE. Eruption of cheek teeth in Insectivora and Carnivora. J Mammal. 1974;55:115–125. [PubMed] [Google Scholar]
- 27.Smuts GL, Anderson JL, Austin J. Age determination of the African lion (Panthera leo). J Zool Lond. 1978;185:115–146. [Google Scholar]
- 28.Johnson NF, Brown BA, Bosomworth JC. Age and sex characteristics of bobcat canines and their use in population assessment. Wildl Soc Bull. 1981;9:202–206. [Google Scholar]
- 29.Jackson DL, Gluesing EA, Jacobson HA. Dental eruption in bobcats. J Wildl Managm. 1988;52:515–517. [Google Scholar]
- 30.Stander PE. Field age determination of leopards by tooth wear. Afr J Ecol. 1997;35:156–161. [Google Scholar]
- 31.Meachen-Samuels J, Binder WJ. Sexual dimorphism and ontogenetic growth in the American lion and sabertoothed cat from Ranho la Brea. J Zool Lond. 2010;280:271–279. [Google Scholar]
- 32.Crowe DM. Aspects of ageing, growth, and reproduction of bobcats from Wyoming. J Mammal. 1975;56:177–198. [PubMed] [Google Scholar]
- 33.Garcia-Perea R. Patterns of postnatal development in skulls of lynxes, genus Lynx (Mammalia: Carnivora). J Morphol. 1996;229:241–254. doi: 10.1002/(SICI)1097-4687(199609)229:3<241::AID-JMOR1>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 34.Petersen MK, Petersen MK. Growth rate and other postnatal developmental changes in margays. Carnivore. 1978;1:87–92. [Google Scholar]
- 35.Stuart CHT, Stuart TD. Age determination and development of foetal and juvenile Felis caracal Schreber, 1776. Säugetierk Mitt. 1985;32:217–229. [Google Scholar]
- 36.Stehlik J. Breeding jaguars Panthera onca at Ostrava Zoo. Intl Zoo Yearbk. 1971;11:116–118. [Google Scholar]
- 37.Mazák V. Der Tiger. 1983. Neue Brehm-Bücherei 356: A. Ziemsen Verlag.
- 38.Smuts GL, Robinson GA, Whyte IJ. Comparative growth of wild male and female lions (Panthera leo). J Zool Lond. 1980;190:365–373. [Google Scholar]
- 39.Segura V, Flores DA. Aproximación cualitativa y aspectos funcionales en la ontogenia craneana de Puma concolor (Felidae). Mastozool Neotropical. 2009;16:169–182. [Google Scholar]
- 40.Giannini NP, Segura V, Giannini MI, Flores D. A quantitative approach to the cranial ontogeny of the puma. Mamm Biol. 2009;75:547–554. [Google Scholar]
- 41.Gay SW, Best TL. Age-related variation in skulls of the puma (Puma concolor). J Mammal. 1996;77:191–198. [Google Scholar]
- 42.Laird AK. Postnatal growth of birds and mammals. Growth. 1966;30:349–263. [PubMed] [Google Scholar]
- 43.Laird AK, Tyler SA, Barton AD. Dynamics of normal growth. Growth. 1965;29:233–248. [PubMed] [Google Scholar]
- 44.Zullinger EM, Ricklefs RE, Redford KH, Mace GM. Fitting sigmoidal equations to mammalian growth curves. J Mammal. 1984;65:607–635. [Google Scholar]
- 45.Benoit MH. What's the difference? A multiphasic allometric analysis of fossil and living lions. In: Goswami A, Friscia A, editors. Carnivoran evolution. New Views on phylogeny, form, and function. Cambridge Univ Press; 2010. pp. 165–188. [Google Scholar]
- 46.Rawn-Schatzinger V. Development and eruption sequence of deciduous and permanent teeth in the saber-tooth cat Homotherium serum Cope. J Vert Paleontol. 1983;3:49–57. [Google Scholar]
- 47.Rawn-Schatzinger V. The scimitar cat Homotherium serum Cope. Illinois State Mus Rep Invest. 1992;47:1–79. [Google Scholar]
- 48.Merriam JC, Stock C. The Felidae of Rancho La Brea. Carnegie Inst Washington Publ. 1932;422:1–232. [Google Scholar]
- 49.Miller GJ. On the age distribution of Smilodon californicus Bovard from Rancho la Brea. Contr Sci Los Angeles County Mus. 1968;131:1–17. [Google Scholar]
- 50.Tejada-Flores AE, Shaw CA. Tooth replacements and skull growth in Smilodon from Rancho la Brea. J Vert Paleontol. 1984;4:114–121. [Google Scholar]
- 51.Christiansen P, Harris JM. The body size of Smilodon (Mammalia: Felidae). J Morphol. 2005;266:369–384. doi: 10.1002/jmor.10384. [DOI] [PubMed] [Google Scholar]
- 52.Rusconi C. Noticias acerca de un esqueleto juvenil perteneciente a los grandes carnívoros extinguidos Smilodon bonariensis. Anales de la Sociedad Científica Argentina. 1931;111:147–156. [Google Scholar]
- 53.Turner A. Dental sex dimorphism in European lions (Panthera leo L.) of the Upper Pleistocene: Palaeoecological and palaeoethological implications. Ann Zool Fennici. 1984;21:1–8. [Google Scholar]
- 54.Wiig Ø, Andersen T. Sexual size dimorphism in the skull of Norwegian Lynx. Acta Theriol. 1986;31:147–155. [Google Scholar]
- 55.Gittleman JL, Van Valkenburgh B. Sexual dimorphism in the canines and skulls of carnivores: Effects of size, phylogeny and behavioural ecology. J Zool Lond. 1997;242:97–117. [Google Scholar]
- 56.Sunquist M, Sunquist F. Wild cats of the World. Chicago: Univ Chicago Press; 2002. [Google Scholar]
- 57.Gust S, Harris JM, Shaw C. Discerning age and sex in the post-cranial skeleton of large cats. Paleobios. 2003;(Suppl 1):2–3. [Google Scholar]
- 58.Mazak JH. On the sexual dimorphism in the skull of the tiger (Panthera tigris). Mamm Biol. 2004;69:392–400. [Google Scholar]
- 59.Palmquist P, Torregrosa V, Pérez-Claros JA, Martínez-Navarro B, Turner A. A re-evaluation of the diversity of Megantereon (Mammalia, Carnivora, Machairodontinae) and the problem of species identification in extinct carnivores. J Vert Paleontol. 2007;27:160–175. [Google Scholar]
- 60.Pocock RI. On the tooth-change, cranial characters, and classification of the snow-leopard or ounce (Felis uncia). Ann Mag Nat Hist. 1916;18:306–316. [Google Scholar]
- 61.Bookstein FL. Principal warps: thin-plate splines and the decomposition of deformation. IEEE Trans Pattern Anal Mach Intell. 1989;11:567–585. [Google Scholar]
- 62.Bookstein FL. Morphometric Tools for Landmark Analysis: Geometry and Biology. New York: Cambridge Univ Press; 1991. [Google Scholar]
- 63.Zelditch ML, Swiderski DL, Sheets HD, Fink WL. Geometric Morphometrics for Biologists. A Primer. San Diego: Elsevier Acad Press; 2004. [Google Scholar]
- 64.Thompson D'Arcy W. On Growth and Form. Cambridge: Cambridge Univ Press; 1917. [Google Scholar]
- 65.Gower JC. Generalized Procrustes analysis. Psychometrika. 1975;40:33–51. [Google Scholar]
- 66.Rohlf FJ, Slice DE. Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst Biol. 1990;39:40–59. [Google Scholar]
- 67.Zelditch ML, Bookstein FL, Lundrigan BL. Ontogeny of integrated skull shape in the cotton rat, Sigmodon. Evolution. 1992;46:1164–1180. doi: 10.1111/j.1558-5646.1992.tb00626.x. [DOI] [PubMed] [Google Scholar]
- 68.Zelditch ML, Swiderski DL, Fink WL. Discovery of phylogenetic characters in morphometric data. In: Wiens JJ, editor. Phylogenetic analysis of morphological data. Smithsonian Inst. Press, Washington; 2000. pp. 37–83. [Google Scholar]
- 69.Swiderski DL. Morphological evolution of the scapula in tree squirrels, chipmunks, and ground squirrels (Sciuridae): An analysis using Thin-Plate Splines. Evolution. 1993;47:1854–1873. doi: 10.1111/j.1558-5646.1993.tb01274.x. [DOI] [PubMed] [Google Scholar]
- 70.Rohlf FJ. tpsDig, digitize landmarks and outlines, ver. 2.0. 2004a. Department of Ecology and Evolution, State University of New York at Stony Brook [computer program and documentation]
- 71.Rohlf FJ. tpsRelw, ver. 1.39. 2004b. Department of Ecology and Evolution, State University of New York at Stony Brook [computer program and documentation]
- 72.Rohlf FJ. Relative warp analysis and an example of its application to mosquito wings. In: Marcus LF, Bello E, Garcia-Valdecasas A, editors. Contributions to morphometrics, vol 8. Madrid: Museo Nacional de Ciencias Naturales (CSIC); 1993. pp. 131–159. [Google Scholar]
- 73.Bookstein FL. Combining the tools of geometric morphometrics. In: Marcus LF, Corti M, Loy A, Naylor GJP, Slice DE, editors. Advances in Morphometrics. Proceedings of the 1993 NATO Advanced Studies Institute on Morphometrics, IL Ciocco, Italy. New York: Plenum Publ. Corp; 1996. pp. 131–151. [Google Scholar]
- 74.Birch JM. Comparing wing shape of bats: The merits of principal components analysis and relative-warp analysis. J Mammal. 1997;78:1187–1198. [Google Scholar]
- 75.Rohlf FJ. tpsSpline, thin-plate-spline, version 1.20. 2004c. Department of Ecology and Evolution, State University of New York at Stony Brook [computer program and documentation]
- 76.Sneath PHA. Trend-surface analysis of transformation grids. J Zool Lond. 1967;151:65–122. [Google Scholar]
- 77.Goswami A. Morphological integration in the carnivoran skull. Evolution. 2006;60:169–183. [PubMed] [Google Scholar]
- 78.Atchley WR, Hall BK. A model for development and evolution of complex morphological structures. Biol Rev Cambridge Phil Soc. 1991;66:101–157. doi: 10.1111/j.1469-185x.1991.tb01138.x. [DOI] [PubMed] [Google Scholar]
- 79.Smith MM, Hall BK. Development and ontogenetic origins of vertebrate skeletogenic and odontogenic tissues. Biol Rev Cambridge Phil Soc. 1990;65:277–373. doi: 10.1111/j.1469-185x.1990.tb01427.x. [DOI] [PubMed] [Google Scholar]
- 80.Klingenberg CP, Leamy LJ, Cheverud JM. Integration and modularity of quantitative trait locus effects on geometric shape in the mouse mandible. Genetics. 2004;166:1909–1921. doi: 10.1534/genetics.166.4.1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Klingenberg CP, Mebus K, Auffray JC. Developmental integration in a complex morphological structure: now distinct are the modules in the mouse mandible? Evol Dev. 2003;5:522–531. doi: 10.1046/j.1525-142x.2003.03057.x. [DOI] [PubMed] [Google Scholar]
- 82.Ravosa MJ. Cranial allometry and geographic variation in slow lorises (Nycticebus). Am J Primatol. 1998;45:225–243. doi: 10.1002/(SICI)1098-2345(1998)45:3<225::AID-AJP1>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 83.Willmore KE, Roseman CC, Rogers J, Cheverud JM, Richtsmeier JT. Comparison of mandibular phenotypic and genetic integration between baboon and mouse. Evol Biol. 2009;36:19–36. doi: 10.1007/s11692-009-9056-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Daegling DJ. Growth in mandibles of African apes. J Human Evol. 1996;30:315–341. [Google Scholar]
- 85.Polanski JM. Morphological integration of the modern human mandible during ontogeny. Intl J Evol Biol. 2011;2011:1–11. doi: 10.4061/2011/545879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ackermann RR. Patterns of covariation in the hominoid craniofacial skeleton: implications for paleoanthropological models. J Human Evol. 2002;43:167–187. doi: 10.1006/jhev.2002.0569. [DOI] [PubMed] [Google Scholar]
- 87.Ackermann RR. Ontogenetic integration of the hominoid face. J Human Evol. 2005;48:175–197. doi: 10.1016/j.jhevol.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 88.Bastir M, Rosas A. Facial heights: Evolutionary relevance of postnatal ontogeny for facial orientation and skull morphology in humans and chimpanzees. J Human Evol. 2004;47:359–381. doi: 10.1016/j.jhevol.2004.08.009. [DOI] [PubMed] [Google Scholar]
- 89.Polanski JM, Franciscus RG. Patterns of craniofacial integration in extant Homo, Pan, and Gorilla. Am J Phys Anthropol. 2006;131:38–49. doi: 10.1002/ajpa.20421. [DOI] [PubMed] [Google Scholar]
- 90.Ravosa MJ, Daniel AN. Ontogeny and phyletic size change in living and fossil lemurs. Am J Primatol. 2010;72:161–172. doi: 10.1002/ajp.20767. [DOI] [PubMed] [Google Scholar]
- 91.Oppenheimer JM. Embryology and evolution: Nineteenth Century hopes and Twentieth Century realities. Quart Rev Biol. 1959;34:271–277. doi: 10.1086/402827. [DOI] [PubMed] [Google Scholar]
- 92.Gould SJ. Ontogeny and phylogeny. Massachusetts: Harvard Univ Press; 1977. [Google Scholar]
- 93.Mayr E. Recapitulation reinterpreted: The somatic program. Quart Rev Biol. 1994;69:223–232. [Google Scholar]
- 94.Rasmussen N. The decline of recapitulationism in early Twentieth-Century biology: Disciplinary conflict and consensus on the battleground of theory. J Hist Biol. 1991;24:51–89. [Google Scholar]
- 95.Haeckel E. Natürliche Schöpfungsgeschichte. Gemeinverständliche wissenschaftliche Vorträge über die Entwicklungslehre im Allgemeinen und diejenige von Darwin, Goethe und Lamarck im Besonderen, über die Anwendung derselben auf den Ursprung des Menschen und andere damit zusammenhängende Grundfragen der Naturwissenschaft. Berlin: G. Reimer; 1868. [Google Scholar]
- 96.Gilbert SF. Developmental biology. Sunderland: Sinauer Assoc. Inc; 2011. [Google Scholar]
- 97.Robb RC. A study of mutations in evolution. I. Evolution in the equine skull. J Genet. 1935a;31:39–46. [Google Scholar]
- 98.Robb RC. A study of mutations in evolution. II. Ontogeny in the equine skull. J Genet. 1935b;31:47–52. [Google Scholar]
- 99.Radinsky LB. Ontogeny and phylogeny in horse skull evolution. Evolution. 1984;38:1–15. doi: 10.1111/j.1558-5646.1984.tb00254.x. [DOI] [PubMed] [Google Scholar]
- 100.Salesa MJ, Antón M, Turner A, Morales J. Aspects of the functional morphology in the cranial and cervical skeleton of the sabre-toothed cat Paramachairodus ogygia (Kaup 1832) (Felidae, Machairodontinae) from the Late Miocene of Spain: Implications for the origins of the machairodont killing bite. Zool J Linn Soc. 2005;144:363–377. [Google Scholar]
- 101.Salesa MJ, Antón M, Turner A, Morales J. Inferred behaviour and ecology of the primitive sabre-toothed cat Paramachairodus ogygia (Felidae, Machairodontinae) from the Late Miocene of Spain. J Zool Lond. 2006;268:243–254. [Google Scholar]
- 102.Salesa MJ, Antón M, Turner A, Alcalá L, Montoy P, et al. Systematic revision of the Late Miocene sabre-toothed felid Paramachaerodus in Spain. Palaeontol. 2010;53:1369–1391. [Google Scholar]
- 103.Peigné S, de Bonis L. Juvenile cranial anatomy of Nimravidae (Mammalia, Carnivora): Biological and phylogenetic implications. Zool J Linn Soc. 2003;138:477–493. [Google Scholar]
- 104.Slaughter BH. Some observations concerning the genus Smilodon with special reference to Smilodon fatalis. Texas J Sci. 1963;55:68–81. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Juvenile specimen of Smilodon populator from Naturhistoriska riksmuseet in Stockholm.
(DOC)
Relative Warps analysis of juvenile and adult cranial shape in Smilodon spp. and extant pantherines.
(DOC)
Relative Warps analysis of juvenile and adult mandible shape in Smilodon spp. and extant pantherines.
(DOC)