Table 4.
Steady-state kinetic parameters and 15N and 13C KIE for the reaction catalyzed by Pnc1 with alternate substrates. Assays were performed as described under Experimental Procedures at pH 7.5.
| Substrate | Structure | kcat (s−1)a | Km (µM)a | kcat/Km (M−1s−1) | 15N KIE | 13C KIE |
|---|---|---|---|---|---|---|
| nicotinamide | 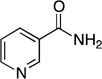 |
0.69 ± 0.04 | 9.6 ± 2.1 | (7.2 ± 1.2) × 104 | 1.0122 ± 0.0002 | 1.0125 ± 0.0004 |
| pyrazinamide | 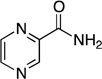 |
2.56 ± 0.10 | 157 ± 16 | (1.6 ± 0.1) × 104 | 1.0231 ± 0.0007 | 1.0196 ± 0.0023 |
| 5-methylnicotinamide | 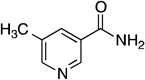 |
1.75 ± 0.05 | 61 ± 6 | (2.9 ± 0.2) × 104 | 1.0151 ± 0.0009 | 1.0269 ± 0.0037 |
| 1-methylnicotinamide | 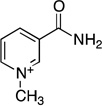 |
NAb | NAb | (8.6 ± 1.7) × 100 | NDd | NDd |
| benzamide | 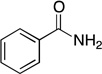 |
0.0088 ± 0.0004 | 25 ± 7 | (3.5 ± 0.9) × 102 | NDd | NDd |
| thionicotinamide | 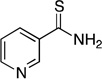 |
NAb | NAb | (5.0 ± 1.7) × 101 | 1.0262 ± 0.0019 | NDd |
| nicotinamide mononucleotide (NMN+) | < 10−5 c | |||||
| nicotinamide adenine dinucleotide (NAD+) | < 10−5 c |
kcat and Km values were determined from fitting the data to the Michaelis-Menten equation using Kaleidagraph (Synergy Software, Reading, PA). Errors represent the error of the fit to the data.
Saturation was not obtained, therefore only kcat/Km values are reported.
No activity was observed above the detection limit of the assay of 10−5 s−1.
Not determined.
