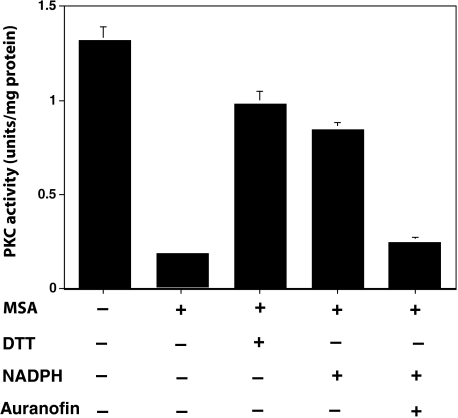
FIGURE 8.
Reversal of PKC redox modification by an endogenous reductase and NADPH in the cell homogenates. Cells grown to confluency were homogenized in 20 mm Tris-HCl, pH 7.4, 1 mm EDTA, 0.5 mm phenylmethylsulfonyl fluoride (2 ml of buffer per 100-mm Petri dish) and centrifuged at 10,000 × g. The soluble fraction was incubated with MSA (5 μm) for 10 min at 30 °C. Then MSA was removed by gel filtration. Aliquots of this desalted cell extract were treated with DTT (1 mm) or NADPH (1 mm) for 10 min at 30 °C, and PKC isoenzymes (combined) were isolated by using a small DEAE-cellulose column. PKC activity was determined using neurogranin polypeptide as a substrate. The values are expressed as mean ± S.E. of triplicate estimations.