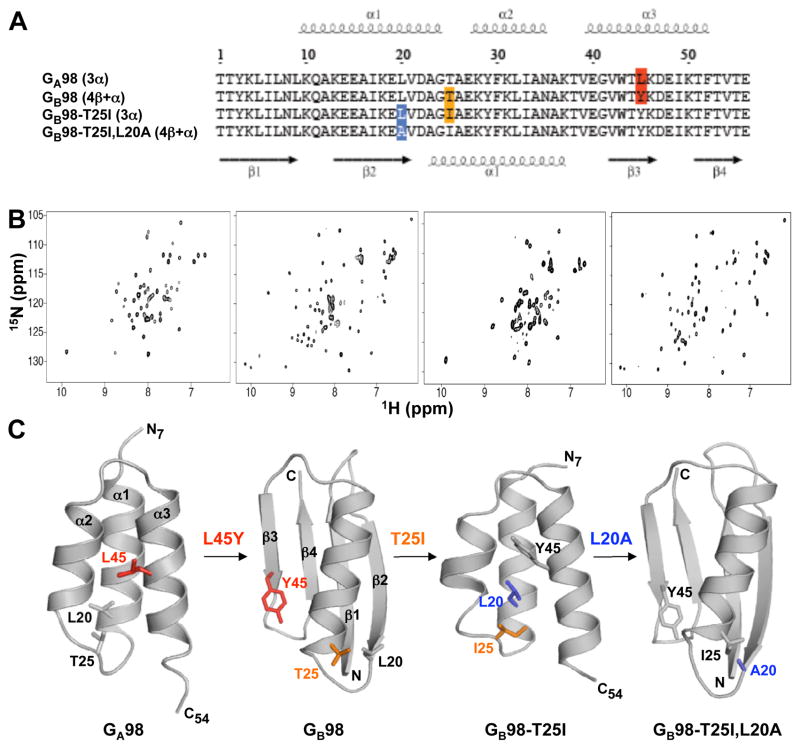
Figure 1.
Single amino acid mutations leading to fold switching. (A) Alignment of amino acid sequences for the four proteins in this study, highlighting the positions at which changes lead to switching between 3α and 4β+α folds. (B) 2D 15N-HSQC spectra for GA98 (left), GB98 (center left), GB98-T25I (center right), and GB98-T25I,L20A (right). Viewing NMR spectra from left to right, large differences are observed from one spectrum to the next as the three successive single site mutations, L45Y, T25I, and L20A, are made (see also Figure S1). (C) Representative structures from the NMR ensembles of GA98, GB98, GB98-T25I, and GB98-T25I,L20A. Residues mutated are highlighted.