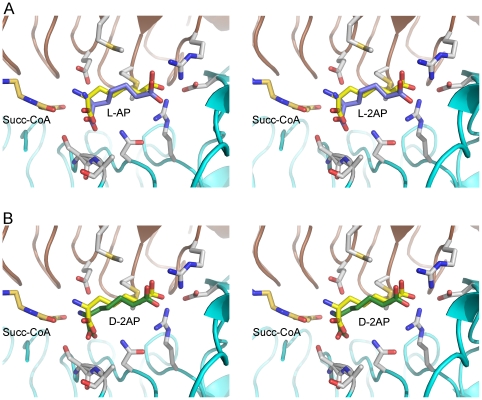
Figure 8. Structural basis of inhibition by D-2-aminopimelate.
A. Composite stereo view of the active site of PaDapD with bound succinamide-CoA, and the substrate L-2-AP. The position of the inactive substrate analogue succinamide-CoA (orange) and the L-2-aminopimelate (yellow) bound in the ternary complex were derived from a superposition with the DapD-succinamide-CoA-L-2AP complex (1KGQ). The bound L-2AP in the PaDapD-L-2AP complex is shown in blue. B. View of a composite model of the catalytically incompetent complex of PaDapD with bound succinamide-CoA, and the inhibitor D-2AP. The model was created using the same templates as in (A), the bound D-2AP in the structure of the complex of PaDaD with this ligand is shown in green.