Abstract
A T2-Relaxation-Under-Spin-Tagging (TRUST) technique was recently developed to estimate cerebral blood oxygenation, providing potentials for non-invasive assessment of the brain's oxygen consumption. A limitation of the current sequence is the need for long TR, as shorter TR causes an over-estimation in blood R2. The present study proposes a post-saturation TRUST by placing a non-selective 90° pulse after the signal acquisition to reset magnetization in the whole brain. This scheme was found to eliminate estimation bias at a slight cost of precision. To improve the precision, TE of the sequence was optimized and it was found that a modest TE shortening of 3.4ms can reduce the estimation error by 49%. We recommend the use of post-saturation TRUST sequence with a TR of 3000ms and a TE of 3.6ms, which allows the determination of global venous oxygenation with scan duration of 1 minute 12 seconds and an estimation precision of ±1% (in units of oxygen saturation percentage).
Keywords: arterial spin labeling, oxygenation, oxygen saturation, magnetization saturation, brain, non-invasive measurement
Introduction
Venous oxygenation (Yv) is defined as the fraction of oxygenated hemoglobin in the venous blood. Quantification of Yv in the brain allows the assessment of tissue oxygen homeostasis and, when combined with other physiologic measures such as cerebral blood flow, one can estimate cerebral metabolic rate of oxygen, CMRO2 (1–3). The interplay of these parameters forms the basis of functional MRI (fMRI), thus the ability to quantify Yv is important for understanding the mechanism of fMRI signal (4–9). Furthermore, alterations in Yv and CMRO2 are implicated in a number of conditions including aging (10), multiple sclerosis (11), carotid artery disease (12), and congenital heart disease (13), again underscoring the significance to develop a non-invasive, cost-effective, and reliable technique for the measurement of these parameters.
MRI has been used to obtain quantitative values of Yv based on the effects of deoxyhemoglobin on extravascular tissue (14,15) or intravascular blood (2,16,17). We have recently reported a T2-Relaxation-Under-Spin-Tagging (TRUST) MRI technique to estimate Yv by measuring blood R2 (=1/T2) (18). Compared to previous R2-based methods (19–22), TRUST MRI utilizes the spin-tagging principle to separate the blood signal from surrounding tissue, thereby minimizing partial volume effects and avoiding the need for judicious selection of voxels containing blood. Methodological studies have validated this technique in humans (23), and have also confirmed the ability of this technique in detecting oxygenation increases due to hypercapnia (24) and hyperoxia (25) as well as oxygenation decreases following hypoxia (25) and caffeine consumption (18).
At present, typical duration of TRUST MRI is 4 minutes with a precision of about ±2% in Yv estimation (in absolute units) (18). Our goal is to reduce the scan time to less than 2 minutes with a similar or even improved precision, thereby enhancing its feasibility in clinical applications. A limitation of the current TRUST protocol is that an excessively long TR (e.g. 7500ms) needs to be used, as a shorter TR results in an over-estimation of R2 due to magnetization disturbance from RF pulses in the previous TR (18). In this work, we modified the TRUST sequence by inserting a non-selective 90° RF pulse immediately following the EPI acquisition, termed post-saturation pulse, to reset the magnetization of all spins, and we showed that unbiased R2 estimation can now be achieved for all TR values tested (1500–7500ms at intervals of 750ms). Shorter TR was found to reduce signal intensity and the precision of R2 fitting. Therefore, to improve the precision of the estimation, we tested the TE-dependence of the TRUST results. Recommendations for TRUST imaging parameters are provided with considerations of tradeoff between scan duration, accuracy, and precision.
Materials and Methods
Pulse sequence and theory
The original TRUST sequence is shown in Fig. 1a and was described previously (18). Briefly, the sequence is similar to Arterial-Spin-Labeling (ASL) MRI, except that the labeling slab is above the imaging slice and that a series of non-slice-selective T2-preparation pulses are placed before the excitation pulse. The modification made in the present study is shown as red symbols in Fig. 1a, which consists of a non-slice-selective 90° pulse followed by dephasing gradients placed at the end of EPI echo train. This post-saturation pulse effectively resets the longitudinal magnetization of water protons in the brain to 0, thus the spin history is removed and the magnetization evolution is independent of what happened in the previous TR periods. Figure 1b shows simulated magnetizations for blood and tissue under the post-saturation sequence. Specifically, the blood signal in the control scan is given by:
| [1] |
where TR is the repetition time, effective TE (eTE) is the duration of T2-preparation, R1,b and R2,b are longitudinal and transverse relaxation rates of the blood, respectively. M0,b is the equilibrium magnetization of the blood. The signal in the labeled scan is written as:
| [2] |
in which TI is the inversion time. The difference signal can then be calculated as:
| [3] |
where S0 = M0,b.(1−e−(TR−TI)·R1,b)·2e−TI·R1,b and C = R1,b − R2,b. From Eq. [3], it is clear that the difference signal is a mono-exponential function of eTE and data fitting would yield the exponent, R1,b−R2,b. Note that R1,b is approximately 20 times smaller than R2,b (26,27), thus the exponent is primarily determined by blood R2. In addition, by assuming R1,b=0.62 s−1 (27), the influence of blood R1 in TRUST MRI is further reduced.
Fig. 1.
TRUST MRI technique. (a) Sequence diagram. The original sequence components are shown in black and the modification proposed in the present study is shown in red. TRUST MRI acquires label and control images at different effective TEs (eTE). The eTE is varied by changing the number of non-selective 180° pulses, which is needed for the R2-fitting. The limitation of the original TRUST sequence is that, at short TR, the non-selective 180° pulses can attenuate the magnetization of the whole brain, which reduces the signal in the next TR period. The extent of attenuation is dependent on eTE, thus would cause a systematic bias to the R2-fitting. The proposed modification places a non-selective 90° pulse after the acquisition echo train, thereby removing all spin history and resetting all magnetizations to zero. While the addition of this RF pulse decreases the signal intensity, it eliminates the eTE dependence and therefore the bias in R2. (b) Simulated magnetizations of blood and tissue under the post-saturation TRUST sequence using TR of 3000ms and eTE of 160ms. The magnetizations are shown for one TR period only. For clarity, only the components relevant for the image intensity are plotted. Specifically, transverse component is used during the effective TE period while longitudinal component is used in the other time. For convenience, the time between pre-sat and labeling pulses is neglected. Similarly, the time between the excitation and the post-saturation pulses is neglected.
It can also be seen that, while TR does not affect the exponential term of the fitting equation, shorter TR results in a lower S0 term (Fig. 2), thereby reducing signal-to-noise ratio (SNR) and estimation precision. The TE dependence study was therefore conducted to improve the precision.
Fig. 2.
TRUST signal intensity (control-label) as a function of TR in the sagittal sinus. Only data from eTE=0 are shown. Data from the original and post-saturation TRUST are shown in black and red symbols, respectively. For post-saturation TRUST, simulation results using Eq. [3] are also plotted (dashed line), demonstrating excellent correspondence with the experimental data. No simulation data are shown for the original TRUST, as the exact analytical expression of the signal is dependent on the magnetization history of individual spins and numerous assumptions are needed.
MRI Experiments
General
The experiments were performed on a 3 Tesla MRI system (Achieva, Philips Medical Systems, The Netherlands) using body coil for RF transmission and an eight-channel sensitivity encoding (SENSE) head coil for receiving. Foam padding was used to stabilize the head and minimize motion. The protocol was approved by University of Texas Southwestern Medical Center's Institutional Review Board and informed written consent was obtained from each participant. Two sub-studies were conducted on separate cohorts: a TR-dependence study and a TE-dependence study. In order to maintain a constant physiological state during the entire session (e.g. not falling asleep), the subjects were allowed to watch a movie.
TR-dependence study
Ten healthy volunteers (27±7 years old, 5 females, 5 males) participated in this study. Both the original TRUST sequence and TRUST with post-saturation were performed. For each sequence, nine TR values ranging from 1500ms to 7500ms with an interval of 750ms were tested. This study used a fixed TE of 7.0ms. The order of TR and sequences (original vs. post-saturation TRUST) was randomized for each subject to avoid time-dependent bias (e.g. due to fatigue or sleepiness). Other imaging parameters were: inversion-recovery time (TI) of 1200ms; T2-preparation times (effective TE, eTE) of 0ms, 40ms, 80ms and 160ms; matrix 64×64; voxel size 3.4×3.4×5mm3; single-shot gradient-echo EPI; single slice perpendicular to sagittal sinus, 3 averages, scan duration ranging from 0.6 (for TR=1500ms) to 3.0 (for TR=7500ms) minutes.
TE-dependence study
Eight healthy volunteers (27±3 years old, 5 females, 3 males) participated in this study. In the ASL literature, it is well known that the shortest possible TE should be used. The same principle applies to TRUST MRI. However, due to the length of EPI echo train, TE cannot practically approach 0. The previous TRUST protocol used parallel imaging to reduce TE to 7.0ms, which is already shorter than that used in most ASL studies (28–30). TE can be further reduced by using a higher SENSE factor and/or a smaller half Fourier factor, which may, however, result in poor SNR as fewer data in the k-space are acquired. Therefore, we tested TE values of 7.0, 4.9 and 3.6ms to assess whether these small variations in TE could yield noticeable differences in the TRUST data. The TE values used here correspond to k-line number of 31 (SENSE factor 2, half Fourier factor 0.9), 20 (SENSE factor 3, half Fourier factor 0.9), 17 (SENSE factor 3, half Fourier factor 0.7), respectively. The TR of this study was fixed at 8000ms and the original TRUST sequence was used. Other imaging parameters were similar to those used in the TR-dependence study.
Pilot study using the optimized TRUST protocol
Based on the TR and TE studies, recommendations were made in terms of optimal TR, TE, and pulse sequences. The optimized protocol was then applied on seven new volunteers (26±4 years old, 3 females, 4 males) to verify its performance. The imaging parameters were identical to those used in the above studies except that the choices of TR, TE, and pulse sequence (e.g. original vs. post-saturation TRUST) were based on outcomes of the TR and TE studies. The duration of the TRUST scan was 1 minute and 12 seconds.
Data analysis
TRUST MRI data were processed using in-house MATLAB (Mathworks, Natick, MA) scripts. The processing method was similar to that described previously (18). Briefly, after motion correction and image subtraction, a preliminary region-of-interest (ROI) was manually drawn to include the sagittal sinus. This ROI tends to have about 30–50 voxels that include the vein as well as some surrounding tissue. To further define the venous voxels, the voxels with the highest blood signals (according to the difference signals) in the ROI were chosen as the final mask for spatial averaging. For the purpose of standardizing protocol, we used 4 voxels in our studies, although we have tested the effect of voxel number and found that the results are relatively insensitive to the number (18). The difference signal was fitted to an exponential function of eTE, from which R2,b was obtained as described in Eq. [3].
Quality of the raw data (i.e. MR images at different TR and TE values) and reliability of the fitted parameters (i.e. estimated R2 values) were quantified using different indices. For the raw data, an SNR was calculated on a voxel-by-voxel basis, which was defined as the mean of the multiple measurements divided by the standard deviation across the measurements. Thus the SNR dependence on TR, TE, and pulse sequences can be examined. Reliability of the fitted parameters was assessed using two alternative indices, accuracy and precision of the estimation. The accuracy refers to the amount of systematic bias, and was evaluated by comparing the estimated R2 to that of the longest TR or shortest TE, which was assumed to reflect the true value. The precision refers to the uncertainty of the estimation, and was quantified by the standard error of R2 (εR2) from the goodness-of-fit assessment of the data fitting.
Yv was estimated from the blood R2 using a calibration curve (23). Error propagation from R2 to Yv was conducted by converting R2±εR2 values to corresponding Yv±εYv. All Yv and εYv values are written in units of oxygen saturation percentage (%).
For statistical analysis, a mixed effect model was used to assess whether there is a dependence of R2 or εR2 on TR or TE. The software R (Wirtschaftsuniversität Wien Vienna University, Austria) was used for this analysis. A P value of less than 0.05 after correcting for multiple comparisons is considered statistically significant.
Results
TR-dependence study
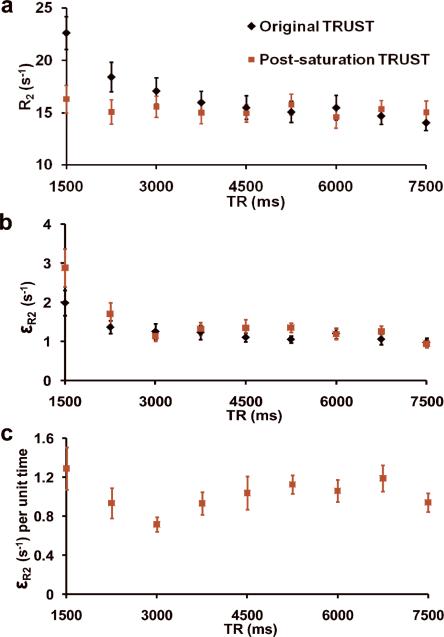
Figure 2 shows the blood signal (i.e. the difference between control and label images) as a function of TR using the original TRUST (black symbols) and post-saturation TRUST (red symbols) sequences. The signal intensity decreased at shorter TR, which is consistent with the simulation results using Eq. [3] (dashed line in Fig. 2). Figure 3a shows the estimated R2 as a function of TR. For original TRUST, shorter TR resulted in an over-estimation in R2 (P<0.001 with mixed-effect model), confirming our previous report (18). No such dependence was observed for the post-saturation TRUST (P=0.67), supporting our hypothesis that the addition of the non-selective pulse can remove the bias and improve the estimation accuracy. Figure 3b plots the standard error of R2, εR2, as a function of TR. Shorter TR resulted in poorer estimation precision for both the original TRUST (P=0.002) and the post-saturation TRUST (P<0.001). This can be explained by the lower signals at shorter TR as shown in Fig. 2. We have also examined εR2 per unit time by accounting for the larger number of repetitions one can obtain with a shorter TR. It was found that the error was lowest at a TR of 3000ms (Fig. 3c).
Fig. 3.
Effect of TR on TRUST MRI results (N=10). (a) Estimated R2 (=1/T2) as a function of TR. Data from the original and post-saturation TRUST are shown in black and red symbols, respectively. The R2 from the original TRUST shows a TR-dependence (P<0.001), while it is not the case for the post-saturation TRUST (P=0.67). (b) Standard error of R2 (εR2) from the goodness-of-fit assessment of the data fitting at different TR. This index represents the uncertainty in the estimation and is used as an indicator for precision. As expected, a shorter TR resulted in poorer precision in both sequences. (c) εR2 per unit time, which was computed by multiplying the corresponding values in (b) by , where 7500 (ms) is the longest TR used in the TR dependence study.
TE-dependence study
In the tissue regions, shorter TE corresponded to a lower SNR (P<0.001 between TE of 3.6ms vs. 7.0ms), which can be attributed to the reduced k-space sampling associated with parallel imaging and partial Fourier acquisition. In the sagittal sinus, on the other hand, shorter TE improved the signal stability. The SNR of the blood signal was 63±7 (mean±SE, N=8), 50±10, and 36±6 for TE of 3.6ms, 4.9ms, and 7.0ms, respectively (P=0.008 with mix-effect model). This SNR increase is considerable given the relatively small change in TE, and is because the outflow effect of the blood is drastically reduced at shorter TE. Figure 4a shows εR2 as a function of TE, demonstrating that the precision of the blood R2 estimation was improved at shorter TE (P<0.001). The estimation error at TE=3.6ms was only 51% of that at TE=7.0ms. A weak dependence of R2 on TE was also observed (Fig. 4b, P=0.034). Data using longer TE were found to slightly over-estimate R2. A more careful investigation suggested that the overestimation (calculated as R2,TE=7.0−R2,TE=3.6) was subject-dependent (Fig. 4c, P<0.001), and an individual with lower SNR tends to have a more pronounced over-estimation. For data with SNR>50, the over-estimation is negligible. Therefore, higher SNR afforded by shorter TE can improve both accuracy and precision of the TRUST data.
Fig. 4.
Effect of TE on TRUST MRI results (N=8). (a) εR2 at different TE values. Shorter TE corresponds to considerably smaller estimation error. (b) Estimated R2 at different TE values. R2 is weakly dependent on TE (P=0.034). R2 from TE of 7.0ms appears to contain some overestimation compared to TE of 3.6ms. (c) The amount of R2 over-estimation is correlated with SNR (P<0.001). An individual with lower SNR tends to have more over-estimation. For data with SNR>50, the over-estimation is negligible.
Pilot study using the optimized TRUST protocol
Based on the TR and TE studies, the optimal protocol was determined to be TR=3000ms and TE=3.6ms using the post-saturation sequence. Data acquired using this protocol revealed R2=15.4±0.7s−1 and εR2=0.39±0.04s−1 (N=7, mean ± standard deviation). Using an in vitro calibration curve, these values correspond to Yv=63.5±1.6% and εYv=0.98±0.11%.
Discussion
The main goal of the present study is to conduct further technical development for TRUST MRI so that it can be performed with a shorter duration without degradation of estimation reliability. The current TRUST protocol takes approximately 4 minutes and a major reason for the relatively long duration is the long TR. A shorter TR can reduce the scan duration, but our previous study has shown that it also causes a systematic bias in the estimated R2 value (18). The post-saturation TRUST proposed in the present study effectively eliminated the bias, allowing the sequence to take advantage of a shorter TR and scan duration. An unwanted effect of shorter TR is the lower estimation precision. Therefore, TE of the sequence was also optimized and it was found that a modest reduction of TE can considerably improve the precision of blood R2. Combining these findings, we recommend the use of post-saturation TRUST sequence with a TR of 3000ms and a TE of 3.6ms for future studies. Compared to the previous TRUST protocol, the choice of this TR value is expected to shorten the scan duration by 60%, while the use of post-saturation can remove the systematic bias which is estimated to be about 3% (in oxygen saturation percentage) at a TR of 3000ms.
A number of techniques have been developed to quantitatively estimate blood oxygenation in the brain. One approach exploits the susceptibility effect of deoxyhemoglobin on extravascular tissue (15,31), and uses simultaneous acquisitions of gradient and spin echoes to obtain Yv (32). An advantage of this technique is that it is capable of providing a spatial map of Yv. A second approach measures the phase angle of intravascular blood signal, and estimates Yv based on the susceptibility effect in venous blood (33–35). The advantage of this susceptometry technique is that it provides greater temporal resolution as the data acquisition can be completed within 30 seconds (2). A third method is based on T2 value of blood signal, which can be converted to Yv using a calibration plot (18–22). Advantages of the T2-based method are that only mono-exponential fitting is needed and that it is less sensitive to vessel orientation. A further advance brought about by TRUST is that partial voluming between vessel and tissue can be accounted for and the estimation is no longer susceptible to subjective ROI selection, rendering the technique a high test-retest reproducibility (18) and sensitivity to physiologic alterations (24,25). At present, the scan duration is somewhat long for certain patient populations. With the improvement introduced in this study, we can complete the TRUST scan within 1 minute and 12 seconds, and this protocol is now routinely used in the authors' application studies. It is also important to point out that, with this protocol, the estimation error on R2 is about 0.4s−1, corresponding to 1% error in venous oxygenation (in units of oxygen saturation percentage).
Using the original TRUST sequence, shorter TR showed an over-estimation of blood R2. Conceptually, this is because the RF pulses (specifically the T2-preparation pulses) in the previous TR period attenuated the magnetization in the next TR. Furthermore, the attenuation effect is greater when more T2-preparation pulses are used. That is, signal at longer eTE is attenuated more than that at shorter eTE. Thus, the T2-dependent signal decay appears faster than it should be. An alternative way to illustrate this effect is by signal equations. Assuming that the spins measured during the present TR had experienced the T2-preparation pulses in the previous TR, one can write the difference signal as:
In contrast to Eq. [3], the term in the bracket in Eq. [4] is now dependent on eTE. Thus, when using a mono-exponential function to fit these data, the estimated exponent will be greater than its true value. It should also be noted that the exact signal in the original TRUST is even more complicated than Eq. [4] as the magnetization could also be affected by labeling and excitation RF pulses as well as RF pulses in the earlier TR periods, which was not accounted for in the above equation.
Post-saturation pulses similar to that used in the present study have been employed previously in ASL MRI (36) and Vascular-Space-Occupancy (VASO) MRI (37). In those studies, the purpose of the post-saturation was to reset magnetization of the arterial blood instead of the venous blood. For example, Pell and colleagues devised a modified Flow-sensitive alternating inversion recovery (FAIR) approach in which a post-saturation pulse was inserted after the acquisition echo train. The authors showed that the modified FAIR sequence can reduce the time for the spins to reach a steady state and simplifies the flow quantification (36). However, the authors also pointed out that the method works most effectively when the RF transmission coil covers the entire subject (e.g. including the heart), in which all spins being imaged have previously experienced the saturation pulse (thus their magnetization has been reset). Unfortunately, in human MRI systems, the body coil does not really provide a full-body coverage when used for brain imaging. Consequently, the performance of the post-saturation pulse is dependent on other parameters such as arterial transit time, inversion time, and delay time, as shown by both Pell et al. (36) and Lu (37). The application of post-saturation pulse in TRUST MRI provides a unique advantage in that the venous spins being imaged originate from smaller veins or parenchyma tissues and, since these spins were inside the body coil coverage, their magnetization has been reset before entering the imaging slice. That is, the post-saturation pulse works more effectively for the venous side compared to the arterial side.
The finding that shorter TE resulted in more precise estimation is not as trivial as it seems at first sight. While shorter TE would correspond to higher signal based on theory, it is important to note that a shorter TE is achieved at the expense of k-space coverage (via higher SENSE factor and/or lower partial Fourier factor). Fewer data samples in k-space are associated with higher noise levels, thus shorter TE does not always result in higher SNR. As a matter of fact, our data showed that a TE of 3.6ms yielded lowest SNR in the tissue regions. This is consistent with the common practice in the ASL literature that few studies used a TE of less than 5ms (29,30,38,39). For the blood signal in the TRUST data, however, shorter TE resulted in a drastic increase in SNR and estimation precision (Fig. 4a). This improvement was attributed to both a signal increase (by 31%) and a noise reduction (by 34%) at shorter TE. Numerical simulations confirmed that such a large increase in signal intensity is expected for spins flowing at a velocity characteristic of the sagittal sinus (24), in which the excited spins may have flown out of the original voxel by the time the signal is acquired. The extent of the noise reduction was also consistent with simulations using pulsatile flow (22–60 cm/s within one cardiac cycle, mean velocity 35 cm/s). Therefore, we conjecture that the noise reduction at shorter TE is primarily attributed to a reduction in pulsation-related fluctuation. An alternative strategy to the use of a short TE is to apply flow-compensating gradients. Although the use of additional gradients will increase the TE value, flow-compensation are expected to improve spin coherence within a voxel. The benefit of such schemes shall be assessed in future studies.
The findings from the present study should be interpreted in view of a few limitations. First, the TR and TE dependence studies were conducted in separate cohorts, making the results not directly comparable. This design was chosen because of the relatively long duration of each study. The TR dependence study included 18 TRUST scans with varying TR and took about 1 hour. The TE dependence study took about half an hour. We were concerned that excessive scan time might result in greater motion and physiologic fluctuations. Thus we separated the TR and TE studies. During the scan, subjects were allowed to watch a movie in order to prevent them from falling asleep. Second, in explaining the TR-dependent over-estimation in the original TRUST sequence, we have primarily focused on the spin history and magnetization evolution. The lower SNR in the shorter TR scans could have also contributed to the higher R2, as demonstrated in the TE-dependence study. However, we note that the effect of SNR on R2 is on the order of 1 s-1, which is much less than that observed in the experimental data (Fig. 3a). Thus, the spin history effect is the predominant factor in the TR-dependence data. Finally, the TRUST technique has only been used to measure Yv in large vessels. In order to estimate Yv in smaller vessels, more advanced tagging schemes such as velocity selective labeling are needed (35).
Conclusion
We improved the speed and reliability of cerebral oxygenation measurement using a modified implementation of TRUST MRI. We recommend the use of post-saturation TRUST sequence with a TR of 3000ms and a TE of 3.6ms, which allows the determination of global venous oxygenation with scan duration of 1 minute 12 seconds and an estimation precision of ±1%.
Acknowledgments
Grant sponsors: NIH R01 MH084021, NIH R01 NS067015, NIH R01 AG033106
References
- 1.Xu F, Ge Y, Lu H. Noninvasive quantification of whole-brain cerebral metabolic rate of oxygen (CMRO2) by MRI. Magn Reson Med. 2009;62:141–148. doi: 10.1002/mrm.21994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jain V, Langham MC, Wehrli FW. MRI estimation of global brain oxygen consumption rate. J Cereb Blood Flow Metab. 2010;30:1598–1607. doi: 10.1038/jcbfm.2010.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kety SS, Schmidt CF. The Effects of Altered Arterial Tensions of Carbon Dioxide and Oxygen on Cerebral Blood Flow and Cerebral Oxygen Consumption of Normal Young Men. J Clin Invest. 1948;27:484–492. doi: 10.1172/JCI101995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Silvennoinen MJ, Clingman CS, Golay X, Kauppinen RA, van Zijl PC. Comparison of the dependence of blood R2 and R2* on oxygen saturation at 1.5 and 4.7 Tesla. Magn Reson Med. 2003;49:47–60. doi: 10.1002/mrm.10355. [DOI] [PubMed] [Google Scholar]
- 5.Lu H, Zhao C, Ge Y, Lewis-Amezcua K. Baseline blood oxygenation modulates response amplitude: Physiologic basis for intersubject variations in functional MRI signals. Magn Reson Med. 2008;60:364–372. doi: 10.1002/mrm.21686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.van Zijl PC, Eleff SM, Ulatowski JA, Oja JM, Ulug AM, Traystman RJ, Kauppinen RA. Quantitative assessment of blood flow, blood volume and blood oxygenation effects in functional magnetic resonance imaging. Nat Med. 1998;4:159–167. doi: 10.1038/nm0298-159. [DOI] [PubMed] [Google Scholar]
- 7.Buxton RB, Wong EC, Frank LR. Dynamics of blood flow and oxygenation changes during brain activation: the balloon model. Magn Reson Med. 1998;39:855–864. doi: 10.1002/mrm.1910390602. [DOI] [PubMed] [Google Scholar]
- 8.Hoge RD, Atkinson J, Gill B, Crelier GR, Marrett S, Pike GB. Linear coupling between cerebral blood flow and oxygen consumption in activated human cortex. Proc Natl Acad Sci U S A. 1999;96:9403–9408. doi: 10.1073/pnas.96.16.9403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Davis TL, Kwong KK, Weisskoff RM, Rosen BR. Calibrated functional MRI: mapping the dynamics of oxidative metabolism. Proc Natl Acad Sci U S A. 1998;95:1834–1839. doi: 10.1073/pnas.95.4.1834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lu H, Xu F, Rodrigue KM, Kennedy KM, Cheng Y, Flicker B, Hebrank AC, Uh J, Park DC. Alterations in Cerebral Metabolic Rate and Blood Supply across the Adult Lifespan. Cereb Cortex. 2011;21:1426–1434. doi: 10.1093/cercor/bhq224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sun X, Tanaka M, Kondo S, Okamoto K, Hirai S. Clinical significance of reduced cerebral metabolism in multiple sclerosis: a combined PET and MRI study. Ann Nucl Med. 1998;12:89–94. doi: 10.1007/BF03164835. [DOI] [PubMed] [Google Scholar]
- 12.Tsuchida C, Kimura H, Sadato N, Tsuchida T, Tokuriki Y, Yonekura Y. Evaluation of brain metabolism in steno-occlusive carotid artery disease by proton MR spectroscopy: a correlative study with oxygen metabolism by PET. J Nucl Med. 2000;41:1357–1362. [PubMed] [Google Scholar]
- 13.Durduran T, Zhou C, Buckley EM, Kim MN, Yu G, Choe R, Gaynor JW, Spray TL, Durning SM, Mason SE, Montenegro LM, Nicolson SC, Zimmerman RA, Putt ME, Wang J, Greenberg JH, Detre JA, Yodh AG, Licht DJ. Optical measurement of cerebral hemodynamics and oxygen metabolism in neonates with congenital heart defects. J Biomed Opt. 2010;15:037004. doi: 10.1117/1.3425884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.An H, Lin W, Celik A, Lee YZ. Quantitative measurements of cerebral metabolic rate of oxygen utilization using MRI: a volunteer study. NMR Biomed. 2001;14:441–447. doi: 10.1002/nbm.717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.He X, Yablonskiy DA. Quantitative BOLD: mapping of human cerebral deoxygenated blood volume and oxygen extraction fraction: default state. Magn Reson Med. 2007;57:115–126. doi: 10.1002/mrm.21108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wright GA, Hu BS, Macovski A. 1991 I.I. Rabi Award. Estimating oxygen saturation of blood in vivo with MR imaging at 1.5 T. J Magn Reson Imaging. 1991;1:275–283. doi: 10.1002/jmri.1880010303. [DOI] [PubMed] [Google Scholar]
- 17.Haacke EM, Lai S, Reichenbach JR, Kuppusamy K, Hoogenraad FG, Takeichi H, Lin W. In vivo measurement of blood oxygen saturation using magnetic resonance imaging: a direct validation of the blood oxygen level-dependent concept in functional brain imaging. Hum Brain Mapp. 1997;5:341–346. doi: 10.1002/(SICI)1097-0193(1997)5:5<341::AID-HBM2>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 18.Lu H, Ge Y. Quantitative evaluation of oxygenation in venous vessels using T2-Relaxation-Under-Spin-Tagging MRI. Magn Reson Med. 2008;60:357–363. doi: 10.1002/mrm.21627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wright GA, Hu BS, Macovski A. Estimating oxygen saturation of blood in vivo with MR imaging at 1.5 T. J Magn Reson Imaging. 1991;1:275–283. doi: 10.1002/jmri.1880010303. [DOI] [PubMed] [Google Scholar]
- 20.Golay X, Silvennoinen MJ, Zhou J, Clingman CS, Kauppinen RA, Pekar JJ, van Zijl PC. Measurement of tissue oxygen extraction ratios from venous blood T(2): increased precision and validation of principle. Magn Reson Med. 2001;46:282–291. doi: 10.1002/mrm.1189. [DOI] [PubMed] [Google Scholar]
- 21.Oja JM, Gillen JS, Kauppinen RA, Kraut M, van Zijl PC. Determination of oxygen extraction ratios by magnetic resonance imaging. J Cereb Blood Flow Metab. 1999;19:1289–1295. doi: 10.1097/00004647-199912000-00001. [DOI] [PubMed] [Google Scholar]
- 22.Qin Q, Grgac K, van Zijl PC. Determination of whole-brain oxygen extraction fractions by fast measurement of blood T(2) in the jugular vein. Magn Reson Med. 2011;65:471–479. doi: 10.1002/mrm.22556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lu H, Xu F, Grgac K, Liu P, Qin Q, van Zij PC. Calibration and validation of TRUST MRI for the estimation of cerebral blood oxygenation. Magn Reson Med. 2011 doi: 10.1002/mrm.22970. doi: 10.1002/mrm.22970, In-press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xu F, Uh J, Brier MR, Hart J, Jr., Yezhuvath US, Gu H, Yang Y, Lu H. The influence of carbon dioxide on brain activity and metabolism in conscious humans. J Cereb Blood Flow Metab. 2011;31:58–67. doi: 10.1038/jcbfm.2010.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xu F, Yezhuvath US, Liu P, Lu H. Proc Intl Soc Mag Reson Med. Stockholm, Sweden: 2010. Hypoxia and hyperoxia alter brain metabolism in awake human; p. 1032. [Google Scholar]
- 26.Zhao JM, Clingman CS, Narvainen MJ, Kauppinen RA, van Zijl PC. Oxygenation and hematocrit dependence of transverse relaxation rates of blood at 3T. Magn Reson Med. 2007;58:592–597. doi: 10.1002/mrm.21342. [DOI] [PubMed] [Google Scholar]
- 27.Lu H, Clingman C, Golay X, van Zijl PC. Determining the longitudinal relaxation time (T1) of blood at 3.0 Tesla. Magn Reson Med. 2004;52:679–682. doi: 10.1002/mrm.20178. [DOI] [PubMed] [Google Scholar]
- 28.Chalela JA, Alsop DC, Gonzalez-Atavales JB, Maldjian JA, Kasner SE, Detre JA. Magnetic resonance perfusion imaging in acute ischemic stroke using continuous arterial spin labeling. Stroke. 2000;31:680–687. doi: 10.1161/01.str.31.3.680. [DOI] [PubMed] [Google Scholar]
- 29.Sutton BP, Ouyang C, Ching BL, Ciobanu L. Functional imaging with FENSI: flow-enhanced signal intensity. Magn Reson Med. 2007;58:396–401. doi: 10.1002/mrm.21325. [DOI] [PubMed] [Google Scholar]
- 30.van Osch MJ, Teeuwisse WM, van Walderveen MA, Hendrikse J, Kies DA, van Buchem MA. Can arterial spin labeling detect white matter perfusion signal? Magn Reson Med. 2009;62:165–173. doi: 10.1002/mrm.22002. [DOI] [PubMed] [Google Scholar]
- 31.An H, Lin W. Impact of intravascular signal on quantitative measures of cerebral oxygen extraction and blood volume under normo- and hypercapnic conditions using an asymmetric spin echo approach. Magn Reson Med. 2003;50:708–716. doi: 10.1002/mrm.10576. [DOI] [PubMed] [Google Scholar]
- 32.Yablonskiy DA, Haacke EM. Theory of NMR signal behavior in magnetically inhomogeneous tissues: the static dephasing regime. Magn Reson Med. 1994;32:749–763. doi: 10.1002/mrm.1910320610. [DOI] [PubMed] [Google Scholar]
- 33.Haacke EM, Lai S, Reichenbach JR, Kuppusamy K, Hoogenraad FG, Takeichi H, Lin W. In vivo measurement of blood oxygen saturation using magnetic resonance imaging: a direct validation of the blood oxygen level dependent concept in functional brain imaging. Human Brain Mapping. 1997;5:341–346. doi: 10.1002/(SICI)1097-0193(1997)5:5<341::AID-HBM2>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 34.Fernandez-Seara MA, Techawiboonwong A, Detre JA, Wehrli FW. MR susceptometry for measuring global brain oxygen extraction. Magn Reson Med. 2006;55:967–973. doi: 10.1002/mrm.20892. [DOI] [PubMed] [Google Scholar]
- 35.Bolar DS, Rosen BR, Sorensen AG, Adalsteinsson E. Proc Intl Soc Mag Reson Med. Honolulu, HI: 2009. Quantitative imaging of extraction of oxygen and tissue consumption (QUIXOTIC) using velocity selective arterial spin labeling; p. 628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pell GS, Thomas DL, Lythgoe MF, Calamante F, Howseman AM, Gadian DG, Ordidge RJ. Implementation of quantitative FAIR perfusion imaging with a short repetition time in time-course studies. Magn Reson Med. 1999;41:829–840. doi: 10.1002/(sici)1522-2594(199904)41:4<829::aid-mrm24>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- 37.Lu H. Proc Intl Soc Mag Reson Med. Toronto, Canada: 2008. Magnetization “reset” for non-steady-state blood spins in Vascular-Space-Occupancy (VASO) fMRI; p. 406. [Google Scholar]
- 38.Hendrikse J, Lu H, van der Grond J, Van Zijl PC, Golay X. Measurements of cerebral perfusion and arterial hemodynamics during visual stimulation using TURBO-TILT. Magn Reson Med. 2003;50:429–433. doi: 10.1002/mrm.10525. [DOI] [PubMed] [Google Scholar]
- 39.Wang J, Alsop DC, Li L, Listerud J, Gonzalez-At JB, Schnall MD, Detre JA. Comparison of quantitative perfusion imaging using arterial spin labeling at 1.5 and 4.0 Tesla. Magn Reson Med. 2002;48:242–254. doi: 10.1002/mrm.10211. [DOI] [PubMed] [Google Scholar]