Abstract
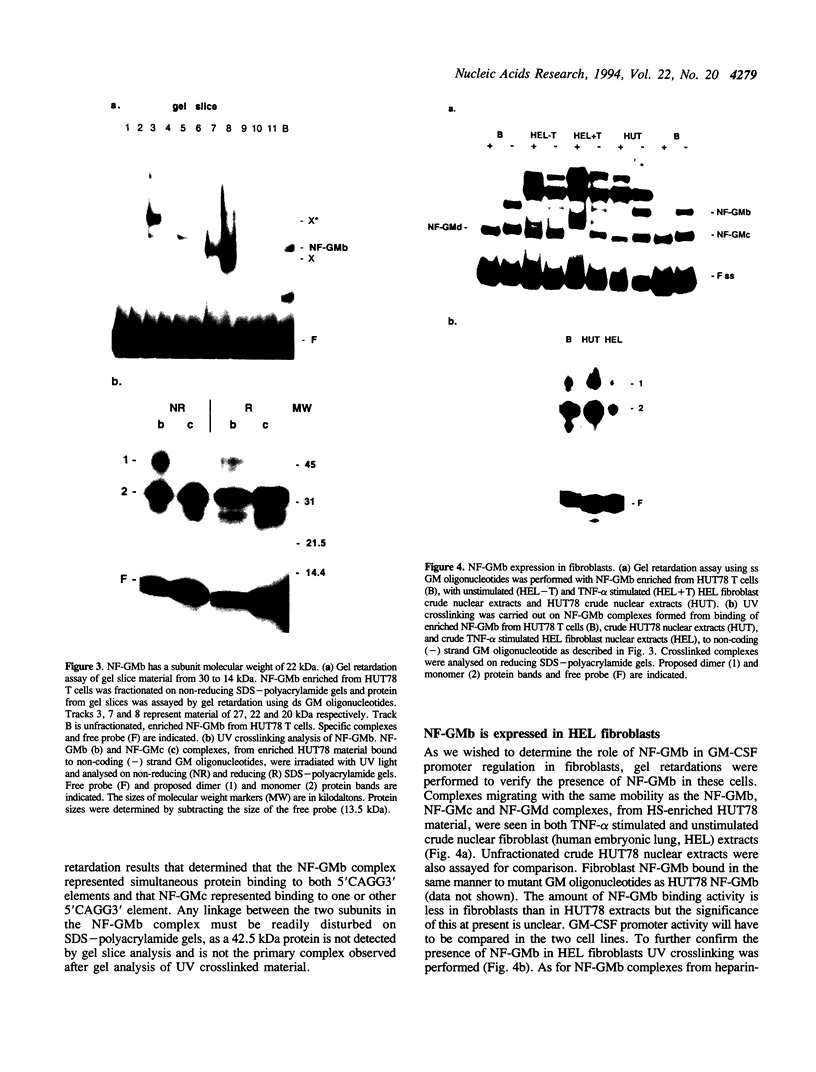
NF-GMb is a nuclear factor that binds to the proximal promoter of the human granulocyte-macrophage colony stimulating factor (GM-CSF) gene. NF-GMb has a subunit molecular weight of 22 kDa, is constitutively expressed in embryonic fibroblasts and binds to sequences within the adjacent CK-1 and CK-2 elements (CK-1/CK-2 region), located at approximately -100 in the GM-CSF gene promoter. These elements are conserved in haemopoietic growth factor (HGF) genes. NF-GMb binding requires the presence of repeated 5'CAGG3' sequences that overlap the binding sites for positive activators. Surprisingly, NF-GMb was found to bind solely to single-strand DNA, namely the non-coding strand of the GM-CSF CK-1/CK-2 region. NF-GMb may belong to a family of single-strand DNA binding (ssdb) proteins that have 5'CAGG3' sequences within their binding sites. Functional analysis of the proximal GM-CSF promoter revealed that sequences in the -114 to -79 region of the promoter containing the NF-GMb binding sites had no intrinsic activity in fibroblasts but could, however, repress tumour necrosis factor-alpha (TNF-alpha) inducible expression directed by downstream promoter sequences (-65 to -31). Subsequent mutation analysis showed that sequences involved in repression correlated with those required for NF-GMb binding.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arai K. I., Lee F., Miyajima A., Miyatake S., Arai N., Yokota T. Cytokines: coordinators of immune and inflammatory responses. Annu Rev Biochem. 1990;59:783–836. doi: 10.1146/annurev.bi.59.070190.004031. [DOI] [PubMed] [Google Scholar]
- Baeuerle P. A., Baltimore D. Activation of DNA-binding activity in an apparently cytoplasmic precursor of the NF-kappa B transcription factor. Cell. 1988 Apr 22;53(2):211–217. doi: 10.1016/0092-8674(88)90382-0. [DOI] [PubMed] [Google Scholar]
- Brorson K. A., Beverly B., Kang S. M., Lenardo M., Schwartz R. H. Transcriptional regulation of cytokine genes in nontransformed T cells. Apparent constitutive signals in run-on assays can be caused by repeat sequences. J Immunol. 1991 Nov 15;147(10):3601–3609. [PubMed] [Google Scholar]
- Broudy V. C., Kaushansky K., Segal G. M., Harlan J. M., Adamson J. W. Tumor necrosis factor type alpha stimulates human endothelial cells to produce granulocyte/macrophage colony-stimulating factor. Proc Natl Acad Sci U S A. 1986 Oct;83(19):7467–7471. doi: 10.1073/pnas.83.19.7467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunel F., Alzari P. M., Ferrara P., Zakin M. M. Cloning and sequencing of PYBP, a pyrimidine-rich specific single strand DNA-binding protein. Nucleic Acids Res. 1991 Oct 11;19(19):5237–5245. doi: 10.1093/nar/19.19.5237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan J. Y., Slamon D. J., Nimer S. D., Golde D. W., Gasson J. C. Regulation of expression of human granulocyte/macrophage colony-stimulating factor. Proc Natl Acad Sci U S A. 1986 Nov;83(22):8669–8673. doi: 10.1073/pnas.83.22.8669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L. L., Clawson M. L., Bilgrami S., Carmichael G. A sequence-specific single-stranded DNA-binding protein that is responsive to epidermal growth factor recognizes an S1 nuclease-sensitive region in the epidermal growth factor receptor promoter. Cell Growth Differ. 1993 Dec;4(12):975–983. [PubMed] [Google Scholar]
- Cockerill P. N., Shannon M. F., Bert A. G., Ryan G. R., Vadas M. A. The granulocyte-macrophage colony-stimulating factor/interleukin 3 locus is regulated by an inducible cyclosporin A-sensitive enhancer. Proc Natl Acad Sci U S A. 1993 Mar 15;90(6):2466–2470. doi: 10.1073/pnas.90.6.2466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dignam J. D., Lebovitz R. M., Roeder R. G. Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 1983 Mar 11;11(5):1475–1489. doi: 10.1093/nar/11.5.1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan R., Bazar L., Michelotti G., Tomonaga T., Krutzsch H., Avigan M., Levens D. A sequence-specific, single-strand binding protein activates the far upstream element of c-myc and defines a new DNA-binding motif. Genes Dev. 1994 Feb 15;8(4):465–480. doi: 10.1101/gad.8.4.465. [DOI] [PubMed] [Google Scholar]
- Finocchiaro L. M., Amati P., Glikin G. C. Single strand binding protein specific for the polyoma early-coding strand of PEA1 (AP1) regulatory sequence. Nucleic Acids Res. 1991 Aug 11;19(15):4279–4287. doi: 10.1093/nar/19.15.4279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaillard C., Strauss F. Sequence-specific single-strand-binding protein for the simian virus 40 early promoter stimulates transcription in vitro. J Mol Biol. 1990 Sep 20;215(2):245–255. doi: 10.1016/S0022-2836(05)80343-2. [DOI] [PubMed] [Google Scholar]
- Gaillard C., Weber M., Strauss F. A sequence-specific single-strand-binding protein for the late-coding strand of the simian virus 40 control region. J Virol. 1988 Jul;62(7):2380–2385. doi: 10.1128/jvi.62.7.2380-2385.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasson J. C. Molecular physiology of granulocyte-macrophage colony-stimulating factor. Blood. 1991 Mar 15;77(6):1131–1145. [PubMed] [Google Scholar]
- Grayhack E. J. The yeast alpha 1 and MCM1 proteins bind a single strand of their duplex DNA recognition site. Mol Cell Biol. 1992 Aug;12(8):3573–3582. doi: 10.1128/mcb.12.8.3573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heike T., Miyatake S., Yoshida M., Arai K., Arai N. Bovine papilloma virus encoded E2 protein activates lymphokine genes through DNA elements, distinct from the consensus motif, in the long control region of its own genome. EMBO J. 1989 May;8(5):1411–1417. doi: 10.1002/j.1460-2075.1989.tb03522.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Himes S. R., Coles L. S., Katsikeros R., Lang R. K., Shannon M. F. HTLV-1 tax activation of the GM-CSF and G-CSF promoters requires the interaction of NF-kB with other transcription factor families. Oncogene. 1993 Dec;8(12):3189–3197. [PubMed] [Google Scholar]
- Hollingsworth M. A., Closken C., Harris A., McDonald C. D., Pahwa G. S., Maher L. J., 3rd A nuclear factor that binds purine-rich, single-stranded oligonucleotides derived from S1-sensitive elements upstream of the CFTR gene and the MUC1 gene. Nucleic Acids Res. 1994 Apr 11;22(7):1138–1146. doi: 10.1093/nar/22.7.1138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jansen-Dürr P., Boshart M., Lupp B., Bosserhoff A., Frank R. W., Schütz G. The rat poly pyrimidine tract binding protein (PTB) interacts with a single-stranded DNA motif in a liver-specific enhancer. Nucleic Acids Res. 1992 Mar 25;20(6):1243–1249. doi: 10.1093/nar/20.6.1243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaushansky K. Control of granulocyte-macrophage colony-stimulating factor production in normal endothelial cells by positive and negative regulatory elements. J Immunol. 1989 Oct 15;143(8):2525–2529. [PubMed] [Google Scholar]
- Kilpatrick M. W., Torri A., Kang D. S., Engler J. A., Wells R. D. Unusual DNA structures in the adenovirus genome. J Biol Chem. 1986 Aug 25;261(24):11350–11354. [PubMed] [Google Scholar]
- Kolluri R., Torrey T. A., Kinniburgh A. J. A CT promoter element binding protein: definition of a double-strand and a novel single-strand DNA binding motif. Nucleic Acids Res. 1992 Jan 11;20(1):111–116. doi: 10.1093/nar/20.1.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuczek E. S., Shannon M. F., Pell L. M., Vadas M. A. A granulocyte-colony-stimulating factor gene promoter element responsive to inflammatory mediators is functionally distinct from an identical sequence in the granulocyte-macrophage colony-stimulating factor gene. J Immunol. 1991 Apr 1;146(7):2426–2433. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Larsen A., Weintraub H. An altered DNA conformation detected by S1 nuclease occurs at specific regions in active chick globin chromatin. Cell. 1982 Jun;29(2):609–622. doi: 10.1016/0092-8674(82)90177-5. [DOI] [PubMed] [Google Scholar]
- Masuda E. S., Tokumitsu H., Tsuboi A., Shlomai J., Hung P., Arai K., Arai N. The granulocyte-macrophage colony-stimulating factor promoter cis-acting element CLE0 mediates induction signals in T cells and is recognized by factors related to AP1 and NFAT. Mol Cell Biol. 1993 Dec;13(12):7399–7407. doi: 10.1128/mcb.13.12.7399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathey-Prevot B., Andrews N. C., Murphy H. S., Kreissman S. G., Nathan D. G. Positive and negative elements regulate human interleukin 3 expression. Proc Natl Acad Sci U S A. 1990 Jul;87(13):5046–5050. doi: 10.1073/pnas.87.13.5046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metcalf D. Hemopoietic regulators. Trends Biochem Sci. 1992 Aug;17(8):286–289. doi: 10.1016/0968-0004(92)90436-d. [DOI] [PubMed] [Google Scholar]
- Metcalf D. The molecular control of cell division, differentiation commitment and maturation in haemopoietic cells. Nature. 1989 May 4;339(6219):27–30. doi: 10.1038/339027a0. [DOI] [PubMed] [Google Scholar]
- Miyatake S., Seiki M., Yoshida M., Arai K. T-cell activation signals and human T-cell leukemia virus type I-encoded p40x protein activate the mouse granulocyte-macrophage colony-stimulating factor gene through a common DNA element. Mol Cell Biol. 1988 Dec;8(12):5581–5587. doi: 10.1128/mcb.8.12.5581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyatake S., Shlomai J., Arai K., Arai N. Characterization of the mouse granulocyte-macrophage colony-stimulating factor (GM-CSF) gene promoter: nuclear factors that interact with an element shared by three lymphokine genes--those for GM-CSF, interleukin-4 (IL-4), and IL-5. Mol Cell Biol. 1991 Dec;11(12):5894–5901. doi: 10.1128/mcb.11.12.5894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukherjee R., Chambon P. A single-stranded DNA-binding protein promotes the binding of the purified oestrogen receptor to its responsive element. Nucleic Acids Res. 1990 Oct 11;18(19):5713–5716. doi: 10.1093/nar/18.19.5713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munker R., Gasson J., Ogawa M., Koeffler H. P. Recombinant human TNF induces production of granulocyte-monocyte colony-stimulating factor. Nature. 1986 Sep 4;323(6083):79–82. doi: 10.1038/323079a0. [DOI] [PubMed] [Google Scholar]
- Nickol J. M., Felsenfeld G. DNA conformation at the 5' end of the chicken adult beta-globin gene. Cell. 1983 Dec;35(2 Pt 1):467–477. doi: 10.1016/0092-8674(83)90180-0. [DOI] [PubMed] [Google Scholar]
- Nicola N. A. Hemopoietic cell growth factors and their receptors. Annu Rev Biochem. 1989;58:45–77. doi: 10.1146/annurev.bi.58.070189.000401. [DOI] [PubMed] [Google Scholar]
- Nimer S. D., Gasson J. C., Hu K., Smalberg I., Williams J. L., Chen I. S., Rosenblatt J. D. Activation of the GM-CSF promoter by HTLV-I and -II tax proteins. Oncogene. 1989 Jun;4(6):671–676. [PubMed] [Google Scholar]
- Nimer S. D., Morita E. A., Martis M. J., Wachsman W., Gasson J. C. Characterization of the human granulocyte-macrophage colony-stimulating factor promoter region by genetic analysis: correlation with DNase I footprinting. Mol Cell Biol. 1988 May;8(5):1979–1984. doi: 10.1128/mcb.8.5.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimer S., Fraser J., Richards J., Lynch M., Gasson J. The repeated sequence CATT(A/T) is required for granulocyte-macrophage colony-stimulating factor promoter activity. Mol Cell Biol. 1990 Nov;10(11):6084–6088. doi: 10.1128/mcb.10.11.6084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimer S. Tax responsiveness of the GM-CSF promoter is mediated by mitogen-inducible sequences other than kappa B. New Biol. 1991 Oct;3(10):997–1004. [PubMed] [Google Scholar]
- Nourbakhsh M., Hoffmann K., Hauser H. Interferon-beta promoters contain a DNA element that acts as a position-independent silencer on the NF-kappa B site. EMBO J. 1993 Feb;12(2):451–459. doi: 10.1002/j.1460-2075.1993.tb05677.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peritz L. N., Fodor E. J., Silversides D. W., Cattini P. A., Baxter J. D., Eberhardt N. L. The human growth hormone gene contains both positive and negative control elements. J Biol Chem. 1988 Apr 15;263(11):5005–5007. [PubMed] [Google Scholar]
- Rajavashisth T. B., Taylor A. K., Andalibi A., Svenson K. L., Lusis A. J. Identification of a zinc finger protein that binds to the sterol regulatory element. Science. 1989 Aug 11;245(4918):640–643. doi: 10.1126/science.2562787. [DOI] [PubMed] [Google Scholar]
- Santoro I. M., Yi T. M., Walsh K. Identification of single-stranded-DNA-binding proteins that interact with muscle gene elements. Mol Cell Biol. 1991 Apr;11(4):1944–1953. doi: 10.1128/mcb.11.4.1944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreck R., Baeuerle P. A. NF-kappa B as inducible transcriptional activator of the granulocyte-macrophage colony-stimulating factor gene. Mol Cell Biol. 1990 Mar;10(3):1281–1286. doi: 10.1128/mcb.10.3.1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seelentag W., Mermod J. J., Vassalli P. Interleukin 1 and tumor necrosis factor-alpha additively increase the levels of granulocyte-macrophage and granulocyte colony-stimulating factor (CSF) mRNA in human fibroblasts. Eur J Immunol. 1989 Jan;19(1):209–212. doi: 10.1002/eji.1830190135. [DOI] [PubMed] [Google Scholar]
- Shannon M. F., Gamble J. R., Vadas M. A. Nuclear proteins interacting with the promoter region of the human granulocyte/macrophage colony-stimulating factor gene. Proc Natl Acad Sci U S A. 1988 Feb;85(3):674–678. doi: 10.1073/pnas.85.3.674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shannon M. F., Pell L. M., Lenardo M. J., Kuczek E. S., Occhiodoro F. S., Dunn S. M., Vadas M. A. A novel tumor necrosis factor-responsive transcription factor which recognizes a regulatory element in hemopoietic growth factor genes. Mol Cell Biol. 1990 Jun;10(6):2950–2959. doi: 10.1128/mcb.10.6.2950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shoemaker S. G., Hromas R., Kaushansky K. Transcriptional regulation of interleukin 3 gene expression in T lymphocytes. Proc Natl Acad Sci U S A. 1990 Dec;87(24):9650–9654. doi: 10.1073/pnas.87.24.9650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sieff C. A., Niemeyer C. M., Mentzer S. J., Faller D. V. Interleukin-1, tumor necrosis factor, and the production of colony-stimulating factors by cultured mesenchymal cells. Blood. 1988 Oct;72(4):1316–1323. [PubMed] [Google Scholar]
- Smith M. R., Greene W. C. The same 50-kDa cellular protein binds to the negative regulatory elements of the interleukin 2 receptor alpha-chain gene and the human immunodeficiency virus type 1 long terminal repeat. Proc Natl Acad Sci U S A. 1989 Nov;86(21):8526–8530. doi: 10.1073/pnas.86.21.8526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stark H. C., Weinberger O., Weinberger J. Common double- and single-stranded DNA binding factor for a sterol regulatory element. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2180–2184. doi: 10.1073/pnas.89.6.2180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugimoto K., Tsuboi A., Miyatake S., Arai K., Arai N. Inducible and non-inducible factors co-operatively activate the GM-CSF promoter by interacting with two adjacent DNA motifs. Int Immunol. 1990;2(8):787–794. doi: 10.1093/intimm/2.8.787. [DOI] [PubMed] [Google Scholar]
- Thorens B., Mermod J. J., Vassalli P. Phagocytosis and inflammatory stimuli induce GM-CSF mRNA in macrophages through posttranscriptional regulation. Cell. 1987 Feb 27;48(4):671–679. doi: 10.1016/0092-8674(87)90245-5. [DOI] [PubMed] [Google Scholar]
- Wang Z. Y., Lin X. H., Nobuyoshi M., Deuel T. F. Identification of a single-stranded DNA-binding protein that interacts with an S1 nuclease-sensitive region in the platelet-derived growth factor A-chain gene promoter. J Biol Chem. 1993 May 15;268(14):10681–10685. [PubMed] [Google Scholar]
- Wilkison W. O., Min H. Y., Claffey K. P., Satterberg B. L., Spiegelman B. M. Control of the adipsin gene in adipocyte differentiation. Identification of distinct nuclear factors binding to single- and double-stranded DNA. J Biol Chem. 1990 Jan 5;265(1):477–482. [PubMed] [Google Scholar]
- Williams T. M., Moolten D., Burlein J., Romano J., Bhaerman R., Godillot A., Mellon M., Rauscher F. J., 3rd, Kant J. A. Identification of a zinc finger protein that inhibits IL-2 gene expression. Science. 1991 Dec 20;254(5039):1791–1794. doi: 10.1126/science.1840704. [DOI] [PubMed] [Google Scholar]
- Wong G. G., Witek J. S., Temple P. A., Wilkens K. M., Leary A. C., Luxenberg D. P., Jones S. S., Brown E. L., Kay R. M., Orr E. C. Human GM-CSF: molecular cloning of the complementary DNA and purification of the natural and recombinant proteins. Science. 1985 May 17;228(4701):810–815. doi: 10.1126/science.3923623. [DOI] [PubMed] [Google Scholar]
- Yu Y. T., Manley J. L. Structure and function of the S1 nuclease-sensitive site in the adenovirus late promoter. Cell. 1986 Jun 6;45(5):743–751. doi: 10.1016/0092-8674(86)90788-9. [DOI] [PubMed] [Google Scholar]