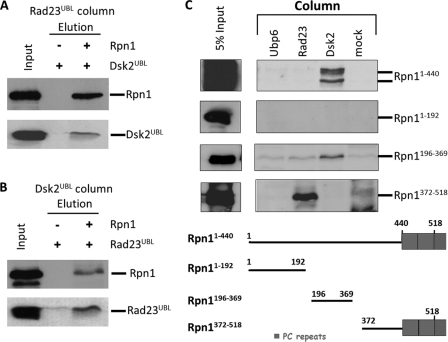
FIGURE 5.
Dsk2 and Rad23 bind to two different sites at Rpn1. A and B, immobilized Rad23UBL was incubated with either Dsk2UBL or a premixed Rpn1 + Dsk2UBL mixture and washed, and bound proteins were eluted and immunoblotted for the presence of Dsk2. The reciprocal experiment was repeated with Dsk2UBL immobilized and incubated with either Rad23UBL or a Rpn1 + Rad23UBL mixture. The formation of these Rad23-Rpn1-Dsk2 ternary complexes demonstrates that Rad23 and Dsk2 can bind simultaneously to Rpn1, via at least two discrete binding sites. C, purified recombinant N-terminal fragments of Rpn1 were washed over immobilized binding partners, washed, and elution immunoblotted for presence of Rpn1. A Dsk2-binding site is located between residues 196 and 369 of Rpn1, whereas Rad23 binds tighter to an Rpn1 fragment spanning residues 372–518.