FIGURE 6.
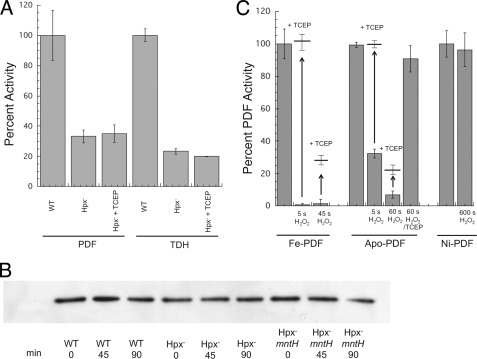
Extended exposure to H2O2 converts PDF and TDH to nonreactivatible forms in vivo and in vitro. A, TCEP did not restore PDF and TDH activities in the extracts of Hpx− cells that had been grown in aerobic medium. Both enzymes were assayed in the presence of metal (500 μm Ni2+ and 500 μm Fe2+ respectively) to ensure full activity of undamaged enzymes. B, PDF polypeptide was not degraded in vivo. Anaerobic cells expressing PDF-FLAG were treated with chloramphenicol and aerated starting at time 0. Polypeptide content was monitored by Western blot. The strains used were MG1655/pPDF-FLAG, LC106/pPDF-FLAG (Hpx−) and AA30/pPDF-FLAG (Hpx− ΔmntH). C, iron-PDF and apo-PDF can be overoxidized in vitro. Iron-charged PDF, apo-PDF, and nickel-charged PDF were exposed to H2O2 (133, 100, and 500 μm, respectively) for the indicated times. They were then treated ± TCEP and reconstituted with nickel prior to assay.