Abstract
AIM: To study the expression of eukaryotic translation initiation factor 4E (eIF4E), which is closely correlated with malignant tumors, and its relationship to prognosis in hepatocellular carcinoma.
METHODS: Western blotting was performed to quantify the elF4E protein expression in the normal human liver cell line L02 and the hepatoma cell lines Hep3B, HepG2, and Huh7. Forty-six hepatocellular carcinoma samples with complete clinical data were obtained from Changzheng Hospital during the period of December 2008 to July 2009. The expression of eIF4E in the tumor samples and their adjacent tissues were detected by immunohistochemistry. The relationship between the test results and hepatocellular carcinoma (HCC) prognosis was statistically analysed by using a COX proportional hazard model.
RESULTS: Western blotting analysis showed that there were distinct eIF4E protein bands in all three of the hepatoma cell lines. In particular, the HepG2 cell line had the highest level of eIF4E protein expression. The L02 cell group had a low eIF4E expression. Immunohistochemical assay showed that there were 32 cases in which the tumour tissue expression was higher than their adjacent tissues, accounting for 69.57%. There were also 14 cases in which the tumour tissue expression was lower or no significant difference was found, accounting for 30.43%. COX proportional hazards model analysis showed that HCC prognosis was related to the depth of invasion, the overexpression of eIF4E and p53, possibly as independent HCC prognostic predictors.
CONCLUSION: In summary, eIF4E expression is associated with liver cancer, and patients with high eIF4E expression levels have a higher risk of recurrence.
Keywords: Hepatocellular carcinoma, Eukaryotic translation initiation factor 4E, Western blotting, Immunohistochemistry, Prognosis
INTRODUCTION
Eukaryotic translation initiation factor 4E (eIF4E) is a member of the eIF family. It can specifically bind to the cap structure located at the 5’ end of mRNAs named the “m7GpppN cap”, which is necessary for mRNA translation initiation, and affects mRNA metabolism, processing, transportation and translation[1]. It plays an important role in regulating the initial stage protein synthesis[2,3]. eIF4E is highly expressed in a variety of human malignancies[4-7], which has been confirmed to be relevant to the occurrence, invasion and metastasis of carcinomas such as head and neck squamous cell carcinoma[8], laryngeal cancer, non-small cell lung cancer[9-11], breast cancer[12-18], thyroid cancer, oesophageal cancer, stomach cancer, cholangiocarcinoma, colon cancer[19], non-Hodgkin’s lymphoma, acute or chronic myeloid leukaemia[20], and lymphoma[21,22]. Experiments have also confirmed that eIF4E is closely related to the prognosis of many carcinomas. However, eIF4E-related studies in the context of hepatocellular carcinoma (HCC) are still rare.
In this study, we separately compared the eIF4E expression levels in normal liver cells with liver cancer cell lines and liver cancer tissues with precancerous tissues. Additionally, we investigated the influence of eIF4E expression on the prognosis of liver cancer. This research may provide an experimental basis for exploring new ways to treat liver cancer.
MATERIALS AND METHODS
Study objects
We selected 46 patients with pathological evidence of HCC and complete clinical data from Shanghai Changzheng Hospital who had liver surgery from January 2007 to January 2009. In these 46 cases, there were 40 males and 6 females who ranged in age from 31 years to 77 years (median age: 52.26 years). With regards to histological grade, there were 42 cases of moderately differentiated HCC, and 4 cases were poorly differentiated. A total of 33 patients had a cancer embolus in the intrahepatic bile duct or vein or had an infiltrated peplos, and 13 patients had no cancer tissue in cutting edge and gallbladder and no infiltrated peplos. p53 pathological testing was positive in 39 patients and negative in 7 cases. None of the patients received preoperative radiotherapy or chemotherapy. The follow-up time was 24 mo, and no case was lost.
Major materials and reagents
The cell lines used for Western blotting were the human liver cancer cell lines Hep3B, HepG2, Huh7, and the normal human liver cell line L02, which was provided by Shanghai Cell Biology Institution of Academia Sinica.
eIF4E (P-2) is a mouse anti-human monoclonal antibody raised against full-length eIF4E (Santa Cruz Biotechnology, Inc.). It is recommended for the detection of eIF4E by Western blotting (dilution: 1:200; dilution range: 1:100-1:1000) and immunohistochemistry (including paraffin-embedded sections; dilution: 1:50; dilution range: 1:50-1:500). The streptavidin-perosidase (SP) kit was provided by Fuzhou Maixin Biotechnology, Inc.
Detection methods
Western blotting analysis: We tested the eIF4E gene expression level in normal liver cells and different liver cancer cell lines. The four of cell lines (i.e., HepG2, Hep3B, Huh7, and L02) were incubated with high glucose Dulbecco's modified Eagle's medium containing 10% fetal bovine serum at 37.0 °C with 5% CO2 until the cell concentration reached 5 × 106 cells/mL. Then, we sequentially performed the protein extraction, bicinchoninic acid protein quantification, sodium dodecyl sulfate-polyacrylamide gel electrophoresis electrophoresis, protein transfer, membrane closure, antibody incubation, and Bio-Rad chemiluminescence.
Immunohistochemistry: We detected the eIF4E protein expression levels in HCC and their adjacent tissues (SP, a particular type of immunohistochemistry). The tumour and adjacent tissues from the same patient were fixed, dehydrated, sectioned, and made into paraffin biopsies. We made 46 paraffin sections. The steps of the SP kit included heating on a baking sheet, incubation, washing, sealing, staining, drying, dehydration, and mounting. To analyse the results, we used two scoring methods. The samples were placed in an electron microscope and were scored for staining intensity as follows. 0: No colour; 1: A yellow colour; 2: A claybank colour; and 3: A brown colour. We then graded the samples for the positivity rate as follows. 0: No positive tumour cell staining; 1: ≤ 10% positive cells; 2: 11% to 50% positive cells; 3: 51% to 75% positive cells; and 4: > 75% positive cells. Finally, we added the two scores together, and the sum represented the immunohistochemical score as follows. -: 0; +: 1 to 4; ++: 5 to 8; and + + +: 9 to 12. Each cancer tissue section was compared with its adjacent tissue.
Follow up: We analysed the number of cases that had HCC recurrence and metastasis during the post-operative 24 mo. The liver cancer recurrence risk was measured using COX proportional hazards model for statistical analysis. The patient age, gender, histological grade, depth of invasion, eIF4E, p53 status and other prognostic indicators were used for the COX proportional hazards model analysis.
Statistical analysis
We used the SPSS 17.0 statistical software for the statistical analysis. A COX proportional hazards model testing level of α = 0.05 and a P < 0.05 was considered statistically significant.
RESULTS
eIF4E protein expression in liver cancer cell lines by western blotting
We tested the eIF4E protein expression level in the liver cell line L02 and the liver cancer cell lines Huh7, HepG2, Hep3B. The eIF4E protein bands are shown in Figure 1. The bands were detected by Bio-Rad chemiluminescence to obtain the data shown in Table 1.
Figure 1.
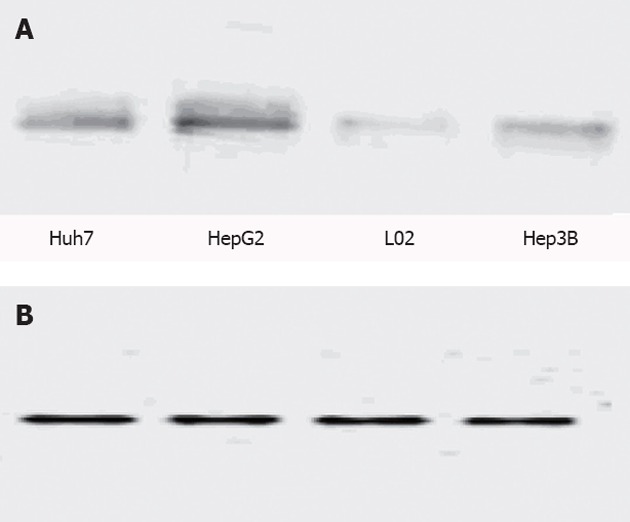
Eukaryotic translation initiation factor 4E protein and glyceraldehyde-3-phosphate dehydrogenase protein bands in a normal human liver cell line and three hepatoma carcinoma cell lines. A: Eukaryotic translation initiation factor 4E protein bands; B: Glyceraldehyde-3-phosphate dehydrogenase protein bands.
Table 1.
Eukaryotic translation initiation factor 4E protein expression in the liver cells at different levels
| Huh7 | HepG2 | L02 | Hep3B | |
| GAPDH | 827.165 | 884.682 | 885.437 | 848.552 |
| eIF4E | 3161.861 | 5651.885 | 775.440 | 4496.191 |
GAPDH: Glyceraldehyde-3-phosphate dehydrogenase; eIF4E: Eukaryotic translation initiation factor 4E.
The liver cancer cell lines HepG2, Huh7, Hep3B significantly expressed the eIF4E protein, and in particular, the HepG2 cell line had the highest level of eIF4E protein expression. The normal liver cell L02 also expressed eIF4E; however, its expression level was low. Glyceraldehyde-3-phosphate dehydrogenase, which was used as the internal reference had bands in each cell line, and no obvious differences were observed with this protein.
eIF4E protein expression in liver cancer and adjacent tissues by immunohistochemistry
We next detected the eIF4E protein expression level in HCC and adjacent tissues. There were 46 pathological tissue paraffin blocks in which 32 HCC tissue cases had higher eIF4E protein expression than their adjacent tissues, accounting for 69.57%. A total of 14 HCC tissue cases had lower expression or no significant difference compared with their adjacent tissues, accounting for 30.43%. The scores were weighted 154:97 = 1.59:1, meaning that, in general, HCC tissues had a higher eIF4E protein expression level than the adjacent tissues.
Figure 2 show that HCC tissues stained significantly stronger than adjacent tissues, indicating that tumour tissues had a higher eIF4E protein expression level. The lightly stained central area in Figure 2A represents necrotic tissue.
Figure 2.
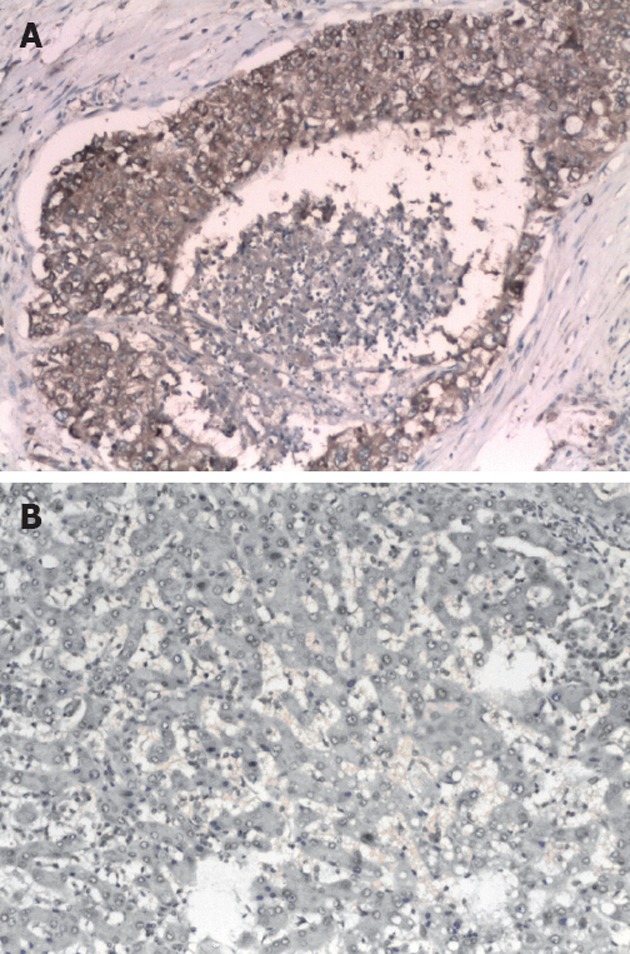
Photo of hepatocellular carcinoma tissue and adjacent tissue by electron microscopy. A: Hepatocellular carcinoma tissue and adjacent interstitial tissue; B: Adjacent tissue. (Hematoxylin and eosin stain, ×100).
eIF4E may be an independent risk factor for liver cancer prognosis
Follow-up statistics showed recurrence in 33 cases and death in 12 cases. The patient age, gender, histological grade, depth of invasion, eIF4E overexpression, p53 positive status and other prognostic indicators were used for COX proportional hazards model for screening analysis. Ultimately, the depth of invasion, eIF4E, and p53 were included in the model with a Sig < 0.05 as shown in Table 2. The statistical significance suggests that these three factors are independent risk factors for liver cancer prognosis.
Table 2.
COX proportional hazards model analysis
|
95% Cl for Exp(B) |
||||||||
| B | SE | Wald | df | Sig | Exp(B) | Lower | Upper | |
| Eukaryotic translation initiation factor 4E | 1.971 | 0.926 | 4.529 | 1 | 0.033 | 7.179 | 1.169 | 44.100 |
| Depth of invasion | 3.122 | 1.211 | 6.650 | 1 | 0.010 | 22.690 | 2.115 | 243.423 |
| Histological grade | 0.410 | 1.156 | 0.126 | 1 | 0.723 | 1.506 | 0.156 | 14.527 |
| Gender | 1.671 | 1.152 | 2.104 | 1 | 0.147 | 5.319 | 0.556 | 50.890 |
| Age | -0.017 | 0.028 | 0.354 | 1 | 0.552 | 0.983 | 0.930 | 1.040 |
| p53 | -3.208 | 0.825 | 15.118 | 1 | 0.000 | 0.040 | 0.008 | 0.204 |
B: Coefficient of regression; SE.: Standard error; Wald: The index of regression effect; df: Degrees of freedom; Sig: P value; Exp(B): Odds ratio.
DISCUSSION
In eukaryotic cells, translational regulation plays an important role in gene expression. eIF4E is involved in the regulation of the mRNA translation process. It can enhance the translation of some important growth factors and cell growth regulators and affect protein synthesis, the cell cycle, cancer gene activation, and apoptosis; it also plays an important role in malignant transformation and metastasis.
eIF4E regulates the translation of cancer-related mRNAs (i.e., it is involved in the activation of proto-oncogenes, angiogenesis, apoptosis, invasion and metastasis) that are involved in tumour occurrence and development. Normal tissues have a low eIF4E expression level. eIF4E was overexpressed, in a variety of malignant tumours including head and neck squamous cell carcinoma, laryngeal cancer, lung cancer, breast cancer, thyroid cancer and other cancer tissues[4,23]. Its high expression was correlated with tumour invasion and metastasis. However, studies of eIF4E in liver cancer are rare. At present, there are studies that involve the targeting eIF4E in head and neck squamous cell carcinoma[24,25], breast cancer[13-18], non-small cell lung cancer[26], blood malignancies[27-29] and other studies[6,30-32]. However, few studies have focused on targeting eIF4E in HCC.
In this study, we tested the expression of eIF4E protein in a normal human liver cell line and three different liver cancer cell lines. eIF4E protein expression was high in the three liver cancer cell lines and higher than in the normal liver cell L02. The HepG2 cell line had an especially high level of eIF4E protein expression. By comparing 46 cases of human liver cancer and adjacent tissues, we found that eIF4E protein expression was higher in most of the cancer tissues than in the adjacent tissues. COX proportional hazards model analysis showed that the depth of invasion, eIF4E, and p53 status were independent risk factors of liver cancer prognosis.
Based on these studies, we believe that eIF4E protein expression may be closely associated with the occurrence of human liver cancer development and prognosis. It has been confirmed in vivo and in vitro that sorafenib treatment can inhibit the RAF/MEK/ERK signal transduction pathway, reduce the eIF4E phosphorylation level, reduce Mcl-l protein, and induce hepatoma cell apoptosis[33,34]. Accordingly, we suggest that lower levels of eIF4E gene expression may inhibit liver cancer. Targeting and adjusting the eIF4E level and activity may inhibit cancer cell growth[6,30,31,35], which may become a new paradigm in the field of the biological treatment of liver cancer[36].
COMMENTS
Background
Hepatocellular carcinoma (HCC), which has a poor prognosis and a low five-year survival rate, is the most common malignant tumour in our country. At present, there are no effective therapies including radiotherapy, chemotherapy, and surgery. Eukaryotic translation initiation factor 4E (eIF4E) plays an important role in the translation initiation phase of a eukaryotic cell. It has been confirmed that eIF4E can specifically bind to the 5’ mRNA cap (m7GpppN) and modulate its translation and expression. Its expression is closely associated with the generation, infiltration, and metastasis of many tumours such as head and neck, larynx, lung, mammary gland, thyroid gland, oesophagus, stomach, bile duct, colon.
Research frontiers
There are many researchers that are targeting eIF4E in head and neck squamous cell carcinoma, breast cancer, non-small cell lung cancer, blood malignancies and other carcinomas; however, studies that involve the targeting eIF4E in HCC are rare.
Innovations and breakthroughs
Research concerning the effects of eIF4E on HCC is limited. The authors tested the expression of the eIF4E protein in liver cancer cell lines and cancer tissues and used COX proportional hazards model analysis to show that eIF4E was an independent risk factor HCC prognosis.
Applications
The targeted regulation of the level and activity of eIF4E may inhibit cancer cell growth, which may become a new treatment paradigm in the liver cancer field.
Terminology
eIF4E is a member of the eIF family. It can specifically bind to the cap structure located at the 5’ end of mRNAs named the “m7GpppN cap”, which is necessary for mRNA translation initiation, and affects mRNA metabolism, processing, transportation and translation. It plays an important role in regulating the initial stage protein synthesis. eIF4E is highly expressed in a variety of human malignancies, which has been confirmed to be relevant to the occurrence, invasion and metastasis of carcinomas such as head and neck squamous cell carcinoma, laryngeal cancer, non-small cell lung cancer, breast, thyroid cancer, oesophageal cancer, stomach cancer, cholangiocarcinoma, colon cancer, non-Hodgkin’s lymphoma, acute or chronic myeloid leukaemia, and lymphoma. Experiments have also confirmed that eIF4E is closely related to the prognosis of many carcinomas.
Peer review
This paper is interesting and worth being published if authors can satisfactorily address the concerns raised regarding immunohistochemical expression of eIF4E.
Footnotes
Peer reviewer: Ezio Laconi, Professor, University of Cagliari, Via Porcell 4, 09125 Cagliari, Italy
S- Editor Gou SX L- Editor A E- Editor Zhang DN
References
- 1.Rhoads RE. eIF4E: new family members, new binding partners, new roles. J Biol Chem. 2009;284:16711–16715. doi: 10.1074/jbc.R900002200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sonenberg N. eIF4E, the mRNA cap-binding protein: from basic discovery to translational research. Biochem Cell Biol. 2008;86:178–183. doi: 10.1139/O08-034. [DOI] [PubMed] [Google Scholar]
- 3.Xu X, Vatsyayan J, Gao C, Bakkenist CJ, Hu J. Sumoylation of eIF4E activates mRNA translation. EMBO Rep. 2010;11:299–304. doi: 10.1038/embor.2010.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Furic L, Rong L, Larsson O, Koumakpayi IH, Yoshida K, Brueschke A, Petroulakis E, Robichaud N, Pollak M, Gaboury LA, et al. eIF4E phosphorylation promotes tumorigenesis and is associated with prostate cancer progression. Proc Natl Acad Sci USA. 2010;107:14134–14139. doi: 10.1073/pnas.1005320107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fan S, Ramalingam SS, Kauh J, Xu Z, Khuri FR, Sun SY. Phosphorylated eukaryotic translation initiation factor 4 (eIF4E) is elevated in human cancer tissues. Cancer Biol Ther. 2009;8:1463–1469. doi: 10.4161/cbt.8.15.8960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Thumma SC, Kratzke RA. Translational control: a target for cancer therapy. Cancer Lett. 2007;258:1–8. doi: 10.1016/j.canlet.2007.08.022. [DOI] [PubMed] [Google Scholar]
- 7.Noske A, Lindenberg JL, Darb-Esfahani S, Weichert W, Buckendahl AC, Röske A, Sehouli J, Dietel M, Denkert C. Activation of mTOR in a subgroup of ovarian carcinomas: correlation with p-eIF-4E and prognosis. Oncol Rep. 2008;20:1409–1417. [PubMed] [Google Scholar]
- 8.Sunavala-Dossabhoy G, Palaniyandi S, Clark C, Nathan CO, Abreo FW, Caldito G. Analysis of eIF4E and 4EBP1 mRNAs in head and neck cancer. Laryngoscope. 2011;121:2136–2141. doi: 10.1002/lary.22144. [DOI] [PubMed] [Google Scholar]
- 9.Khoury T, Alrawi S, Ramnath N, Li Q, Grimm M, Black J, Tan D. Eukaryotic initiation factor-4E and cyclin D1 expression associated with patient survival in lung cancer. Clin Lung Cancer. 2009;10:58–66. doi: 10.3816/CLC.2009.n.009. [DOI] [PubMed] [Google Scholar]
- 10.Wang R, Geng J, Wang JH, Chu XY, Geng HC, Chen LB. Overexpression of eukaryotic initiation factor 4E (eIF4E) and its clinical significance in lung adenocarcinoma. Lung Cancer. 2009;66:237–244. doi: 10.1016/j.lungcan.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 11.Yoshizawa A, Fukuoka J, Shimizu S, Shilo K, Franks TJ, Hewitt SM, Fujii T, Cordon-Cardo C, Jen J, Travis WD. Overexpression of phospho-eIF4E is associated with survival through AKT pathway in non-small cell lung cancer. Clin Cancer Res. 2010;16:240–248. doi: 10.1158/1078-0432.CCR-09-0986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McClusky DR, Chu Q, Yu H, Debenedetti A, Johnson LW, Meschonat C, Turnage R, McDonald JC, Abreo F, Li BD. A prospective trial on initiation factor 4E (eIF4E) overexpression and cancer recurrence in node-positive breast cancer. Ann Surg. 2005;242:584–590; discussion 590-592. doi: 10.1097/01.sla.0000184224.55949.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Holm N, Byrnes K, Johnson L, Abreo F, Sehon K, Alley J, Meschonat C, Md QC, Li BD. A prospective trial on initiation factor 4E (eIF4E) overexpression and cancer recurrence in node-negative breast cancer. Ann Surg Oncol. 2008;15:3207–3215. doi: 10.1245/s10434-008-0086-9. [DOI] [PubMed] [Google Scholar]
- 14.Zhou S, Wang GP, Liu C, Zhou M. Eukaryotic initiation factor 4E (eIF4E) and angiogenesis: prognostic markers for breast cancer. BMC Cancer. 2006;6:231. doi: 10.1186/1471-2407-6-231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wolfort R, de Benedetti A, Nuthalapaty S, Yu H, Chu QD, Li BD. Up-regulation of TLK1B by eIF4E overexpression predicts cancer recurrence in irradiated patients with breast cancer. Surgery. 2006;140:161–169. doi: 10.1016/j.surg.2006.05.001. [DOI] [PubMed] [Google Scholar]
- 16.Flowers A, Chu QD, Panu L, Meschonat C, Caldito G, Lowery-Nordberg M, Li BD. Eukaryotic initiation factor 4E overexpression in triple-negative breast cancer predicts a worse outcome. Surgery. 2009;146:220–226. doi: 10.1016/j.surg.2009.05.010. [DOI] [PubMed] [Google Scholar]
- 17.Coleman LJ, Peter MB, Teall TJ, Brannan RA, Hanby AM, Honarpisheh H, Shaaban AM, Smith L, Speirs V, Verghese ET, et al. Combined analysis of eIF4E and 4E-binding protein expression predicts breast cancer survival and estimates eIF4E activity. Br J Cancer. 2009;100:1393–1399. doi: 10.1038/sj.bjc.6605044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hiller DJ, Chu Q, Meschonat C, Panu L, Burton G, Li BD. Predictive value of eIF4E reduction after neoadjuvant therapy in breast cancer. J Surg Res. 2009;156:265–269. doi: 10.1016/j.jss.2009.03.060. [DOI] [PubMed] [Google Scholar]
- 19.Rosenwald IB, Chen JJ, Wang S, Savas L, London IM, Pullman J. Upregulation of protein synthesis initiation factor eIF-4E is an early event during colon carcinogenesis. Oncogene. 1999;18:2507–2517. doi: 10.1038/sj.onc.1202563. [DOI] [PubMed] [Google Scholar]
- 20.Topisirovic I, Guzman ML, McConnell MJ, Licht JD, Culjkovic B, Neering SJ, Jordan CT, Borden KL. Aberrant eukaryotic translation initiation factor 4E-dependent mRNA transport impedes hematopoietic differentiation and contributes to leukemogenesis. Mol Cell Biol. 2003;23:8992–9002. doi: 10.1128/MCB.23.24.8992-9002.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Inamdar KV, Romaguera JE, Drakos E, Knoblock RJ, Garcia M, Leventaki V, Medeiros LJ, Rassidakis GZ. Expression of eukaryotic initiation factor 4E predicts clinical outcome in patients with mantle cell lymphoma treated with hyper-CVAD and rituximab, alternating with rituximab, high-dose methotrexate, and cytarabine. Cancer. 2009;115:4727–4736. doi: 10.1002/cncr.24506. [DOI] [PubMed] [Google Scholar]
- 22.Ruggero D, Montanaro L, Ma L, Xu W, Londei P, Cordon-Cardo C, Pandolfi PP. The translation factor eIF-4E promotes tumor formation and cooperates with c-Myc in lymphomagenesis. Nat Med. 2004;10:484–486. doi: 10.1038/nm1042. [DOI] [PubMed] [Google Scholar]
- 23.Hagner PR, Schneider A, Gartenhaus RB. Targeting the translational machinery as a novel treatment strategy for hematologic malignancies. Blood. 2010;115:2127–2135. doi: 10.1182/blood-2009-09-220020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nathan CO, Liu L, Li BD, Abreo FW, Nandy I, De Benedetti A. Detection of the proto-oncogene eIF4E in surgical margins may predict recurrence in head and neck cancer. Oncogene. 1997;15:579–584. doi: 10.1038/sj.onc.1201216. [DOI] [PubMed] [Google Scholar]
- 25.Culjkovic B, Borden KL. Understanding and Targeting the Eukaryotic Translation Initiation Factor eIF4E in Head and Neck Cancer. J Oncol. 2009;2009:981679. doi: 10.1155/2009/981679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang B, Zhu C, Chen B, Zhang X, Ye M, Lin A. [Expression and its clinical significance of eIF4E in non-small cell lung cancer] Zhongguo Feiai Zazhi. 2010;13:1132–1135. doi: 10.3779/j.issn.1009-3419.2010.12.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Borden KL, Culjkovic-Kraljacic B. Ribavirin as an anti-cancer therapy: acute myeloid leukemia and beyond? Leuk Lymphoma. 2010;51:1805–1815. doi: 10.3109/10428194.2010.496506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Muta D, Makino K, Nakamura H, Yano S, Kudo M, Kuratsu J. Inhibition of eIF4E phosphorylation reduces cell growth and proliferation in primary central nervous system lymphoma cells. J Neurooncol. 2011;101:33–39. doi: 10.1007/s11060-010-0233-6. [DOI] [PubMed] [Google Scholar]
- 29.Assouline S, Culjkovic B, Cocolakis E, Rousseau C, Beslu N, Amri A, Caplan S, Leber B, Roy DC, Miller WH, et al. Molecular targeting of the oncogene eIF4E in acute myeloid leukemia (AML): a proof-of-principle clinical trial with ribavirin. Blood. 2009;114:257–260. doi: 10.1182/blood-2009-02-205153. [DOI] [PubMed] [Google Scholar]
- 30.Graff JR, Konicek BW, Carter JH, Marcusson EG. Targeting the eukaryotic translation initiation factor 4E for cancer therapy. Cancer Res. 2008;68:631–634. doi: 10.1158/0008-5472.CAN-07-5635. [DOI] [PubMed] [Google Scholar]
- 31.Hsieh AC, Ruggero D. Targeting eukaryotic translation initiation factor 4E (eIF4E) in cancer. Clin Cancer Res. 2010;16:4914–4920. doi: 10.1158/1078-0432.CCR-10-0433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Choi CH, Lee JS, Kim SR, Lee YY, Kim CJ, Lee JW, Kim TJ, Lee JH, Kim BG, Bae DS. Direct inhibition of eIF4E reduced cell growth in endometrial adenocarcinoma. J Cancer Res Clin Oncol. 2011;137:463–469. doi: 10.1007/s00432-010-0902-z. [DOI] [PubMed] [Google Scholar]
- 33.Liu L, Cao Y, Chen C, Zhang X, McNabola A, Wilkie D, Wilhelm S, Lynch M, Carter C. Sorafenib blocks the RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor cell apoptosis in hepatocellular carcinoma model PLC/PRF/5. Cancer Res. 2006;66:11851–11858. doi: 10.1158/0008-5472.CAN-06-1377. [DOI] [PubMed] [Google Scholar]
- 34.Huynh H, Ngo VC, Koong HN, Poon D, Choo SP, Thng CH, Chow P, Ong HS, Chung A, Soo KC. Sorafenib and rapamycin induce growth suppression in mouse models of hepatocellular carcinoma. J Cell Mol Med. 2009;13:2673–2683. doi: 10.1111/j.1582-4934.2009.00692.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Graff JR, Konicek BW, Vincent TM, Lynch RL, Monteith D, Weir SN, Schwier P, Capen A, Goode RL, Dowless MS, et al. Therapeutic suppression of translation initiation factor eIF4E expression reduces tumor growth without toxicity. J Clin Invest. 2007;117:2638–2648. doi: 10.1172/JCI32044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jiang Y, Zhang SH, Han GQ, Qin CY. Interaction of Pdcd4 with eIF4E inhibits the metastatic potential of hepatocellular carcinoma. Biomed Pharmacother. 2010;64:424–429. doi: 10.1016/j.biopha.2010.01.015. [DOI] [PubMed] [Google Scholar]


