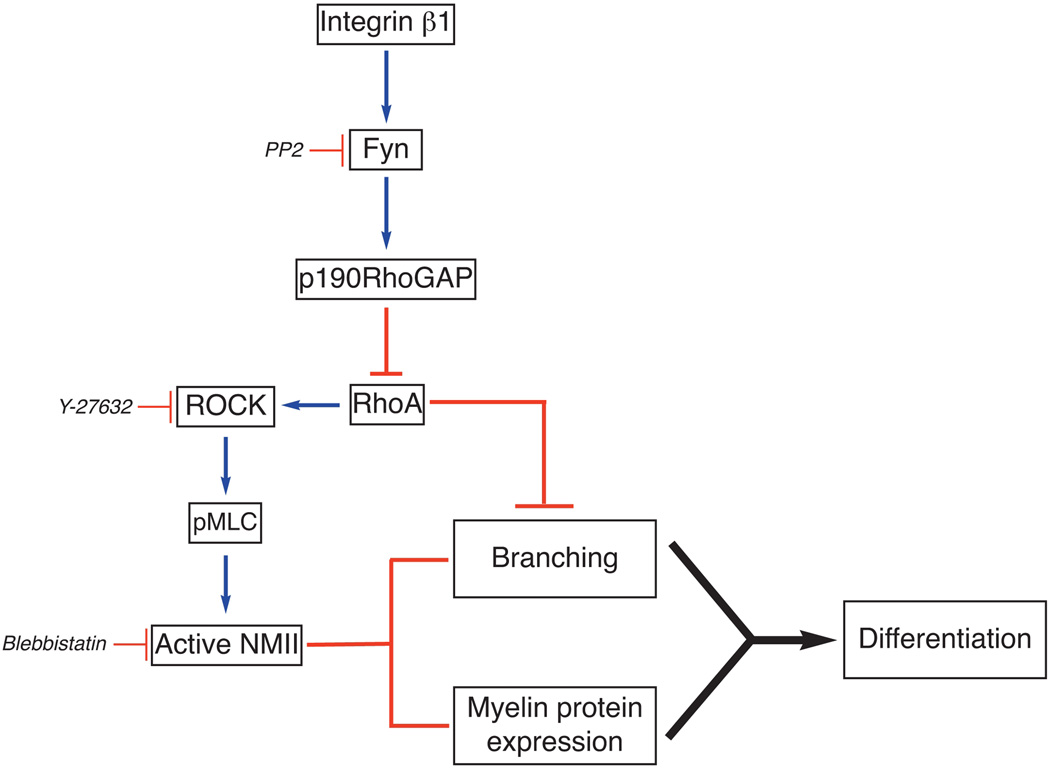
Figure 5. Model of Fyn-RhoA signaling and NMII inactivation in oligodendrocyte.
Engagement of integrins by their ECM ligands activates Fyn (Colognato et al. 2004). Fyn phosphorylates p190RhoGAP (Wolf et al. 2001), a GTPase activating factor, thus reducing the levels of active RhoA. Activation of ROCK downstream of RhoA promotes phosphorylation of MLC and activation of NMII (Somlyo and Somlyo 2000), which inhibits oligodendrocyte branching and differentiation. Inhibition of RhoA downstream of Fyn prevents NMII phosphorylation/activation by ROCK thus promoting process extension by oligodendrocytes. This model is supported by our data showing that inhibition of Fyn (PP2, siRNA) increases pMLC levels, while inhibition of ROCK (Y-27632) or NMII (blebbistatin, siRNA) reduces pMLC levels and promotes oligodendrocyte process extension. In the absence of NMII activity, expression of myelin protein and myelination are also enhanced. However, branching and myelin protein expression can be independently regulated.