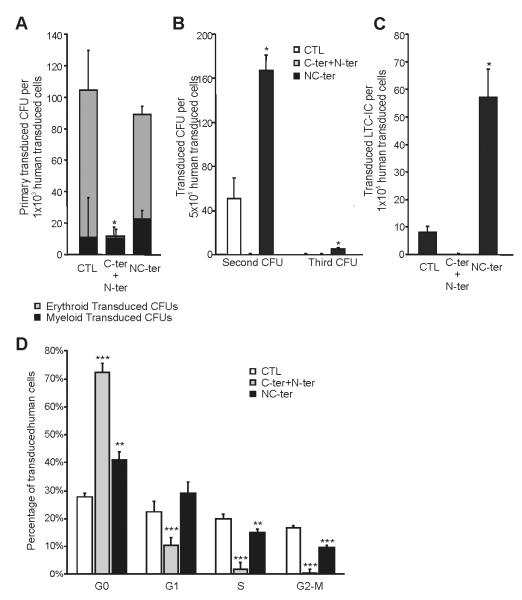
Figure 4. Biallelic and Double monoallelic mutations on C/EBPα modifies differently human progenitor compartment and Cell Cycle.
(A) Effect of C-terminal with N-terminal mutated C/EBPα (biallelic mutations) or NC-terminal mutated C/EBPα (double monoallelic mutation) in Human Hematopoietic Progenitors were determines by CFU assays. The number of erythroid (grey) and myeloid (black) CFUs in the tranduced cells are presented. (B) Serial replating of Control, N-ter+C-ter or NC-ter mutated C/EBP transduced progenitors are presented. (C) LTC-IC assay to evaluate effect in HSC after expressing C-ter+N-ter or NC-ter C/EBPα. (D) Cell Cycle profile of control, N-ter + C-ter or NC-ter expressing after 14 days of culture. Results are shown as mean ± standard deviation (SD), with significant differences of p ≤ 0.05, p ≤ 0.01 or p ≤ 0.001 are indicating by (*), (**) or (***) respectively. All the data are from a minimum of triplicates of two independent experiments.