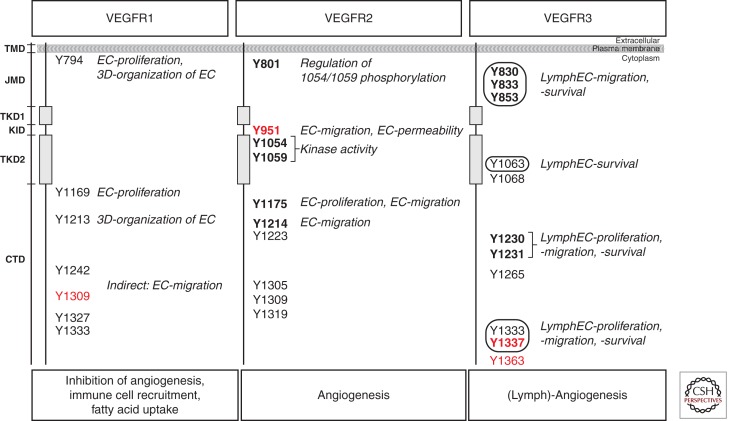
Figure 2.
Tyrosine phosphorylation sites in VEGFRs. Intracellular domains of VEGFR monomers are shown schematically with phosphorylated tyrosine residues indicated as Y followed by a number indicating the position in the amino acid sequence of the receptor. Red numbers indicate selective phosphorylation, i.e., Y1309 in VEGFR1 is phosphorylated on PlGF-, but not VEGF-binding, Y951 in VEGFR2 is phosphorylated in endothelial cells without pericyte contact, and Y1337/Y1363 are phosphorylated in VEGFR3 homodimers, but not in VEGFR2/-3 heterodimers. Tyrosines with identified biological function are bold; tyrosines without known biological function are regular. Encircled residues in VEGFR3 show tyrosines, phosphorylated by extracellular matrix independent of ligand (see text for details and references). Functions are indicated for respective tyrosines in italic. TMD, Transmembrane domain; JMD, juxtamembrane domain; TKD1, ATP binding domain; KID, kinase insert domain; TKD2, phosphotransferase domain; CTD, carboxy-terminal domain; EC, endothelial cell.