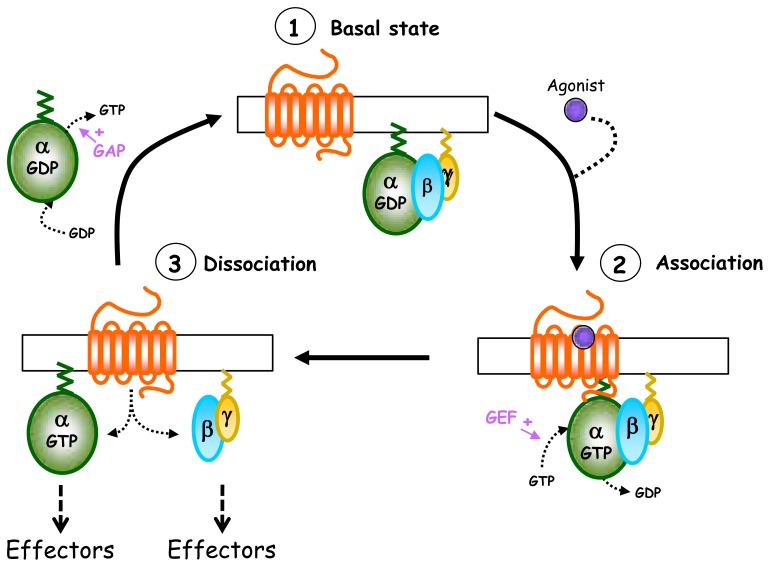
Fig. 2.
Heterotrimeric G protein activation cycle. In the absence of agonist (1, basal state), GαGDP-βγ heterotrimeric G protein forms a tight inactive complex dissociated from the receptor. The activation of the receptor by the agonist promotes recruitment of Gαβγ to the receptor and the subsequent GDP/GTP exchange at the level of the Gα subunit (2, association). This nucleotide exchange then leads to the dissociation of the receptor and also of the Gα-GTP and Gβγ subunits, which are now able to activate their effectors (3, dissociation). The activation cycle is terminated by the Gα intrinsic GTPase activity which allows GTP hydrolysis and the reassociation of Gα-GDP with Gβγ subunits so to restore the inactive basal state (1).