Abstract
Acquisition of anoikis resistance is a prerequisite for the metastasis of hepatocellular carcinoma (HCC) cells. Activation of growth factor signaling pathways and rearrangement of the cytoskeleton have been reported as vital steps in this process. However, key molecules involved in anoikis resistance remain to be determined. The aim of this study was to investigate the effect of CD147 on HCC cells resistant to anoikis. The human SMMC-7221 human HCC cell line was used. Immunofluorescence was used to investigate the expression levels of CD147. Anoikis-induced cell death was assessed using trypan blue exclusion. In the present study, the results showed that SMMC-7721 HCC cells exhibited significant morphological changes when suspended in culture medium supplemented with 1% methocel and a subpopulation of cells resistant to anoikis was acquired with higher viability and invasion ability. CD147 was identified to be significantly increased in cells resistant to anoikis, when compared to the parental cells. CD147 knockdown by siRNA notably induced cell anoikis, partially through the inactivation of PI3K/Akt pathway. All of these evidence provide a novel CD147-related mechanism underlying the metastasis of HCC cells.
Keywords: anoikis resistance, CD147, PI3K/Akt, hepatocellular carcinoma metastasis
Introduction
Metastasis is a multi-step biological process and becomes the primary cause of cancer-related mortality in most solid tumors. Therefore, preventing the metastatic spread of cancer cells is a principal task in cancer therapy. During the metastatic process, cells were required to detach from their primary site, and then to migrate to the lymphatic and circulatory systems (1). In metastasis, the majority of cells undergo anoikis, which is induced by the detachment of anchorage-dependent cells from the surrounding extracellular matrix (2). Acquisition of anoikis-resistance is therefore a prerequisite for the metastasis of cancer cells. Recently, several reports have confirmed that the breakdown of anoikis contributes prominently to the malignancy of breast (3), colon (4), lung (5) and head and neck carcinomas (6). However, few studies have focused on the role of anoikis-resistance in hepatocellular carcinoma (HCC) metastasis.
In recent years, accumulating studies indicate that anoikis is mainly mediated by the death receptor pathway activated by caspase (7,8). This process occurs in hepatocytes (9). Activation of growth factor signaling pathways, which commonly occurs in carcinoma cells, enables cells to acquire anoikis resistance (10,11). Numerous kinase/phosphatase signaling molecules are known to be involved in anoikis as central regulators, such as Ras (12), Akt and raf (13). Other reports have also confirmed that cell cytoskeletal rearrangement is critical for the cells to survive anoikis resistance (14,15). Moreover, this process is especially associated with cadherin family members, which are the key elements in the complex network of survival signaling (16).
CD147, a member of the immunoglobulin superfamily, was initially characterized as an inducer of matrix metalloproteinase (MMP) synthesis (17). CD147 is known to play a vital role in tumor progression. High expression levels of CD147 in epithelial carcinoma tissue are significantly associated with poor prognosis (18,19). Recently, CD147 has been found to promote epithelial-mesenchymal transition (EMT) through transforming growth factor (TGF)-β signaling (20) and serve as an anoikis suppressor by the inhibition of Bim, a pro-apoptotic protein interacting with Bcl-2 to mediate cell death in breast cancer cells (21). In addition, a radio-immunoconjugate 131I-labeled antibody HAb18 F(ab’)2 against CD147, registered as Licartin, was used to treat primary HCC and prevent tumor recurrence of post-orthotopic liver transplantation in advanced HCC patients (22). However, the role of CD147-induced anoikis resistance in HCC metastasis remains poorly elucidated.
The aim of the present study was to investigate the upregulation of CD147-conferred HCC cell resistance to anoikis partially through activation of the phosphoinositide-3 kinase (PI3K)/Akt pathway, thereby providing a novel mechanism supporting HCC metastasis.
Materials and methods
Cell culture and anoikis assay
The SMMC-7721 human HCC cell line was purchased from the Shanghai Institute for Biological Sciences (Shanghai, China) and routinely cultured in RPMI-1640 medium (Hyclone Laboratories, Logan, UT, USA) supplemented with 10% fetal bovine serum (Gibco, Rockville, MD, USA). HEK293ar cells were obtained as previously reported (23). The anoikis assay was essentially performed as described by Frisch (24). Briefly, flasks were pre-coated with 2% sterilized agar. Cells were trypsinized and plated onto pre-coated flasks. Suspension medium consisted of RPMI-1640 supplemented with 1% methocel and 10% FBS. According to the process of anoikis adaptation for HEK293ar cells (23), a subpopulation of SMMC-7721 cells resistant to anoikis was obtained. Cultures were maintained routinely at 37°C under a mixture of 95% air and 5% CO2.
Immunofluorescence
SMMC-7721 cells were suspended in methocel medium for the indicated times. Cells were harvested and dried on coverslips. Double-staining of Hoechst 33342 and propidium iodide (PI) was developed to evaluate the phenotype and viability of treated cells. To examine the expression levels of CD147 in treated cells, the cells were firstly fixed and blocked, and then incubated with anti-CD147 monoclonal antibody (1:200) (25). After washing, the cell samples were incubated with FITC-conjugated goat anti-mouse IgG antibody (1:1,000; Pierce, Rockford, IL, USA) and the nuclei were stained with DAPI (1:100; Biotium, Hayward, CA, USA). The stained cells were examined with a laser scanning microscope (Olympus, Tokyo, Japan).
Antibodies and immunoblot analysis
For the immunoblot analysis, cells were lysed in RIPA buffer (Beyotime Inc., NanTong, China). Protein concentration was determined with the Bradford reagent (Beyotime Inc.). Equal amounts of total proteins were separated and transferred to polyvinylidene difluoride membrane (Millipore, Bedford, MA, USA). The membranes were subsequently immunoblotted with the appropriate primary antibody. The following primary antibodies were used in this study: anti-CD147 antibody (25), anti-Akt and anti-phosphorylated Akt (ser 473) antibody from Cell Signaling Technology (Beverly, MA, USA). A secondary horseradish peroxidase-conjugated goat anti-mouse antibody (Pierce) was finally used for signal detection with an ECL kit (Pierce) according to the manufacturer’s instructions.
Trypan blue exclusion assay
The anoikis-induced cell death was evaluated by trypan blue exclusion assay as previously described (26). In brief, the parental SMMC-7721 and SMMC-7721 cells resistant to anoikis were suspended in methocel medium for the indicated times. Cells were harvested, washed three times with PBS and resuspended in 100 μl PBS. After mixing with 100 μl of 0.8% trypan blue, the cells were counted using a hemocytometer.
In vitro invasion assay
The migration ability was measured by in vitro invasion assay as previously reported (27). The parental SMMC-7721 and SMMC-7721 cells resistant to anoikis were suspended in methocel medium for 48 h, resuspended in serum-free DMEM medium containing 0.5% FBS and then added to the upper compartment of a chamber at a total number of 2×105 cells per chamber. After incubation for 24 h, the number of cells migrating through the filter was counted by crystal violet staining and plotted as the mean number of migrating cells per optic field in three independent experiments.
siRNA transfection
siRNA sequences corresponding to the cDNA sequences of CD147 (Genebank accession NM_001728) were designed (5’-GGUUCUUCGUGAGUUCCUCdTdT-3’ and 5’-GAGGAACUCACGAAGAACCdTdT-3’). Briefly, the subpopulation of SMMC-7721 cells resistant to anoikis was transfected with CD147 siRNA (100 nM) using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). Scrambled negative control (SNC) siRNA was used as mock control. Transiently-transfected cells were recovered for 24 h and re-plated on agar-coated 6-well plates.
Apoptosis assay
SMMC-7721 cells resistant to anoikis transfected with CD147 siRNA or control siRNA were suspended in fresh methocel medium in an agar-coated plate for 24 h. Cells were harvested and treated with 10 mmol/l EDTA to disrupt the cell-cell contacts. The cells were then analyzed by double-staining with Annexin V-FITC and PI using an Apoptosis Detection kit (Calbiochem, San Diego, CA, USA) followed by detection using a FACSCalibur flow cytometer. Early apoptotic cells were labeled as Annexin+/PI- and necrotic cells were labeled as PI+. The percentages of cells at early apoptosis or necrotic stage were analyzed at least three times.
Statistical analysis
Statistical analyses were performed using the SPSS 16.0 statistical software package (SPSS Inc., Chicago, IL, USA). Statistical significance of the differences was determined using Student’s t-test. All P-values were based on two-sided tests. P<0.05 was considered statistically significant.
Results
Anoikis of suspended SMMC-7721 cells and formation of multi-cellular spheroids
To characterize the morphological change of SMMC-7721 cells after suspension in methocel medium, typical images representing the morphology of cells after treatment for 0, 24 and 48 h are shown in Fig. 1A. Cell-cell contacts were spontaneously and gradually formed and multi-cellular spheroids were generated in a time-dependent manner. The volume of the aggregated spheroids was significantly increased at the time point of 48 h as indicated in Fig. 1B. The viability of the SMMC-7721 cells was further analyzed by double-staining of Hoechst 33342 and PI under a laser microscope. At 0 h, the staining assay indicated that the treated cells were alive (exclusion of PI). After cells were cultured for 24 h, anoikis with positive staining of PI was induced in some of the cells and apoptotic bodies were also found along the cytoplasmic membrane. Multi-cellular spheroids were found at the time-point of 48 h, when the majority of cells underwent anoikis with a strong staining of PI and a weak staining of Hoechst 33342 (Fig. 3C).
Figure 1.
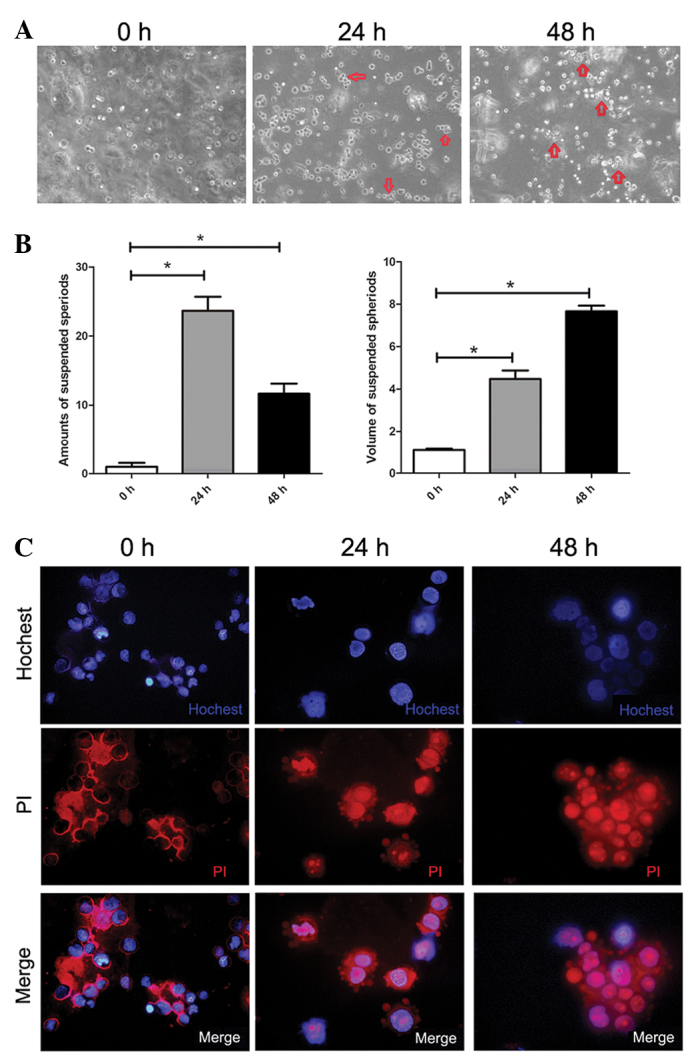
Anoikis of suspended SMMC-7721 cells and formation of multi-cellular spheroids. (A) Morphology of SMMC-7721 cells in agar suspension culture at indicated time-points (magnification, ×100). (B) Characterization of the suspended spheroids during suspension culture of SMMC-7721 cells. The amount of spheroids (3 cells/spheroid) per focus was counted under a phase-contrast microscope. The volume of spheroids referred to the average number of cells per multi-cellular spheroids (magnification, ×100; n=8–10). Each value is the mean ± SD of at least triplicate determinations. (C) Double staining of Hoechst 33342 and PI SMMC-7721 cells after suspension at different time-points by immunofluorescence under a laser scanning microscope (magnification, ×400).
Figure 3.
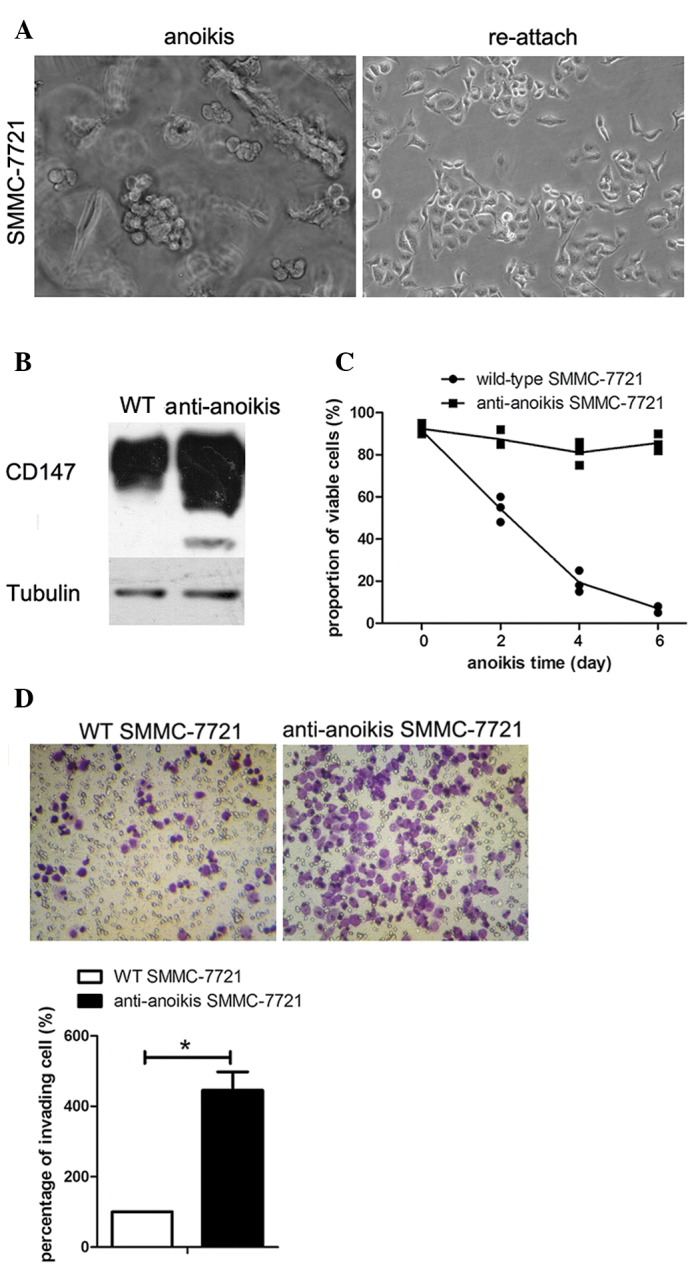
CD147 was significantly upregulated in anoikis-resistant SMMC-7721 cells with an increase in both viability and invasion ability. (A) The process of suspension culture adaptation under a phase-contrast microscope (magnification, ×200). The left panel shows the morphology of SMMC-7721 cells when suspended in methocel medium. The right panel shows the morphology of SMMC-7721 cells following reattachment to the extracellular matrix. The subpopulation of anoikis-resistant SMMC-7721 cells was obtained by sequential cycles of suspension and adhesion culture. (B) Findings of the immunoblot assay with regard to the expression levels of CD147 in wild-type SMMC-7721 cells and the subpopulation of SMMC-7721 cells resistant to anoikis. Tubulin was used as a loading control. (C) The roportion of viable cells under suspension culture of wild-type SMMC-7721 and SMMC-7721 cells resistant to anoikis at the indicated time-points were detected by trypan blue exclusion assay. Data are the means ± SD for eight replicates. (D) Images of invading SMMC-7721 or anoikis-resistant SMMC-7721 cells in a transwell invasion assay after suspension for 48 h, and column graph for three independent experiments. Data were normalized to wild-type SMMC-7721 cells and are the means ± SD (*p<0.05).
CD147 was upregulated on the cell-cell contacts in suspended multi-cellular spheroids
To explore the role of CD147 in anoikis resistance, we examined the expression levels of CD147 in HCC cells. Western blotting showed that SMMC-7721 cells in suspended culture exhibited a notable time-dependent accumulation of the CD147 protein (Fig. 2A). Immunofluorescence staining confirmed the elevated levels of CD147 in SMMC-7721 cells after suspension for 48 h (Fig. 2B). Additionally, in a large multi-cellular spheroid, the strong CD147 staining areas were restricted to the cell-cell contacts for both SMMC-7721 and HEK293ar cells as indicated by the yellow arrows in Fig. 2B.
Figure 2.
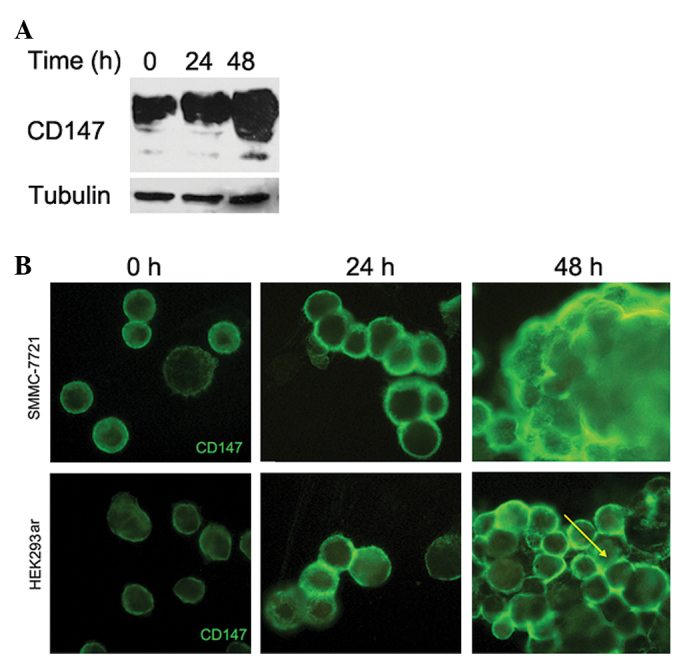
CD147 was upregulated on the cell-cell contacts in suspended multi-cellular spheroids. (A) Western blotting of CD147 expression in SMMC-7721 cells following suspension at the indicated time-points. Two major forms of CD147 (45–66 and 35 kDa) were analyzed. Tubulin was used as a loading control. (B) Expression levels of CD147 in SMMC-7721 and HEK293ar cells following suspension at the indicated time-points as revealed by immunofluorescence under a laser scanning microscope (n=6-10; magnification, ×400). Yellow arrow points to the enhanced expression levels of CD147 between cell-cell contacts in a multi-cellular spheroid.
CD147 was significantly upregulated in anoikis-resistant SMMC-7721 cells with an increase in both viability and invasion ability
Based on a previous study (23), a subpopulation of anoikis-resistant SMMC-7721 cells was obtained to explore the correlation between CD147 expression levels and anoikis resistance. Morphological changes occurred in cells during the adaptation period for SMMC-7721 cells in suspension culture (Fig. 3A). Western blotting indicated that CD147 was significantly upregulated in the anoikis-resistant SMMC-7721 cells, compared to the parental SMMC-7721 cells (Fig. 3B). The trypan blue exclusion assay demonstrated that a high percentage of the anoikis-resistant SMMC-7721 cells remained alive following suspension in culture medium for 6 days, while almost all the parental SMMC-7721 cells underwent death (Fig. 3C). In vitro invasion assay showed that the invasion ability was significantly increased in SMMC-7721 cells resistant to anoikis, compared to the parental SMMC-7721 cells (Fig. 3D).
Anoikis was induced by the knockdown of CD147 expression in the subpopulation of SMMC-7721 cells resistant to anoikis through inactivation of the PI3K/Akt pathway
To prove the direct involvement of CD147 during the adaptation process of anoikis resistance, we knocked down the expression levels of CD147 in the subpopulation of anoikis-resistant SMMC-7721 cells through transfection with specific small interfering RNA (siRNA). Immunofluorescence assay results indicated that the staining intensity of CD147 in the area of cell-cell contacts was significantly reduced in anoikis-resistant SMMC-7721 cells tansfected with CD147 siRNA, compared to those transfected with SNC siRNA (Fig. 4A). Similarly, the cell nuclei tended to be cracked in cells transfected with CD147 siRNA following suspension in methocel medium for 24 h as indicated by the white arrows (Fig. 4A). Flow cytometry showed that the inhibition of CD147 expression significantly induced anoikis (both early apoptosis and necrosis) in anoikis-resistant SMMC-7721 cells (Fig. 4B). To elucidate the possible signaling pathway involved in the CD147-mediated anoikis resistance, western blot analysis was performed to examine the expression levels of CD147, phosphorylated Akt and Akt in anoikis-resistant SMMC-7721 cells transfected with CD147 siRNA and SNC siRNA, respectively. We found that the knockdown of CD147 resulted in a significant decrease of phosphorylated Akt levels, whereas the expression of Akt was not affected (Fig. 4C). These data suggest that activation of the PI3K-Akt signaling pathway may be involved in CD147-mediated anoikis resistance.
Figure 4.
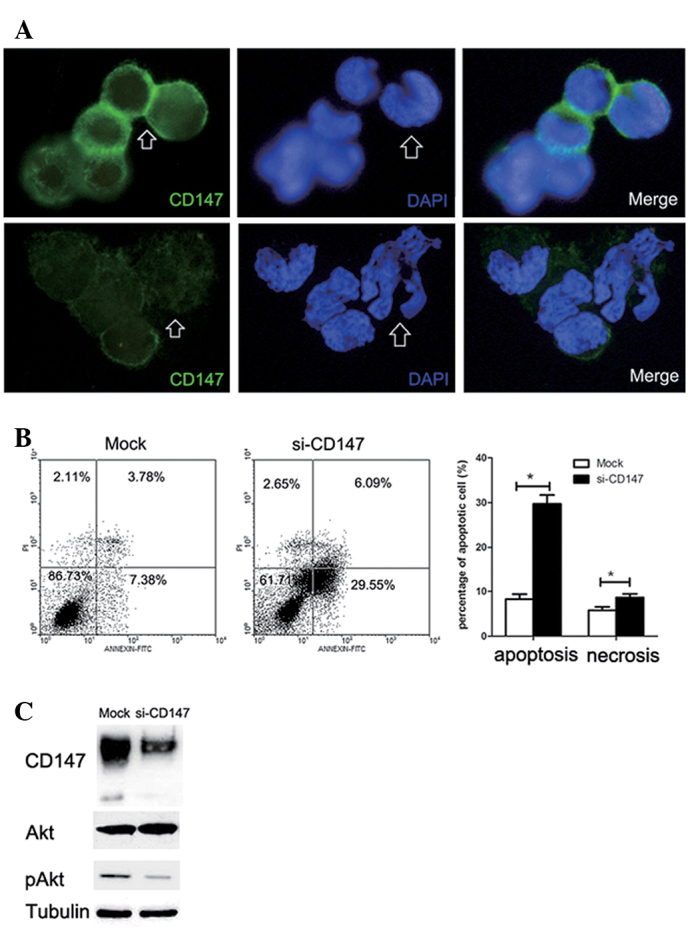
Anoikis was induced by knocking down CD147 expression in the subpopulation of SMMC-7721 cells resistant to anoikis by inactivating the PI3K/Akt pathway. (A) Immunofluorescence of CD147 in SMMC-7721 cells resistant to anoikis transfected with CD147 siRNA and control siRNA (n=6–10; magnification, ×400). Arrows point to the different morphologies of cell-cell contacts and nuclei after the inhibition of CD147. (B) Flow cytometry for apoptosis and necrosis of anoikis-resistant SMMC-7721 cells through double staining of Annexin V-FITC and PI after culture was suspended for 24 h. Each value is the means ± SD of at least triplicate determinations. (C) Western blotting was performed to examine the expression levels of CD147, phosphorylated Akt and Akt in the subpopulation of SMMC-7721 cells resistant to anoikis transfected with CD147 siRNA and control siRNA.
Discussion
This study provides evidence that an elevated endogenous expression of CD147 contributes to anoikis resistance in SMMC-7721 cells partially through activation of the PI3K/Akt survival pathway, which has been considered an important process for the metastasis of HCC cells.
We first established a model for cell suspension culture to characterize the morphology of SMMC-7721 cells following suspension in methocel medium. A number of studies have validated that anoikis can be inhibited despite loss of cell-matrix adhesion when cell-cell adhesion is preserved among various epithelial carcinoma cell lines (16,28,29). Our research model also revealed a multi-cellular spheroid formation in a time-dependent manner. Furthermore, we observed a significant increase of CD147 expression in SMMC-7721 cells under suspension culture, especially restricted to the region along the cytoplasmic membrane of cell-cell contacts, which indicates a potential role of CD147 in mediating the formation of multi-cellular spheroids. Earlier evidence has shown that CD147 promotes breast cancer cell survival by regulating intercellular contacts and inhibiting anoikis through BIM (21). Besides, our previous study has verified a vital role of CD147 involved in cell-cell contacts in an E-cadherin-dependent manner in HEK293 cells (23). These data suggest that CD147 may also be involved in anoikis resistance in HCC when cells are exposed to a condition of extracellular matrix-detached culture.
The direct role of CD147 in the adaptation of anoikis resistance was investigated. The SMMC-7721 cell line has been extensively studied in the anoikis resistance of HCC (30,31). Therefore, we established an anoikis-resistant cell model (SMMC-7721 cells resistant to anoikis) to investigate the mechanism of anoikis suppression. The adaptation process in this cell model is closer to the natural in vivo status of the physiological conditions compared to earlier research models that directly modified the expression levels of target gene by manual intervention (21). The significantly high expression levels of CD147 led to anoikis-resistant SMMC-7721 cells exhibiting a notably higher viability and invasion ability compared to their parental SMMC-7721 cells. To provide direct evidence that CD147 is involved in the process of anoikis resistance in the subpopulation of SMMC-7721 cells resistant to anoikis, we inhibited CD147 through transfection of specific targeting siRNA in anoikis-resistant SMMC-7721 cells. The proportion of the apoptotic and necrotic cells was greatly increased compared to the mock control after the cells were suspended in methocel medium for 24 h. Accordingly, the morphology of nuclei tended to split after the inhibition of CD147. These results strongly suggest that the upregulation of CD147 is important in rendering cell anoikis resistance, which is a premise for the metastasis of primary cells.
The present study also provided a potential molecular mechanism supporting the hypothesis that the upregulation of CD147 induces anoikis resistance in SMMC-7721 cells. A number of published observations have demonstrated the principal role of PI3K/Akt pathway activation in anoikis resistance (32–34). Activation of PI3K/Akt is essential for the integrity of adherent junctions and is associated with the rearrangement of the cytoskeleton (35). Our previous studies have demonstrated a vital role of CD147 in the activation of the PI3K/Akt pathway to protect cell survival under the different stress exposition (26,36). Therefore, we explored the correlation between the upregulation of CD147 expression and activation of the PI3K/Akt pathway in our anoikis-resistant cell model. Our results have shown that the knockdown of CD147 significantly decreased the levels of pAkt, suggesting that activation of the PI3K/Akt pathway is involved in anoikis resistance by the upregulation of CD147. However, our preliminary data only provide a potential molecular mechanism of CD147-induced anoikis resistance in HCC cells. The possible involvement of other signaling molecules cannot be excluded. Moreover, a thorough evaluation of other related signaling pathways would shed light on the mechanisms of CD147-mediated anoikis resistant regulation.
In conclusion, the upregulation of CD147 significantly contributes to anoikis resistance in SMMC-7721 cells following suspension in methocel medium. Spontaneous upregulation of the expression levels of CD147 in SMMC-7721 cells resistant to anoikis may promote the cell-cell contacts and then activate the PI3K/Akt pathway. Thus, our findings provide a new mechanism underlying the metastasis of HCC cells, which emphasizes an important role of CD147 in anoikis resistance regulation (Fig. 5).
Figure 5.
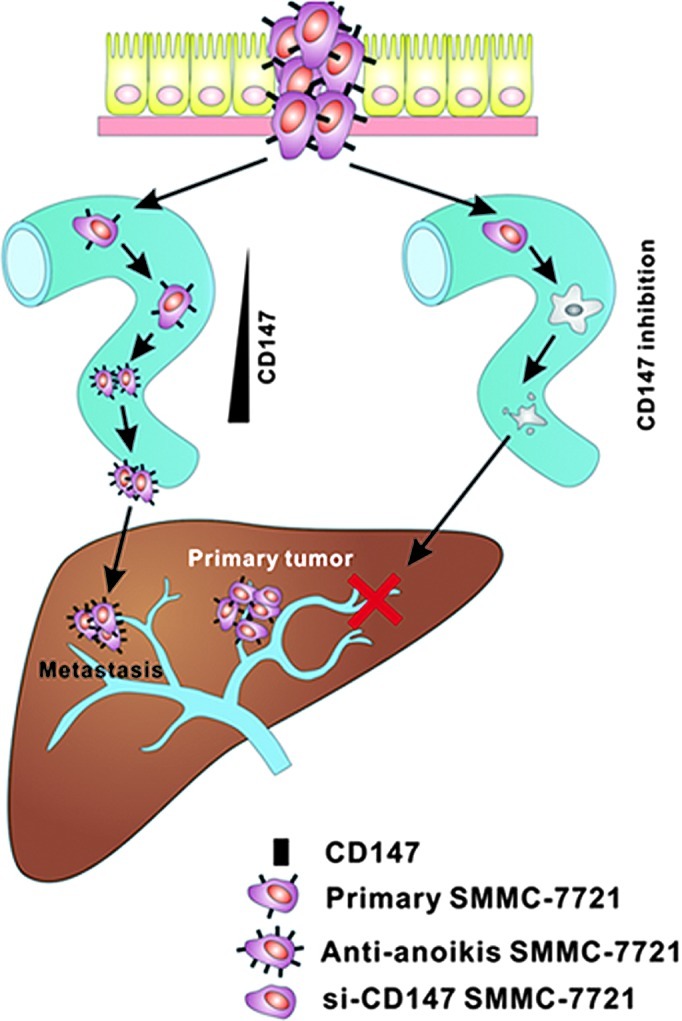
Schematic image of the underlying mechanism of the upregulation of CD147 for anoikis resistance in HCC metastasis. Enhanced expression of CD147 enables HCC cells to form multi-cellular spheroids and to activate the PI3K/Akt pathway, protecting cells from anoikis through a synoikis-like survival style. Thus, metastasis from the primary tumor was eventually induced in the liver. In comparison, during the process of metastatic circulation, the cell-cell contacts were destroyed when CD147 was inhibited and the disassociated cells ultimately underwent anoikis. HCC metastasis was successfully prevented.
Acknowledgements
The authors are thankful to Mr. Kai Qu for his technical assistance.
Abbreviations
- HCC
hepatocellular carcinoma
- EMT
epithelial-mesenchymal transition
- PI3K
phosphoinositide-3 kinase
- PI
propidium iodide
- siRNA
small interfering RNA
- SNC
scrambled negative control
References
- 1.Simpson CD, Anyiwe K, Schimmer AD. Anoikis resistance and tumor metastasis. Cancer Lett. 2008;272:177–185. doi: 10.1016/j.canlet.2008.05.029. [DOI] [PubMed] [Google Scholar]
- 2.Frisch SM, Francis H. Disruption of epithelial cell-matrix interactions induces apoptosis. J Cell Biol. 1994;124:619–626. doi: 10.1083/jcb.124.4.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Simpson CD, Mawji IA, Anyiwe K, et al. Inhibition of the sodium potassium adenosine triphosphatase pump sensitizes cancer cells to anoikis and prevents distant tumor formation. Cancer Res. 2009;69:2739–2747. doi: 10.1158/0008-5472.CAN-08-2530. [DOI] [PubMed] [Google Scholar]
- 4.Taddei ML, Parri M, Angelucci A, et al. EphA2 induces metastatic growth regulating amoeboid motility and clonogenic potential in prostate carcinoma cells. Mol Cancer Res. 2011;9:149–160. doi: 10.1158/1541-7786.MCR-10-0298. [DOI] [PubMed] [Google Scholar]
- 5.Pongrakhananon V, Nimmannit U, Luanpitpong S, Rojanasakul Y, Chanvorachote P. Curcumin sensitizes non-small cell lung cancer cell anoikis through reactive oxygen species-mediated Bcl-2 downregulation. Apoptosis. 2010;15:574–585. doi: 10.1007/s10495-010-0461-4. [DOI] [PubMed] [Google Scholar]
- 6.Kumar P, Yadav A, Patel SN, et al. Tetrathiomolybdate inhibits head and neck cancer metastasis by decreasing tumor cell motility, invasiveness and by promoting tumor cell anoikis. Mol Cancer. 2010;9:206. doi: 10.1186/1476-4598-9-206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Aoudjit F, Vuori K. Matrix attachment regulates Fas-induced apoptosis in endothelial cells: a role for c-flip and implications for anoikis. J Cell Biol. 2001;152:633–643. doi: 10.1083/jcb.152.3.633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mawji IA, Simpson CD, Hurren R, et al. Critical role for Fas-associated death domain-like interleukin-1-converting enzyme-like inhibitory protein in anoikis resistance and distant tumor formation. J Natl Cancer Inst. 2007;99:811–822. doi: 10.1093/jnci/djk182. [DOI] [PubMed] [Google Scholar]
- 9.Smets FN, Chen Y, Wang LJ, Soriano HE. Loss of cell anchorage triggers apoptosis (anoikis) in primary mouse hepatocytes. Mol Genet Metab. 2002;75:344–352. doi: 10.1016/S1096-7192(02)00004-5. [DOI] [PubMed] [Google Scholar]
- 10.Attwell S, Roskelley C, Dedhar S. The integrin-linked kinase (ILK) suppresses anoikis. Oncogene. 2000;19:3811–3815. doi: 10.1038/sj.onc.1203711. [DOI] [PubMed] [Google Scholar]
- 11.Terada LS, Nwariaku FE. Escaping anoikis through ROS: ANGPTL4 controls integrin signaling through Nox1. Cancer Cell. 2011;19:297–299. doi: 10.1016/j.ccr.2011.02.019. [DOI] [PubMed] [Google Scholar]
- 12.Rosen K, Rak J, Leung T, Dean NM, Kerbel RS, Filmus J. Activated Ras prevents downregulation of Bcl-X(L) triggered by detachment from the extracellular matrix. A mechanism of Ras-induced resistance to anoikis in intestinal epithelial cells. J Cell Biol. 2000;149:447–456. doi: 10.1083/jcb.149.2.447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Boisvert-Adamo K, Aplin AE. B-RAF and PI-3 kinase signaling protect melanoma cells from anoikis. Oncogene. 2006;25:4848–4856. doi: 10.1038/sj.onc.1209493. [DOI] [PubMed] [Google Scholar]
- 14.Frisch SM, Vuori K, Ruoslahti E, Chan-Hui PY. Control of adhesion-dependent cell survival by focal adhesion kinase. J Cell Biol. 1996;134:793–799. doi: 10.1083/jcb.134.3.793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhao J, Zhang Y, Ithychanda SS, et al. Migfilin interacts with Src and contributes to cell-matrix adhesion-mediated survival signaling. J Biol Chem. 2009;284:34308–34320. doi: 10.1074/jbc.M109.045021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kang HG, Jenabi JM, Zhang J, et al. E-cadherin cell-cell adhesion in ewing tumor cells mediates suppression of anoikis through activation of the ErbB4 tyrosine kinase. Cancer Res. 2007;67:3094–3105. doi: 10.1158/0008-5472.CAN-06-3259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Caudroy S, Polette M, Tournier JM, et al. Expression of the extracellular matrix metalloproteinase inducer (EMMPRIN) and the matrix metalloproteinase-2 in bronchopulmonary and breast lesions. J Histochem Cytochem. 1999;47:1575–1580. doi: 10.1177/002215549904701209. [DOI] [PubMed] [Google Scholar]
- 18.Pinheiro C, Longatto-Filho A, Simoes K, et al. The prognostic value of CD147/EMMPRIN is associated with monocarboxylate transporter 1 co-expression in gastric cancer. Eur J Cancer. 2009;45:2418–2424. doi: 10.1016/j.ejca.2009.06.018. [DOI] [PubMed] [Google Scholar]
- 19.Guo H, Li R, Zucker S, Toole BP. EMMPRIN (CD147), an inducer of matrix metalloproteinase synthesis, also binds interstitial collagenase to the tumor cell surface. Cancer Res. 2000;60:888–891. [PubMed] [Google Scholar]
- 20.Wu J, Ru NY, Zhang Y, et al. HAb18G/CD147 promotes epithelial-mesenchymal transition through TGF-beta signaling and is transcriptionally regulated by Slug. Oncogene. 2011;30:4410–4427. doi: 10.1038/onc.2011.149. [DOI] [PubMed] [Google Scholar]
- 21.Yang JM, O’Neill P, Jin W, et al. Extracellular matrix metalloproteinase inducer (CD147) confers resistance of breast cancer cells to Anoikis through inhibition of Bim. J Biol Chem. 2006;281:9719–9727. doi: 10.1074/jbc.M508421200. [DOI] [PubMed] [Google Scholar]
- 22.Xu J, Shen ZY, Chen XG, et al. A randomized controlled trial of Licartin for preventing hepatoma recurrence after liver transplantation. Hepatology. 2007;45:269–276. doi: 10.1002/hep.21465. [DOI] [PubMed] [Google Scholar]
- 23.Ma XK, Wang L, Li Y, et al. HAb18G/CD147 cell-cell contacts confer resistance of a HEK293 subpopulation to anoikis in an E-cadherin-dependent manner. BMC Cell Biol. 2010;11:27. doi: 10.1186/1471-2121-11-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Frisch SM. Evidence for a function of death-receptor-related, death-domain-containing proteins in anoikis. Curr Biol. 1999;9:1047–1049. doi: 10.1016/s0960-9822(99)80455-2. [DOI] [PubMed] [Google Scholar]
- 25.Chen ZN. Significance and application of anti-malignant hepatoma MAb HAb18 in radioimmunal diagnosis of human hepatocellular carcinoma. Zhonghua Zhong Liu Za Zhi. 1992;14:9–12. (In Chinese) [PubMed] [Google Scholar]
- 26.Gou X, Ru Q, Zhang H, et al. HAb18G/CD147 inhibits starvation-induced autophagy in human hepatoma cell SMMC7721 with an involvement of Beclin 1 down-regulation. Cancer Sci. 2009;100:837–843. doi: 10.1111/j.1349-7006.2009.01113.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Xu J, Xu HY, Zhang Q, et al. HAb18G/CD147 functions in invasion and metastasis of hepatocellular carcinoma. Mol Cancer Res. 2007;5:605–614. doi: 10.1158/1541-7786.MCR-06-0286. [DOI] [PubMed] [Google Scholar]
- 28.Derksen PW, Braumuller TM, van der Burg E, et al. Mammary-specific inactivation of E-cadherin and p53 impairs functional gland development and leads to pleomorphic invasive lobular carcinoma in mice. Dis Model Mech. 2011;4:347–358. doi: 10.1242/dmm.006395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Singh AB, Sugimoto K, Harris RC. Juxtacrine activation of epidermal growth factor (EGF) receptor by membrane-anchored heparin-binding EGF-like growth factor protects epithelial cells from anoikis while maintaining an epithelial phenotype. J Biol Chem. 2007;282:32890–32901. doi: 10.1074/jbc.M702677200. [DOI] [PubMed] [Google Scholar]
- 30.Wu Y, Zuo J, Ji G, et al. Proapoptotic function of integrin beta(3) in human hepatocellular carcinoma cells. Clin Cancer Res. 2009;15:60–69. doi: 10.1158/1078-0432.CCR-08-1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Feng Y, Wang LY, Cai T, et al. All-trans-retinoic acid increased the expression of integrin alpha5beta1 and induced ‘anoikis’ in SMMC-7721 hepatocarcinoma cell. J Exp Clin Cancer Res. 2001;20:429–438. [PubMed] [Google Scholar]
- 32.McFall A, Ulku A, Lambert QT, Kusa A, Rogers-Graham K, Der CJ. Oncogenic Ras blocks anoikis by activation of a novel effector pathway independent of phosphatidylinositol 3-kinase. Mol Cell Biol. 2001;21:5488–5499. doi: 10.1128/MCB.21.16.5488-5499.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Isakoff SJ, Engelman JA, Irie HY, et al. Breast cancer-associated PIK3CA mutations are oncogenic in mammary epithelial cells. Cancer Res. 2005;65:10992–11000. doi: 10.1158/0008-5472.CAN-05-2612. [DOI] [PubMed] [Google Scholar]
- 34.Jiang K, Sun J, Cheng J, Djeu JY, Wei S, Sebti S. Akt mediates Ras downregulation of RhoB, a suppressor of transformation, invasion, and metastasis. Mol Cell Biol. 2004;24:5565–5576. doi: 10.1128/MCB.24.12.5565-5576.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Frisch SM, Ruoslahti E. Integrins and anoikis. Curr Opin Cell Biol. 1997;9:701–706. doi: 10.1016/s0955-0674(97)80124-x. [DOI] [PubMed] [Google Scholar]
- 36.Chen Y, Zhang H, Gou X, Horikawa Y, Xing J, Chen Z. Upregulation of HAb18G/CD147 in activated human umbilical vein endothelial cells enhances the angiogenesis. Cancer Lett. 2009;278:113–121. doi: 10.1016/j.canlet.2009.01.004. [DOI] [PubMed] [Google Scholar]


