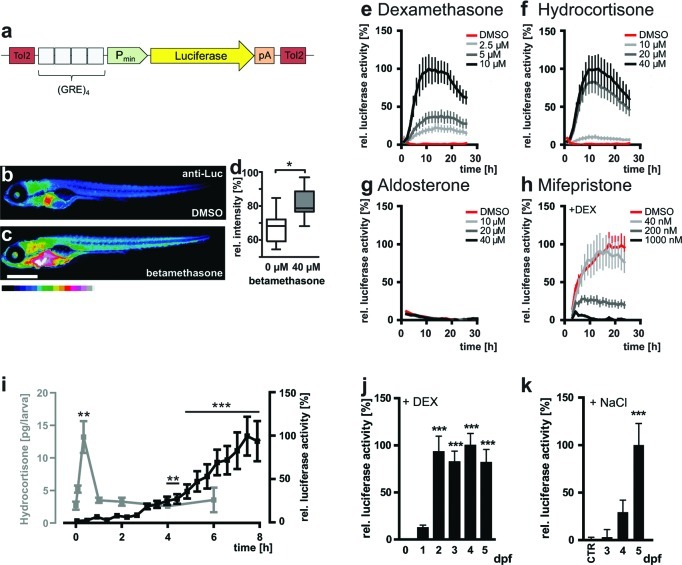
Figure 1.
Characterization of the GRE:Luc reporter line. (a) Schematic showing the structure of the reporter construct. Tol2, Tol2 transposase sites to facilitate integration into the genome; (GRE)4, four concatemerized Glucocorticoid Response Elements (GRE); Pmin, minimal promoter; pA, polyadenylation site. (b–d) Luciferase expression is ubiquitously upregulated upon GC treatment. (b, c) Immunohistochemistry with an anti-luciferase antibody (anti-Luc) and an Alexa Fluor 488 labeled secondary antibody. Fluorescence intensity is shown color coded. Representative examples of 5 dpf larvae treated with solvent control (0.3% DMSO (b)) or 40 μM betamethasone (c). Scale bar 0.5 mm. (d) Quantification of fluorescence intensity shows a significant (p = 0.0115, n = 10) increase in betamethasone treated larvae. (e–h) The bioluminescence response of GRE:Luc larvae (n = 48) to GC signaling is specific. Graphs show the relative reporter activity in response to treatment with the indicated concentrations of a GR agonist (dexamethasone (e)), the natural GC cortisol (hydrocortisone (f)), the mineralocorticoid aldosterone (g), and a GR antagonist (mifepristone, in presence of 5 μM dexamethasone (h)). Red, control treatments. (i) GRE:Luc larvae increase bioluminescence in response to a rise in endogenous cortisol levels under osmotic stress. Upon salt stress, endogenous cortisol levels (gray, left y-axis) rise to a peak within 20 min (p ≤ 0.01, n = 5) and a significant increase in bioluminescence (black, right y-axis) is observed after 4 h of treatment (p ≤ 0.01, n = 22). (j) Developmental time course of GC signaling activation by DEX. Treatment with DEX leads to a significant rise in bioluminescence starting at 2 dpf (black bars, p ≤ 0.001, n ≥ 48). Error bars represent mean values ± SEM. (k) Developmental time course of osmotic stress response. A trend for increased bioluminescence is observed at 4 dpf, which becomes significant at 5 dpf (black bars, p ≤ 0.001, n = 288).