Figure 1.
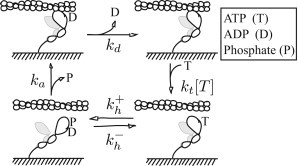
Kinetic scheme for the interaction of actin and myosin. (Starting at the lower left) Myosin releases phosphate, strongly binds to actin, and undergoes a power stroke (not necessarily in that order) at a rate ka. It releases ADP at a rate kd. Strong binding is terminated by the binding of ATP at a rate kt[T], where [T] is the concentration of ATP. Hydrolysis of ATP reverses the power stroke and reprimes the head. Although both heads of myosin are pictured, we only consider the action of the unshaded head and do not imply anything about intramolecular head-head interactions.
