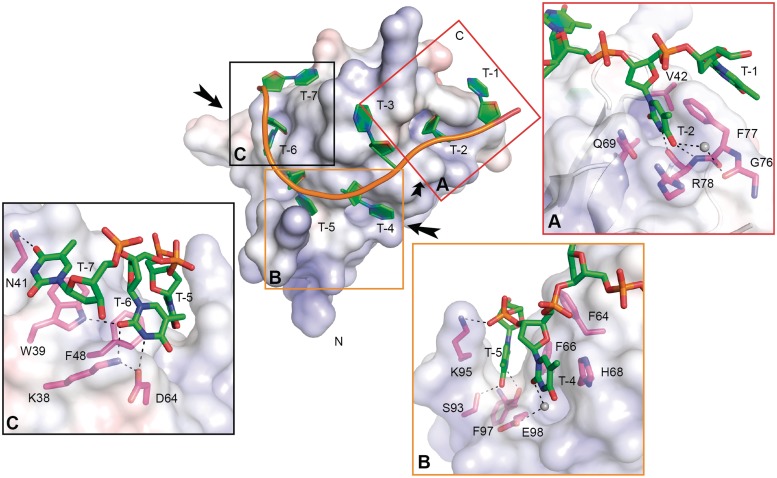
Figure 3.
Molecular interactions that promote binding of dT7 to XtrLin28b CSD. The electrostatic surface potential of XtrLin28b CSD upon binding to dT7 was calculated with APBS for pH 7 with a range from −10 (red) to +10 kT (blue). The bound oligonucleotide is shown in cartoon representation. Water molecules are shown as gray spheres. (A) Detailed view of T-2 bound to the hydrophobic pocket at binding subsite 2. Besides hydrophobic contacts including a three-membered stack with Phe77 and T-1, T/U-specific contacts are mediated via the backbone atoms of Arg78 and Phe77. (B) Detailed view of T-5 bound in the hydrophobic cleft created by Lys95 and Phe66. Ser93 and Phe97 form hydrogen bonds with O4 and HN3 of T-5. Binding of T-4 is only mediated via stacking interactions with His68 and a water molecule-mediated hydrogen bond. (C) Detailed view of binding subsites 6 and 7. T-6 and T-7 are mainly bound via stacking interactions with Phe48 and Trp39, respectively.