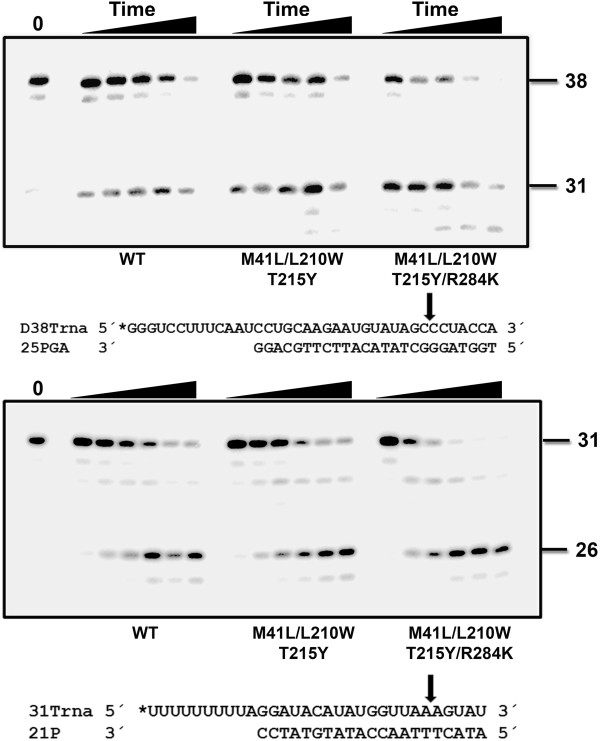
Figure 5.
RNase H activity of wild-type and mutants RTs M41L/L210W/T215Y and M41L/L210W/T215Y/R284K. [32P]RNA/DNA substrates (50 nM) were cleaved at 37°C in the presence of the corresponding RT at 50 nM concentration. Template-primer sequences are shown below. Arrows in the template sequences indicate the cleavage sites. For D38Trna/25PGA, the time points were taken after incubating the samples for 20 s, 40 s, and 1, 2 and 4 minutes. Catalytic rate constants for the cleavage of D38Trna were 0.34 ± 0.15 min-1, 0.39 ± 0.18 min-1 and 1.16 ± 0.54 min-1 for WT, and mutant RTs M41L/L210W/T215Y and M41L/L210W/T215Y/R284K, respectively. For 31Trna/21P, the time points were drawn after 20 s, 40 s, and 1, 2, 3 and 4 minutes. The catalytic rate constants with this substrate were 0.33 ± 0.05 min-1 for WT RT, and 0.35 ± 0.03 min-1 and 0.87 ± 0.12 min-1 for mutants M41L/L210W/T215Y and M41L/L210W/T215Y/R284K, respectively. Kinetic data were averages of three independent experiments.