Table 1.
Comparison of Baseline and Posttreatment Electrocardiographic Measurements for Subjects Completing the Study (n=24)
| |
Baseline |
Posttreatment |
|
||
|---|---|---|---|---|---|
| Interval | Mean (SD) | Range | Mean (SD) | Range | p value |
| PR | 130 (19) | 98–170 | 133 (19) | 100–174 | 0.16 |
| QRS | 82 (13) | 56–106 | 82 (8.9) | 60–92 | 0.92 |
| RR | 0.73 (0.18) | 0.48–1.43 | 0.75 (0.16) | 0.52–1.3 | 0.42 |
| QT | 352 (33) | 290–440 | 353 (33) | 304–446 | 0.70 |
| QTcB | 416 (21) | 368–468 | 412 (16) | 385–439 | 0.23 |
| QTcN | 398 (18) | 375–443 | 395 (15) | 368–419 | 0.39 |
| QTcF | 393 (18) | 368–436 | 391 (16) | 363–414 | 0.49 |
QTcB=Bazett's correction (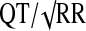 ); QTcN=FDA Neuropharmacology Division's correction (QT/RR0.37); QTcF=Fridericia's correction (
); QTcN=FDA Neuropharmacology Division's correction (QT/RR0.37); QTcF=Fridericia's correction (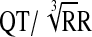 ).
).
There were no significant changes in any of the electrocardiographic parameters after the treatment phase of aripiprazole. All of the numbers measured were in the normal range for their respective intervals. All values are expressed in ms, except RR which is expressed in seconds. Statistical significance is defined as p≤0.05 using a paired, two-tailed t test.
