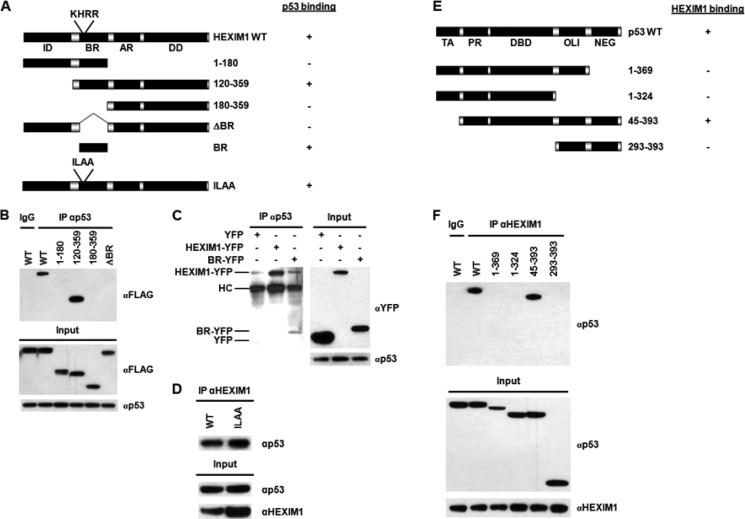
FIGURE 2.
Domain study. A, diagram of HEXIM1 truncations. The HEXIM1 constructs were FLAG- or YFP-tagged, while the HEXIM1-ILAA mutant was not tagged. The p53 binding data are summarized on the right. ID, inhibitory domain; BR, basic region; AR, acidic region; DD, dimerization domain. B, HEK293 cells were transiently transfected with the indicated FLAG-tagged HEXIM1 plasmids. Lysates of the transfected cells were immunoprecipitated with an anti-p53 antibody or a normal IgG (as a negative control). The precipitated protein complexes were analyzed by immunoblotting using an anti-FLAG antibody. C, HEK293 cells were transfected with YFP, BR-YFP, or HEXIM1-YFP plasmids. IPs were performed using the lysates of the transfected cells and an anti-p53 antibody. The immunoprecipitated complexes were analyzed by Western blotting using an anti-GFP antibody, which recognized YFP as well. HC, the heavy chain of anti-p53 antibody. D, cell lysates prepared from the HEXIM1 WT or HEXIM1 ILAA-overexpressing HEK293 cells were utilized for IP analyses. IPs were carried out using an anti-HEXIM1 antibody. The immunoprecipitated protein complexes were analyzed by Western blotting using an anti-p53 antibody. E, diagram of p53 deletions. The HEXIM1 binding data are summarized on the right. TA, transactivation domain; PR, proline-rich region; DBD, DNA binding domain; OLI, oligomerization domain; NEG, negative regulation domain. F, HEK293 cells were transiently transfected with an indicated p53 plasmid. Lysates of the transfected cells were analyzed by IPs with an anti-HEXIM1 antibody or a normal IgG, followed by immunoblotting using an anti-p53 antibody.