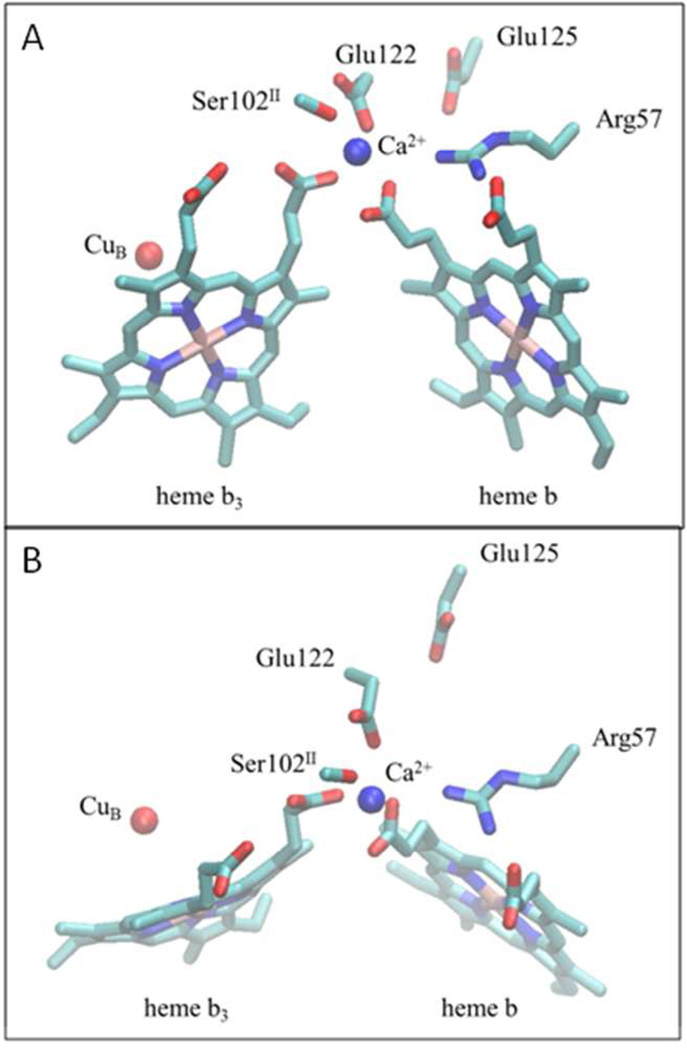
Figure 1. Structure of the Ca+2 binding site in the C-family oxygen reductase from Pseudomonas stutzeri (PDB: 3MK7).
(A) The Ca+2 binding site, located at the interface between subunits I (CcoN) and II (CcoO), as seen from the side. The Ca2+ ion (blue) is ligated to the D-propionates of both the low-spin heme b and the active-site high spin heme b3, along with two conserved amino acid side chains, Glu122 in subunit I (CcoN) and Ser102 in subunit II (CcoO). The A- and D-propionates of the low-spin heme are both hydrogen bonded to a conserved arginine (Arg57) in subunit I. (B) A view of the same region of the protein as seen from the top (periplasmic side). The residue numbering corresponds to the P. stutzeri cbb3 oxygen reductase.