Abstract
Blocks in genetic programs required for terminal myeloid differentiation and aberrant proliferation characterize acute myeloid leukemia (AML) cells. 1,25-Dihydroxy-vitamin D3 (VitD3) arrests proliferation of AML cells and induces their differentiation into mature monocytes. In a previous study, we showed that miR-26a was induced upon VitD3-mediated monocytic differentiation. Here, we identify E2F7 as a novel target of miR-26a. We show that E2F7 significantly promotes cell cycle progression and inhibits monocytic differentiation of AML cells. We also demonstrate that E2F7 binds the cyclin-dependent kinase inhibitor p21CIP1/WAF1 (cyclin-dependent kinase inhibitor 1A) promoter repressing its expression. Moreover, interfering with E2F7 expression results in inhibition of c-Myc (v-myc myelocytomatosis viral oncogene homolog) transcriptional activity. This leads to the downregulation of c-Myc transcriptional target miR-17-92 cluster, whose expression has a well-defined role in contributing to block monocytic differentiation and sustain AML cell proliferation. Finally, we show that the expression of E2F7 is upregulated in primary blasts from AML patients. Thus, these findings indicate that the newly identified miR-26a target E2F7 might have an important role in monocytic differentiation and leukemogenesis.
Keywords: microRNAs, miR-26a, E2F7, acute myeloid leukemia
Acute myeloid leukemia (AML) is a heterogeneous hematopoietic malignancy in which immature progenitor cells accumulate in the bone marrow (BM) and eventually in blood and organs interfering with the production of normal blood cells.1 AML is characterized by genetic and epigenetic alterations in progenitor cells that produce complete or partial blockage at different stages of myeloid differentiation and uncontrolled proliferation.2 Studying the functional interactions between genes that control the correct balance between cell proliferation and differentiation is critical to understand how their deregulated expression may contribute to leukemogenesis.
Cells derived from different AML French–American–British (FAB) subtypes, from M2 to M5, can be induced to differentiate, by specific agents, into cells that resemble normal counterparts.1, 3, 4, 5 In particular, the vitamin D3 metabolite 1,25-dihydroxy-vitamin D3 (VitD3) can arrest proliferation of leukemic cells with a concomitant differentiation into more mature monocyte/macrophage cellular phenotype; thus, it is considered a good model for normal monocytic differentiation studies. One of the mechanisms responsible for the effect of VitD3 on monocyte/macrophage differentiation is the induction of the cyclin-dependent kinase inhibitor p21CIP1/WAF1 (cyclin-dependent kinase inhibitor 1A).6, 7, 8 This multifunctional protein contributes to the cell cycle arrest and protection against apoptosis observed in mature monocytes. MicroRNAs (miRNAs) are short noncoding RNAs that have been shown to be involved in a variety of biological processes, including differentiation and oncogenesis, via the post-transcriptional regulation of protein coding genes. Several studies indicated that miRNAs are critical regulators of molecular networks involved in hematopoietic differentiation.9 Moreover, deregulation of miRNA expression characterizes different subtypes of AML and may contribute to leukemogenesis by altering normal hematopoietic differentiation.9 Taken together with protein factors, miRNAs have important roles in VitD3-induced cell cycle arrest and monocytic differentiation of AML cells.10, 11
Myc proteins are found to have a relevant role in controlling blood cell homeostasis.12 They are transcription factors with essential roles in cell growth and proliferation through their ability to both positively and negatively regulate the expression of coding and noncoding genes.13, 14 C-Myc (v-myc myelocytomatosis viral oncogene homolog) is frequently activated in AML and has an important role in the induction of leukemogenesis.12 Notably, c-Myc transcriptionally represses p21CIP1/WAF1 expression, thereby sustaining AML cell proliferation. Conversely, p21CIP1/WAF1 represses Myc transcriptional activity by interfering with its dimerization with Max protein.15 Repression of tumor-suppressing miRNAs is a fundamental component of the Myc tumorigenic program. One of the relevant miRNAs repressed by c-Myc in hematopoietic malignancies is miR-26a,16, 17 which was shown to act as a tumor suppressor in different types of cancer.18, 19, 20, 21, 22
In a previous study, we showed that miR-26a was induced upon VitD3-mediated monocytic differentiation of AML cell lines.17 In addition, we have shown that miR-26a was downregulated in primary AML blasts and, if reconstituted in AML cell lines, was able to decrease proliferation and induce myeloid differentiation.17 However, the mechanism underlying this effect was not fully understood. Here, we identify the transcriptional repressor E2F7 as a novel target for miR-26a function in AML. We show that E2F7 promotes cell cycle progression and inhibits monocytic differentiation of AML cells. The repression of E2F7 by miR-26a contributes to the increased expression of p21CIP1/WAF1 observed during VitD3-induced monocytic differentiation of AML cells. In addition, interfering with E2F7 expression results in inhibition of c-Myc activity and downregulation of the oncogenic miR-17-92 cluster. Finally, we further show that, conversely to miR-26a, the expression of E2F7 is upregulated in AML blasts. Therefore, we identified a new regulatory network that might have a relevant role in leukemogenesis.
Results
E2F7 is a direct target of miR-26a and is upregulated in AML
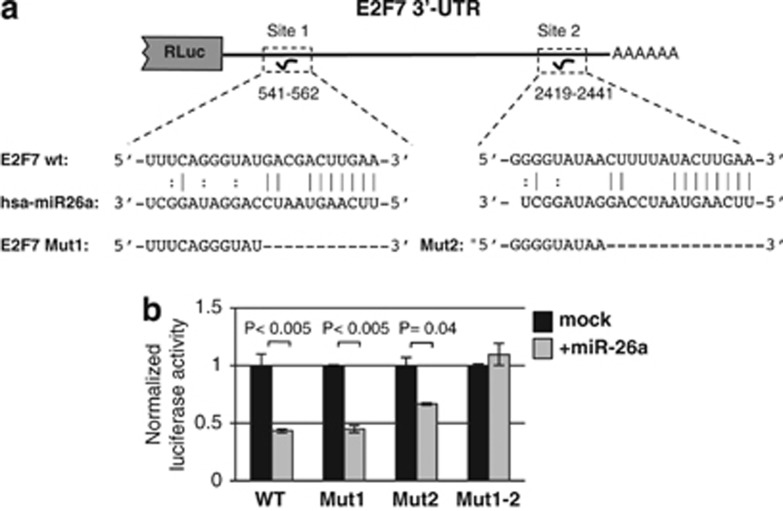
We previously showed that miR-26a negatively regulates cell proliferation and stimulates myeloid differentiation of AML cells.17 Notably, miR-26a negatively regulates G1/S transition in different tumors and this has been attributed, at least in part, to the direct regulation of EZH2 by miR-26a.16, 17, 19, 21 However, we showed that EZH2 is not a direct target of miR-26a in AML cells.17 Thus, we decided to investigate novel relevant targets for miR-26a function in AML. Between the putative targets of miR-26a, we noticed the E2F7 transcription factor (data from TargetScan (www.targetscan.org) and Pictar (pictar.mdc-berlin.de)), an important regulator of the G1/S-phase transition.23 The 3'-untranslated region (3′-UTR) of E2F7 contained two highly conserved putative binding sites for miR-26a (Figure 1a). To show the direct targeting of E2F7 by miR-26a, we constructed luciferase reporters containing the wild type (WT) 3′-UTR of E2F7 (WT), the E2F7 3′-UTR mutated at the miR-26a binding site I (Mut1), the E2F7 3′-UTR mutated at the miR-26a binding site II (Mut2) or the E2F7 3′-UTR mutated at both miR-26a binding sites (Mut1–2). Luciferase reporters were individually co-transfected in HeLa cells with a plasmid expressing miR-26a or control (Figure 1b). The expression of WT reporter was significantly decreased by miR-26a compared with control. Reporter Mut1 behaved similarly to WT, while deletion of Mut2 partially relieved translational repression. On the other hand, expression of the reporter construct mutated at both miR-26a binding sites of E2F7 3′-UTR (Mut1–2) was not sensitive to miR-26a translational repression. This may be explained by the fact that the binding of miR-26a on site II increases the accessibility to site I or, alternatively, binding of one repressive complex on site II may serve as scaffold for attracting cofactors necessary for repression on site I. These results demonstrated that miR-26a regulated E2F7 expression through direct binding of the 3′-UTR.
Figure 1.
E2F7 is a direct target of miR-26a. (a) Schematic representation of the constructs used in the luciferase assays. Positions and sequences of predicted miR-26a binding sites are shown. (b) Luciferase assays in HeLa cells transfected with the constructs indicated in panel a with an expression plasmid for miR-26a (+miR-26a) or control plasmid (mock). P-values are indicated
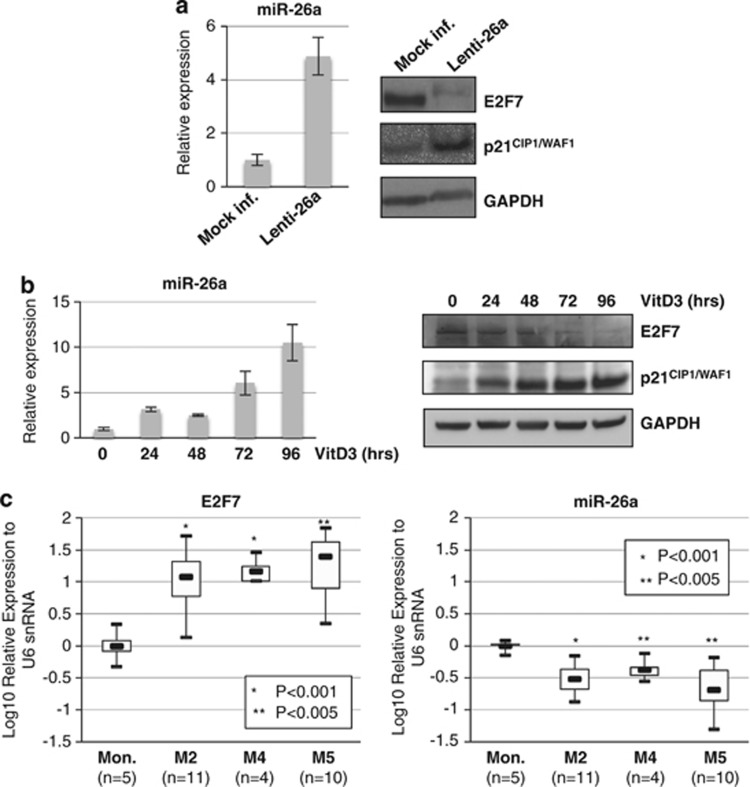
To further demonstrate that miR-26a represses E2F7 expression in AML, U937 cells were infected with a lentivirus containing an miR-26a expression cassette (Lenti-26a) or control (Mock inf.). Enforced expression by lentiviral transduction significantly increased miR-26a levels with a concomitant decrease in E2F7 protein levels (Figure 2a). Similar results were obtained with the HL-60 cell line (Supplementary Figure 1). Altogether, these data indicated that E2F7 is a direct target of miR-26a in AML.
Figure 2.
Expression analysis of miR-26a and E2F7. (a, left panel) qPCR analysis of miR-26a levels in HL-60 and U937 cells infected with a lentivirus for ectopic expression of miR-26a (Lenti-26a) or control (Mock inf.). Values were normalized for U6 expression. The histograms represent the means±S.E.M. from triplicates. (Right panel) Western blot analysis of E2F7 and p21 protein levels from cells transduced with control lentivirus (Mock inf.) or Lenti-26a. GAPDH was utilized as the loading control. (c) qPCR analysis of miR-26a levels during VitD3-induced monocytic differentiation of U937 cell line. Values were normalized for U6 expression. The histograms represent the means±S.E.M. from triplicates. (c) Western blot analysis of E2F7 and p21CIP1/WAF1 protein levels during VitD3-induced monocytic differentiation of U937 cell line. GAPDH was utilized as the loading control. (c) qPCR analysis of E2F7 and miR-26a levels in primary leukemia cells derived from patients with M2, M4 and M5 AML subtypes (n=19) and monocytes (Mon.) isolated from healthy donors (n=5). Box plots graphically represent numerical data and the average of expression levels is indicated. Values were normalized with U6 snRNA. P-values are indicated
We also examined the expression levels of E2F7 protein and miR-26a during monocytic differentiation of U937 induced by VitD3 (Figure 2b). As expected, E2F7 protein levels decreased upon monocytic differentiation and are inversely correlated to miR-26a levels (Figure 2b). Similar results were obtained during monocytic differentiation of HL-60 cells (Supplementary Figure 1). We previously showed that miR-26a is downregulated in primary AML samples.15 Thus, we analyzed E2F7 expression levels in human monocytes isolated from healthy donors and diagnostic samples of AML patients by real-time RT-PCR (Figure 2c). We have restricted the analysis to M2, M4 and M5 FAB subtypes because they can respond to monocytic differentiation in vitro.3, 4 High expression levels of E2F7 were detected in all AML samples compared with mature monocytes. These data indicated that E2F7 is upregulated in primary AML samples and that its expression is inversely correlated with miR-26a.
Decreased E2F7 levels stimulate monocytic differentiation and inhibit proliferation of AML cells
It has been previously demonstrated that miR-26a induced the expression of p21CIP1/WAF1 (Figure 2a) and, concomitantly, monocytic differentiation of AML cell lines.17 Thus, we next investigated the role of E2F7 in monocytic cell differentiation.
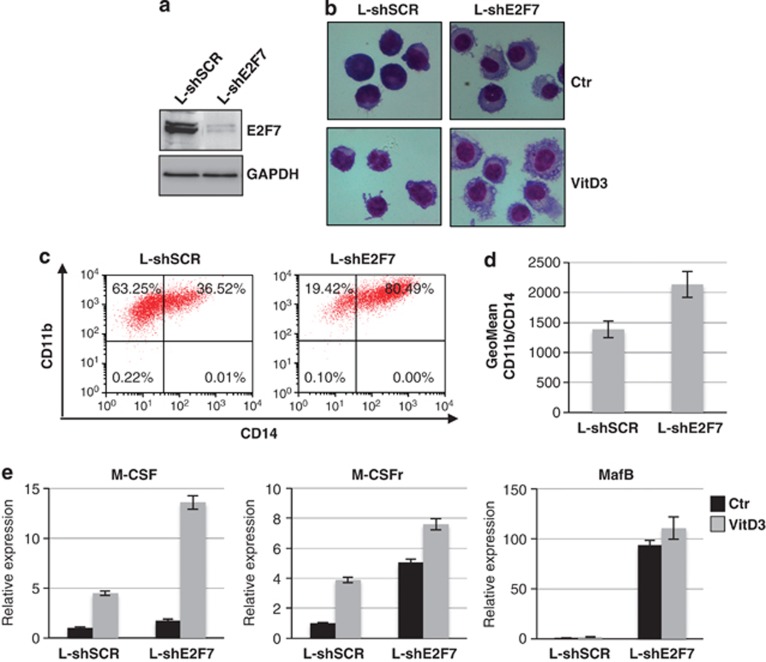
We selected U937 cells for further experiments because they have been extensively used as suitable model systems to study monocytic differentiation. These cells differentiate into mature monocytes by treatment with VitD3, a process mainly mediated by p21CIP1/WAF1 expression and subsequent G0/G1 cell cycle arrest.6, 7 Cells were infected with lentiviral vectors expressing short hairpin RNAs (shRNAs) against E2F7 (L-shE2F7) or scramble shRNA (L-shSCR) and differentiation was evaluated by analyzing the morphology, immunophenotype and induction of specific molecular markers. Cells infected with Lenti-shE2F7 exhibited almost a complete depletion of E2F7 protein level (Figure 3a). Strikingly, the depletion of E2F7 induced drastic changes in the morphology of transduced cells. A nuclear condensation and more abundant cytoplasm were observed, indicating an induction of myeloid differentiation (Figure 3b). Notably, cells interfering with E2F7 presented a more pronounced differentiation upon VitD3 treatment with a feature of matured monocytes/macrophages, such us indented nuclei, increased cytoplasmic processes and granules. Immunophenotype was analyzed to further examine differentiation. A strong increase in cells expressing the surface markers, cluster of differentiation 14 (CD14) and cluster of differentiation 11b (CD11b), which are upregulated during myelomonocytic differentiation, was observed upon depletion of E2F7 and treatment with VitD3 (Figures 3c and d). We also measured mRNA expression levels for marker of monocytic differentiation such as M-CSF, M-CSFr and MafB (Figure 3e). We detected a strong increase in monocytic markers upon inhibition of E2F7 expression by RNAi, even in the absence of any inducer, indicating that the depletion of E2F7 is sufficient to commit U937 toward monocytic differentiation. Similar results were obtained with HL-60 cells (Supplementary Figure 2). Altogether, these results indicate that decreased E2F7 levels stimulated monocytic/macrophage differentiation of AML cells.
Figure 3.
E2F7 counteracts monocytic differentiation of AML cells. (a) Western blot analysis of E2F7 levels from U937 cells infected with L-shE2F7 or L-shSCR. GAPDH was utilized as the loading control. (b) Morphological analysis of L-shE2F7- and L-shSCR-infected cells treated without (Ctr) or with 250 ng/ml of VitD3 for 72 h. (c) Representative FACS analysis of CD11b- and CD14-positive cells after VitD3 treatment. (d) The histogram represents the geometric mean±S.E.M. of CD11b/CD14-positive cells after VitD3 treatment and indicates the average intensity of CD11b/CD14 in a population. (e) qPCR analysis of monocytic specific markers. Data were normalized for GAPDH mRNA. The histograms represent the means±S.E.M. from three independent experiments
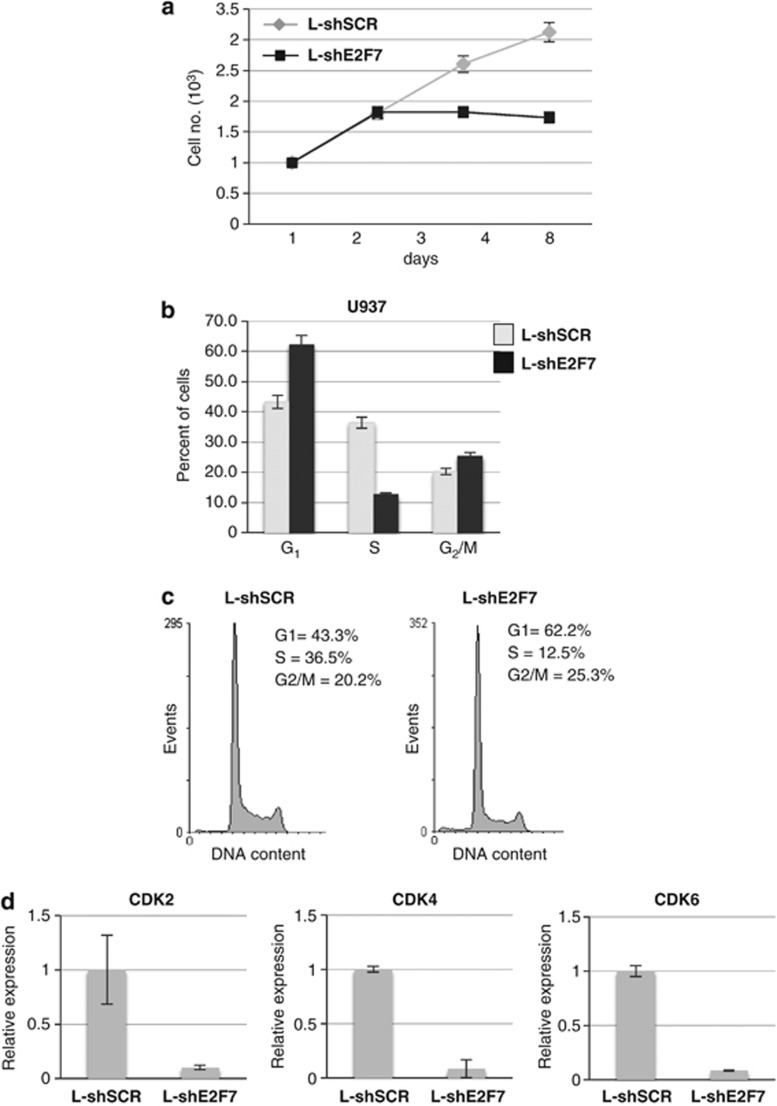
In AML, the terminal differentiation block is strictly related to aberrant proliferation.2 Thus, to better understand the activity of E2F7 in leukemic cells, we analyzed the effect of E2F7 on cellular proliferation and cell cycle. U937 cells infected with L-shE2F7 showed lower proliferation rate than control L-shSCR cells (Figure 4a). In addition, cell cycle analysis showed that silencing of E2F7 expression increased the percentage of cells in G1 arrest and decreased the number of cells in the S phase (Figures 4b and c). Expression analysis of relevant cell cycle regulators by real-time PCR (qPCR) indicated that E2F7 silencing reduced the levels of several positive regulators of the G1 phase, including CDK2, CDK4 and CDK6 (Figure 4d). Similar results were obtained with HL-60 cells (Supplementary Figure 3). These data indicated that endogenous E2F7 could be essential for maintaining cellular proliferation of AML cells.
Figure 4.
Interference against E2F7 inhibits AML cell proliferation. (a) Effect of L-shE2F7 and L-shSCR on cell proliferation. (b) Cell cycle distribution of cells transduced with L-shE2F7 and L-shSCR. The histograms represent the means±S.E.M. from three independent experiments. (c) Representative cell cycle analysis of cells transduced with L-shE2F7 and L-shSCR. (d) qPCR analysis of positive cell cycle regulators. Data were normalized for GAPDH mRNA. The histograms represent the means±S.E.M. from three independent experiments
Increased E2F7 levels counteract monocytic differentiation and stimulate proliferation of AML cells
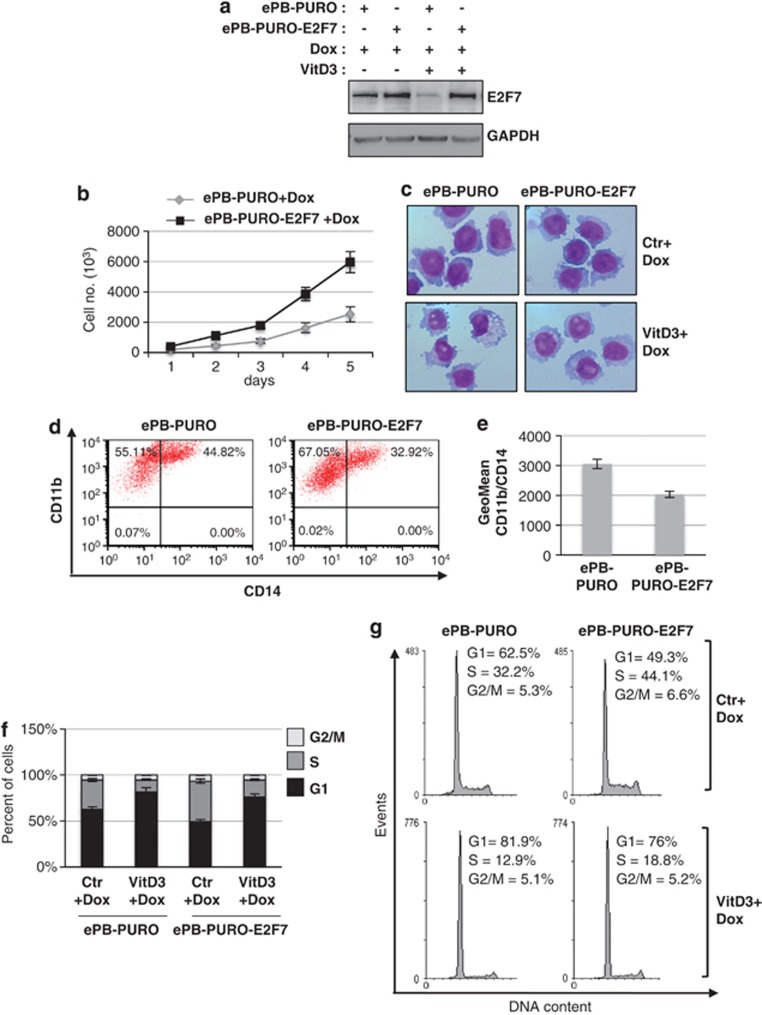
We next evaluated whether enforced expression of E2F7 affected the differentiation status and proliferation of AML cells. Thus, we cloned an E2F7 derivative with a 3′-UTR devoid of miRNA target sites in a stable and Tet-on inducible system derived from the enhanced PiggyBac (ePiggyBac) transposon (EBP-PURO) giving rise to the EBP-PURO-E2F7 construct. Doxycycline (Dox) treatment of U937 cells transformed with EBP-PURO-E2F7 increased the levels of E2F7 as compared with those transformed with the empty vector EBP-PURO (Figure 5a). Moreover, E2F7 overexpression is retained after VitD3 treatment, when endogenous E2F7 protein levels decreased (Figure 5a). High levels of E2F7 resulted in a reduction of VitD3-induced monocytic differentiation assessed by morphological changes and percentage of CD11- and CD14-positive cells compared to cells transfected with the empty vector (Figures 5c–e). Moreover, enforced E2F7 expression resulted in increased proliferation rate and percentage of cells in the S phase, while decreased the number of cells arrested in the G1 phase (Figures 5b, f and g). Notably, this effect is maintained even after VitD3 treatment (Figures 5f and g). Altogether, these data indicated that increased levels of E2F7 stimulated proliferation and counteracted monocytic differentiation of AML cells; therefore, suggesting that one of the pathways by which miR-26a promotes monocytopoiesis of AML cells is through E2F7 repression.
Figure 5.
Enforced E2F7 expression stimulates AML cell proliferation and counteracts monocytic differentiation of AML cells. (a) Western blot analysis of E2F7 protein levels in U937 cells transformed with EPB-PURO and EPB-PURO-E2F7. Data were normalized for GAPDH. (b) Effect of EPB-PURO and EPB-PURO-E2F7 on U937 proliferation upon Dox treatment (200 ng/ml). (c) Morphological analysis of EPB-PURO and EPB-PURO-E2F7 cells treated with Dox alone or in combination with VitD3 for 72 h. (d) Representative FACS analysis of CD11b- and CD14-positive cells after Dox and VitD3 treatment. (e) The histogram represents the geometric mean±S.E.M. of CD11b/CD14-positive cells after Dox and VitD3 treatment and indicates the average intensity of CD11b/CD14 in a population. (f) Cell cycle distribution of cells transduced with EPB-PURO and EPB-PURO-E2F7 and treated with Dox alone or in combination with VitD3 for 72 h. (g) Representative cell cycle analysis of (f)
E2F7 represses p21CIP1/WAF1 expression
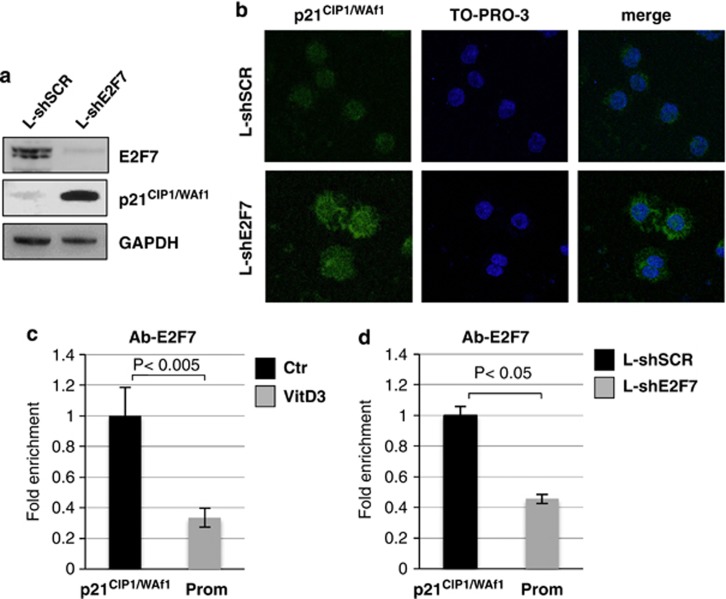
To explain the effect of E2F7 on AML cells, we investigated the level of the cyclin-dependent kinase inhibitor p21CIP1/WAF1, which has a crucial role in inducing cell cycle arrest and monocytic differentiation of AML cells6 and whose levels are increased upon VitD3 treatment (Figure 2b). In addition, E2F repressors, including E2F7, have been recently identified has negative regulators of p21CIP1/WAF1 transcription.23, 24 Suppression of E2F7 by RNAi was sufficient to produce a drastic increase of p21CIP1/WAF1 protein levels in AML cells (Figure 6a). Notably, a p21CIP1/WAF1 increase was also observed by overexpressing miR-26a (Figure 2a).17 Immunofluorescence analysis also indicated that p21CIP1/WAF1 protein accumulated in both nuclear and cytoplasmic compartments (Figure 6b). To clarify whether E2F7 binds the p21CIP1/WAF1 promoter in vivo, we carried out chromatin immunoprecipitation (ChIP) assays with an E2F7 antibody. To assess the specific binding of E2F7, ChIP experiments were performed on cells treated with VitD3 (Figure 5c) or upon repression of E2F7 expression by shRNAs (Figure 6d). A specific enrichment of the p21CIP1/WAF1 promoter regions in E2F7-immunoprecipitated samples from untreated cells was observed. Notably, the immunoprecipitations were strongly reduced after VitD3 treatment or RNAi for E2F7, in accordance with the decreased E2F7 protein levels (Figures 2b and 6a). These data indicate that E2F7 contributes to repress p21CIP1/WAF1 expression in AML cells.
Figure 6.
E2F7 represses the expression of p21CIP1/WAF1 in AML cells. (a) Western blot analysis of p21CIP1/WAF1 protein levels in cells transduced with L-shE2F7 and L-shSCR. Data were normalized for GAPDH. (b) Localization of endogenous p21CIP1/WAF1 protein in cells transduced with L-shE2F7 and L-shSCR. DNA staining with TO-PRO-3 and a merged view of green and blue channels of the same field is shown (merge). (c) ChIP analysis with anti-E2F7 antibody on p21CIP1/WAF1 promoter in U937 cells before and after VitD3 treatment. (d) ChIP analysis with anti-E2F7 antibody on p21CIP1/WAF1 promoter in U937 cells transduced with L-shE2F7 and L-shSCR. The recovered DNA in ChIP experiments was quantified by real-time PCR. Results are expressed as the relative level over control cells after correcting for differences in the amount of starting (input) chromatin materials. Experiments were performed in triplicate. P-values are indicated
E2F7 sustains miR-17-92 cluster expression
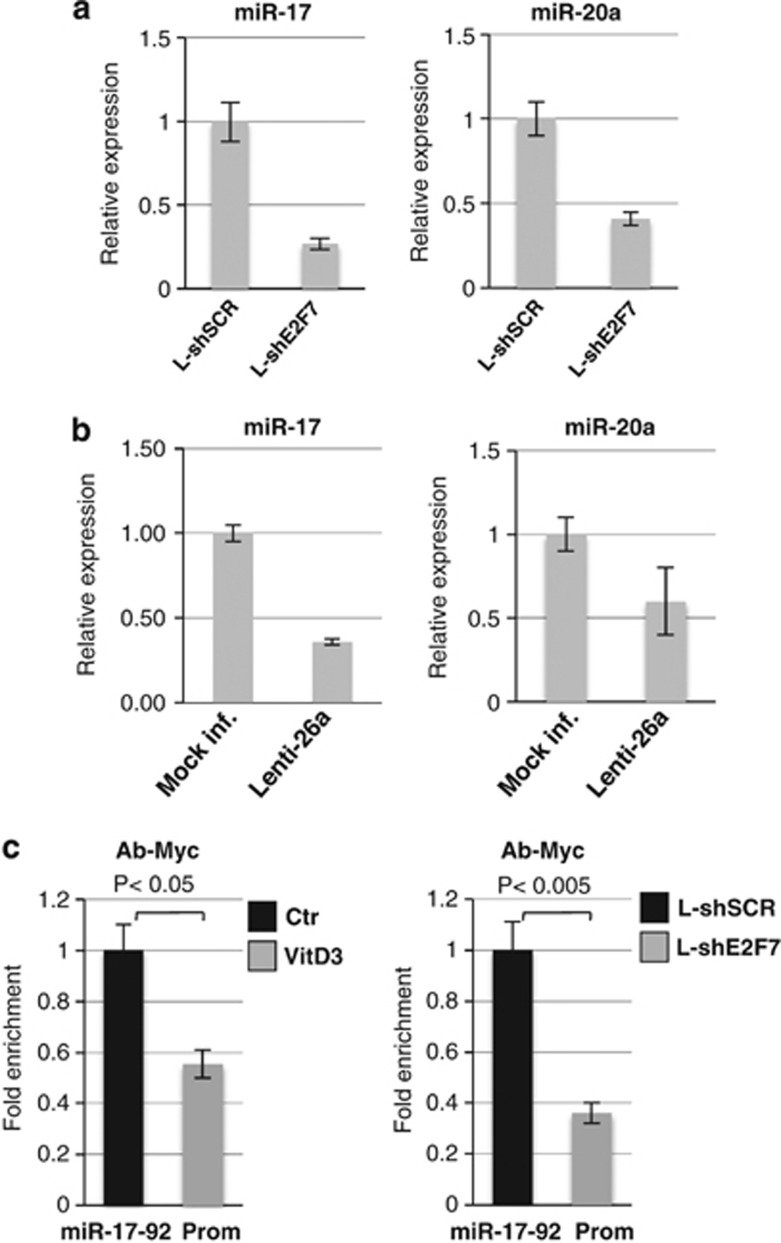
Previous work demonstrated a relevant contribution for the miR-17-92 cluster in blocking monocyte/macrophage differentiation and leukemogenesis.25, 26 More importantly, inhibition of miR-17-92 expression cluster has been associated to monocyte/macrophage differentiation of both normal progenitors and AML cells.25, 26 Thus, we analyzed the expression levels of miR-17-92 upon silencing of E2F7 in U937. All cluster miRNAs were significantly downregulated, and miR-17 and miR-20 levels are shown (Figure 7a). Interestingly, a decrease in miR-17 and miR-20 levels was also obtained upon enforced expression of miR-26a (Figure 7b). ChIP experiments showed that the binding of c-Myc on miR-17-92 promoter region is strongly reduced after VitD3 treatment and downregulation of E2F7 levels (Figure 7c). These results suggested that E2F7 could contribute to maintain c-Myc activity and high level of the miR-17-92 cluster, therein sustaining the differentiation block and aberrant proliferation of AML cells.
Figure 7.
(a) qPCR analysis of miR-17 and miR-20a in cells transduced with L-shE2F7 and L-shSCR. (b) qPCR analysis of miR-17 and miR-20a in cells transduced with Lenti-26a and mock. (c) ChIP analysis with anti-c-Myc antibody on miR-17-92 cluster promoter in cells treated with VitD3 for 72 h or transduced with L-shE2F7 and L-shSCR. The recovered DNA in ChIP experiments was quantified by real-time PCR. Results are expressed as the relative level over control cells after correcting for differences in the amount of starting (input) chromatin materials. Experiments were performed in triplicate. P-values are indicated
Discussion
AML cells are characterized by increased proliferation, block of differentiation programs and enhanced self-renewal. Insights into the molecular networks that control all these processes are critical to fully understand the leukemogenic process and develop effective therapies.
MiRNAs are critical regulators of gene programs that control blood cell homeostasis.9 For this reason, it is not surprising that hematological malignancies are characterized by miRNA misregulation.9 A critical aspect for understanding miRNA function is to link specific miRNAs to their relevant targets. A preliminary study showed that miR-26a was downregulated in AML by the oncogene c-Myc and that reconstitution of miR-26a in AML cell lines induced proliferation arrest and stimulated monocytic differentiation.17 This study identified as a novel target for miR-26a function in AML the transcriptional repressor E2F7. E2F7 belongs to the atypical E2F members, which were initially identified as a negative regulator of cell cycle progression.27 However, different studies revealed that atypical E2Fs might also act as a positive regulator of cellular proliferation.28, 29, 30, 31 Here, for the first time, we showed that E2F7 expression was upregulated in AML primary blasts. Recently, E2F7 was found upregulated in rhabdomyosarcoma, indicating that E2F7 misregulation might have significant role in tumorigenesis.32 Herein, we demonstrated that E2F7 silencing in AML cell lines inhibited the cellular growth and induced monocytic differentiation. Conversely, enforced E2F7 expression stimulated proliferation and counteracted monocytic differentiation. The E2F7 knockdown in AML cells delayed cell cycle progression and in particular G1/S transition, which was not consistent with previous data obtained in fibroblast.33 Notably, similar results have been described for the other atypical E2F factor E2F8 in hepatocellular carcinoma.28, 34 This suggests that E2F7, like E2F8, could act as a positive or negative regulator of cell cycle progression in a cellular context-dependent manner. Therefore, much more work will be necessary to identify E2F7 interacting proteins and assign specific functions to E2F7 in different cell lineages.
Importantly, both miR-26a overexpression and E2F7 inhibition produces p21CIP1/WAF1 accumulation, a factor that has a crucial role in the proliferation arrest and monocytic differentiation of AML cells.6, 8 We showed that the transcriptional repressor E2F7 is able to bind to p21CIP1/WAF1 promoter region in proliferating AML cells; thus, sustaining the differentiation block of AML cells by inhibiting p21 CIP1/WAF1 expression. In summary, we propose a model in which the translational repression of E2F7 by miR-26a could contribute to a detachment of E2F7 from p21CIP1/WAF1 promoter and, in turn, to the induction of p21CIP1/WAF1 expression during VitD3-mediated differentiation of AML cell lines. It is worth noting that just as E2F7 is just one of the hundreds of potential targets of miR-26a, p21CIP1/WAF1 is just one of the downstream effectors of E2F7. Further studies will be needed to understand the relevance of this model in normal monocytopoiesis and AML.
Previous studies have shown that the transcriptional activity of c-Myc is inhibited by p21CIP1/WAF1 protein.15 c-Myc transcriptionally activates the oncogenic miR-17-92 cluster,33 containing miRNAs known to inhibit monocytic/macrophage differentiation of both normal myeloid progenitors and AML cells.25, 26 Moreover, increased expression of miR-17-92 has been reported in AML patients where it helps to maintain self-renewal of leukemic cells.26, 35 Our studies showed that, in our cellular system, E2F7 contributed to control miR-17-92 expression. Indeed, E2F7 inhibition is sufficient to produce a decrease of miR-17-92. This suggests a model in which E2F7 could control miR-17-92 expression through the inhibition of c-Myc activity by p21CIP1/WAF1. However, further experiments will be needed to clarify the relation between p21CIP1/WAF1, c-Myc and miR-17-92 expression. Notably, the p21CIP1/WAF1 mRNA is also a target of the miR-17 family.26, 35, 36 Consequently, the decreased expression of miR-17-92 might contribute to enforce p21CIP1/WAF1 expression during AML cell differentiation. This indicates that upregulation of E2F7 could contribute to oncogenesis in AML by maintaining high levels of miR-17-92.
In conclusion, this study identified a novel aspect of VitD3 action in regulating proliferation and differentiation of AML cells through the miR-26a/E2F7/p21CIP1/WAF1 network. However, it should be considered that the level of complexity of gene regulation during these processes is such that no single circuitry operates in isolation and they are very likely part of fully integrated networks within the cells. Therein, the biological effect of a specific miRNA strongly depends on the cellular context. In particular, miR-26a has been shown to induce proliferation arrest in different tumor model systems, such as lymphomas and liver, breast and nasopharyngeal carcinomas.16, 17, 21, 22, 37 Conversely, it has been shown to act as an oncogene in glioblastoma.38 An important aspect to consider in using miR-26a as an antitumor molecule in AML is that p21CIP1/WAF1 not only has a relevant role in the proliferation arrest during differentiation of normal and pathological hematopoietic progenitors6, 7, 8 but also prevents excess DNA-damage accumulation and exhaustion of cancer stem cells.39 Thus, consideration should be given to the design of antitumor therapies based on miR-26a.
Materials and Methods
Cell cultures and reagents
HL-60 and U937 cell lines were maintained in RPMI 1640 medium supplemented with 1 × penicillin/streptomycin solution, 1 × ℒ-glutamine and 10% fetal bovine serum. VitD3 and Dox were purchased from Sigma-Aldrich (St. Louis, MO, USA) and utilized at a concentration of 250 and 200 ng/ml, respectively, unless differently specified.
Mononuclear cells (MCs) were isolated from the peripheral blood (PB) of informed healthy donors as reported.17 Monocytes were isolated from MCs by adherence. Percentage of monocytes after the isolation was assessed using morphology and FACS analysis and was higher than 80%. PB/BM cells were obtained from 21 informed, newly diagnosed AML patients (Supplementary Table 1). Cases were classified according to the FAB classification and showed an initial percentage of circulating blasts more than 80%.
RNA interference and miR-26a overexpression
E2F7 knockdown was obtained by Mission Lentiviral shRNA clones targeting E2F7 TRCN0000017455 and TRCN000017453 (Sigma-Aldrich). Mission Lentiviral Non-Targeting shRNA clone SHC002 (Sigma-Aldrich) was utilized as control. Lentiviral particles were prepared according to the manufacturer's specifications. Lentiviral vectors for miR-26a expression and control were described previously.17 Infection of AML cell lines was performed as described previously.40
Cell proliferation and differentiation
For cell cycle analysis, 2 × 105 cells were resuspended in PBS/50% FCS, fixed in 70% ethanol for 24 h, incubated with 50 Ag/ml propidium iodide (Sigma-Aldrich) and 50 U/ml DNase-free RNase A (Sigma-Aldrich), and analyzed after 3 h (10 000 events) using an Epics XL Cytometer (Beckman Coulter, Brea, CA, USA). Morphology was evaluated after 7 days from lentiviral transduction in conventional light-field microscopy of Wright–Giemsa-stained cytospins. Differentiation was assessed by direct immunofluorescence staining of cells using an allophycocyanin anti-human CD11b (Becton Dickinson, San Jose, CA, USA), PerCP anti-human CD14 (Becton Dickinson) and PE-IgG1 isotype control (eBiosciences, San Diego, CA, USA) as described previously.17 A minimum of 10 000 events were collected for each sample with flow cytometer (CyAN ADP; Dako Italia S.p.a., Milan, Italy) by using the Summit 4.3 software (Beckman Coulter) for data acquisition and analysis.
Immunofluorescence
A total of 2 × 105 cells were spotted onto the slide by cytospin, fixed with 4% paraformaldehyde in PBS for 15 min at room temperature (RT) and then washed with PBS for 10 min at RT. The cells were incubated with p21 antibody (Cell Signaling Technology, Beverly, MA, USA) and diluted in 1% PBS/BSA for 1 h at RT. The slides were then washed with PBS for 10 min at RT and subsequently incubated with Alexa Fluor 488 (rabbit)-conjugated secondary antibody (Molecular Probes Inc., Eugene, OR, USA) for 1 h at RT. After washing with PBS, slides were counterstained with TO-PRO-3 for 10 min and mounted with Vectashield (Vector Labs, Burlingame, CA, USA). Cells were examined under a Leica TCS SP2 fluorescence confocal microscope (Leica Microsystems, Wetzlar, Germany) (argon 488 nm laser). Photomicrographs were acquired with LAS AF Software (Leica Microsystems).
RNA extraction and analysis
Total RNA was prepared using Qiazol reagent and miRNAeasy kit (Qiagen, Hilden, Germany). Hsa-miR-26a analysis by real-time PCR was performed using miRScript System (Qiagen) and E2F7 analysis was performed with QuantiTect Primer Assay QT00059115 (Qiagen). Delta-delta Ct values were normalized with those obtained from the amplification of the endogenous U6 snRNA (Qiagen). P-values between primary AML samples and monocytes data were obtained with t-test. cDNAs for p21, CDK2, CDK4 and CDK6 analysis were generated with miRScript RT Kit (Qiagen). Oligonucleotides are described in the primers list (shown below). M-CSF, M-CSFr and MafB were detected with TaqMan oligonucleotides Hs00166144_m1, Hs00234622_m1 and Hs00534343_s1, respectively, and normalized by the amplification of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) Hs99999905_m1 (Applied Biosystems, Life Technologies Italia, Monza MB, Italy). MiR-17 and miR-20a were detected with TaqMan microRNA assay TM-393 and TM-580 (Applied Biosystems), respectively. Delta-delta Ct values were normalized with those obtained from the amplification of the endogenous U6 snRNA, TaqMan probe TM-1093 (Applied Biosystems). All reactions were performed in triplicate.
Primers list:

Immunoblot analysis
In all, 40 μg of whole cell extract was separated by 10% SDS-PAGE and electroblotted to nitrocellulose membrane (Protran S&S, Drammen, Norway). Immunoblots were incubated with antibodies to p21 (sc-817; Santa Cruz Biotechnology, Santa Cruz, CA, USA), E2F7 (BCN4776, AntibodyBCN) and GAPDH (sc-25778; Santa Cruz Biotechnology).
ChIP assay
DNA/protein crosslinking and ChIP with anti-E2F7 (BCN4776; AntibodyBCN) and anti-c-Myc (sc-764; Santa Cruz Biotechnology) antibodies were performed as described previously.17 Immuoprecipitation without specific antibody was performed as negative control. Negative control amplifications were performed on intergenic region.17 The relative occupancy of the immunoprecipitated factor at a locus was estimated by using the comparative threshold method.17 Oligonucleotides for promoters and intergenic region detection are described in the primers list. P-values were obtained with t-test.
E2F7 overexpression
The E2F7 cDNA clone was purchased from Open Biosystems (Thermo Scientific, Rockford, IL, USA) (clone 9020354) and subcloned in the ePiggyBac (EPB) vector EPB-PURO41 generating the EPB-PURO-E2F7 plasmid. This plasmid contains a TET-on system for inducible transgene expression. Helper and transposon plasmids were electroporated in U937 with the Neon Transfection System (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instruction. Selection with puromycin was initiated 2 days after transfection and maintained until resistant colonies became visible.
Acknowledgments
We thank Professor L Naldini for lentiviral plasmids, Dr. A Rosa and Dr. A Brivanlou for the ePiggyBac inducible transposon system, O Sthandier, M Marchioni and M Arceci for technical assistance. BS is a recipient of a fellowship from the Italian Foundation for Cancer Research (FIRC). This work was supported by grants from: Italian Association for Cancer Research (AIRC), Italian Institute of Technology (IIT) ‘SEED' project, MIUR-FIRB and ‘Centro di eccellenza BEMM' – Sapienza University of Rome.
Glossary
- AML
acute myeloid leukemia
- CD11b
cluster of differentiation 11b
- CD14
cluster of differentiation 14
- ChIP
chromatin immunoprecipitation
- c-Myc
v-myc myelocytomatosis viral oncogene homolog
- Dox
doxycycline
- FAB
French–American–British
- GAPDH
glyceraldehyde-3-phosphate dehydrogenase
- miRNA
microRNA
- p21CIP1/WAF1
cyclin-dependent kinase inhibitor 1A
- qPCR
real-time PCR
- SCR
scramble
- shRNA
short hairpin RNA
- VitD3
1,25-dihydroxy-vitamin D3
- 3′-UTR
3'-untranslated region
- WT
wild type
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on Cell Death and Disease website (http://www.nature.com/cddis)
Edited by T Brunner
Supplementary Material
References
- Tenen DG. Disruption of differentiation in human cancer: AML shows the way. Nat Rev Cancer. 2003;3:89–101. doi: 10.1038/nrc989. [DOI] [PubMed] [Google Scholar]
- Kumar CC. Genetic abnormalities and challenges in the treatment of acute myeloid leukemia. Genes Cancer. 2011;2:95–107. doi: 10.1177/1947601911408076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manfredini R, Trevisan F, Grande A, Tagliafico E, Montanari M, Lemoli R, et al. Induction of a functional vitamin D receptor in all-trans-retinoic acid-induced monocytic differentiation of M2-type leukemic blast cells. Cancer Res. 1999;59:3803–3811. [PubMed] [Google Scholar]
- Wang Q, Harrison JS, Uskokovic M, Kutner A, Studzinski GP. Translational study of vitamin D differentiation therapy of myeloid leukemia: effects of the combination with a p38 MAPK inhibitor and an antioxidant. Leukemia. 2005;19:1812–1817. doi: 10.1038/sj.leu.2403916. [DOI] [PubMed] [Google Scholar]
- Tagliafico E, Tenedini E, Manfredini R, Grande A, Ferrari F, Roncaglia E, et al. Identification of a molecular signature predictive of sensitivity to differentiation induction in acute myeloid leukemia. Leukemia. 2006;20:1751–1758. doi: 10.1038/sj.leu.2404358. [DOI] [PubMed] [Google Scholar]
- Liu M, Lee MH, Cohen M, Bommakanti M, Freedman LP. Transcriptional activation of the Cdk inhibitor p21 by vitamin D3 leads to the induced differentiation of the myelomonocytic cell line U937. Genes Dev. 1996;10:142–153. doi: 10.1101/gad.10.2.142. [DOI] [PubMed] [Google Scholar]
- Asada M, Yamada T, Fukumuro K, Mizutani S. p21Cip1/WAF1 is important for differentiation and survival of U937 cells. Leukemia. 1998;12:1944–1950. doi: 10.1038/sj.leu.2401228. [DOI] [PubMed] [Google Scholar]
- Asada M, Yamada T, Ichijo H, Delia D, Miyazono K, Fukumuro K, et al. Apoptosis inhibitory activity of cytoplasmic p21(Cip1/WAF1) in monocytic differentiation. EMBO J. 1999;18:1223–1234. doi: 10.1093/emboj/18.5.1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schotte D, Pieters R, Den Boer ML. MicroRNAs in acute leukemia: from biological players to clinical contributors. Leukemia. 2012;26:1–12. doi: 10.1038/leu.2011.151. [DOI] [PubMed] [Google Scholar]
- Gocek E, Wang X, Liu X, Liu CG, Studzinski GP. MicroRNA-32 upregulation by 1,25-dihydroxyvitamin D3 in human myeloid leukemia cells leads to Bim targeting and inhibition of AraC-induced apoptosis. Cancer Res. 2011;71:6230–6239. doi: 10.1158/0008-5472.CAN-11-1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Gocek E, Liu CG, Studzinski GP. MicroRNAs181 regulate the expression of p27Kip1 in human myeloid leukemia cells induced to differentiate by 1,25-dihydroxyvitamin D3. Cell Cycle. 2009;8:736–741. doi: 10.4161/cc.8.5.7870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delgado MD, León J. Myc roles in hematopoiesis and leukemia. Genes Cancer. 2010;1:605–616. doi: 10.1177/1947601910377495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adhikary S, Eilers M. Transcriptional regulation and transformation by Myc proteins. Nat Rev Mol Cell Biol. 2005;6:635–645. doi: 10.1038/nrm1703. [DOI] [PubMed] [Google Scholar]
- Bui TV, Mendell JT. Myc: maestro of microRNAs. Genes Cancer. 2010;1:568–575. doi: 10.1177/1947601910377491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kitaura H, Shinshi M, Uchikoshi Y, Ono T, Iguchi-Ariga SM, Ariga H. Reciprocal regulation via protein-protein interaction between c-Myc and p21 (cip1/waf1/sdi1) in DNA replication and transcription. J Biol Chem. 2000;275:10477–10483. doi: 10.1074/jbc.275.14.10477. [DOI] [PubMed] [Google Scholar]
- Sander S, Bullinger L, Klapproth K, Fiedler K, Kestler HA, Barth TF, et al. MYC stimulates EZH2 expression by repression of its negative regulator miR-26a. Blood. 2008;112:4202–4212. doi: 10.1182/blood-2008-03-147645. [DOI] [PubMed] [Google Scholar]
- Salvatori B, Iosue I, Djodji Damas N, Mangiavacchi A, Chiaretti S, Messina M, et al. Critical role of c-Myc in acute myeloid leukemia involving direct regulation of miR-26a and histone methyltransferase EZH2. Genes Cancer. 2011;2:585–592. doi: 10.1177/1947601911416357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang TC, Yu D, Lee YS, Wentzel EA, Arking DE, West KM, et al. Widespread microRNA repression by Myc contributes to tumorigenesis. Nat Genet. 2008;40:43–50. doi: 10.1038/ng.2007.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kota J, Chivukula RR, O'Donnell KA, Wentzel EA, Montgomery CL, Hwang HW, et al. Therapeutic microRNA delivery suppresses tumorigenesis in a murine liver cancer model. Cell. 2009;137:1005–1017. doi: 10.1016/j.cell.2009.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y, Lu Y, Zhang Q, Liu JJ, Li TJ, Yang JR, et al. MicroRNA-26a/b and their host genes cooperate to inhibit the G1/S transition by activating the pRb protein. Nucleic Acids Res. 2012;40:4615–4625. doi: 10.1093/nar/gkr1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu J, He ML, Wang L, Chen Y, Liu X, Dong Q, et al. MiR-26a inhibits cell growth and tumorigenesis of nasopharyngeal carcinoma through repression of EZH2. Cancer Res. 2011;71:225–233. doi: 10.1158/0008-5472.CAN-10-1850. [DOI] [PubMed] [Google Scholar]
- Chen L, Zheng J, Zhang Y, Yang L, Wang J, Ni J, et al. Tumor-specific expression of microRNA-26a suppresses human hepatocellular carcinoma growth via cyclin-dependent and -independent pathways. Mol Ther. 2011;19:1521–1528. doi: 10.1038/mt.2011.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westendorp B, Mokry M, Groot Koerkamp MJ, Holstege FC, Cuppen E, de Bruin A. E2F7 represses a network of oscillating cell cycle genes to control S-phase progression. Nucleic Acids Res. 2012;40:3511–3523. doi: 10.1093/nar/gkr1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donzelli S, Fontemaggi G, Fazi F, Di Agostino S, Padula F, Biagioni F, et al. MicroRNA-128-2 targets the transcriptional repressor E2F5 enhancing mutant p53 gain of function. Cell Death Differ. 2012;19:1038–1048. doi: 10.1038/cdd.2011.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fontana L, Pelosi E, Greco P, Racanicchi S, Testa U, Liuzzi F, et al. MicroRNAs 17-5p-20a-106a control monocytopoiesis through AML1 targeting and M-CSF receptor upregulation. Nat Cell Biol. 2007;9:775–787. doi: 10.1038/ncb1613. [DOI] [PubMed] [Google Scholar]
- Pospisil V, Vargova K, Kokavec J, Rybarova J, Savvulidi F, Jonasova A, et al. Epigenetic silencing of the oncogenic miR-17-92 cluster during PU.1-directed macrophage differentiation. EMBO J. 2011;30:4450–4464. doi: 10.1038/emboj.2011.317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lammens T, Li J, Leone G, De Veylder L. Atypical E2Fs: new players in the E2F transcription factor family. Trends Cell Biol. 2009;19:111–118. doi: 10.1016/j.tcb.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng Q, Wang Q, Zong WY, Zheng DL, Wen YX, Wang KS, et al. E2F8 contributes to human hepatocellular carcinoma via regulating cell proliferation. Cancer Res. 2010;70:782–791. doi: 10.1158/0008-5472.CAN-09-3082. [DOI] [PubMed] [Google Scholar]
- Sozzani R, Maggio C, Giordo R, Umana E, Ascencio-Ibanez JT, Hanley-Bowdoin L, et al. The E2FD/DEL2 factor is a component of a regulatory network controlling cell proliferation and development in Arabidopsis. Plant Mol Biol. 2010;72:381–395. doi: 10.1007/s11103-009-9577-8. [DOI] [PubMed] [Google Scholar]
- Hazar-Rethinam M, Cameron SR, Dahler AL, Endo-Munoz LB, Smith L, Rickwood D, et al. Loss of E2F7 expression is an early event in squamous differentiation and causes derepression of the key differentiation activator Sp1. J Invest Dermatol. 2011;131:1077–1084. doi: 10.1038/jid.2010.430. [DOI] [PubMed] [Google Scholar]
- Sirma H, Kumar M, Meena JK, Witt B, Weise JM, Lechel A, et al. The promoter of human telomerase reverse transcriptase is activated during liver regeneration and hepatocyte proliferation. Gastroenterology. 2011;141:326–337. doi: 10.1053/j.gastro.2011.03.047. [DOI] [PubMed] [Google Scholar]
- Li L, Sarver AL, Alamgir S, Subramanian S. Downregulation of microRNAs miR-1, -206 and -29 stabilizes PAX3 and CCND2 expression in rhabdomyosarcoma. Lab Invest. 2012;92:571–583. doi: 10.1038/labinvest.2012.10. [DOI] [PubMed] [Google Scholar]
- Di Stefano L, Jensen MR, Helin K. E2F7, a novel E2F featuring DP-independent repression of a subset of E2F-regulated genes. EMBO J. 2003;22:6289–6298. doi: 10.1093/emboj/cdg613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christensen J, Cloos P, Toftegaard U, Klinkenberg D, Bracken AP, Trinh E, et al. Characterization of E2F8, a novel E2F-like cell-cycle regulated repressor of E2F-activated transcription. Nucleic Acids Res. 2005;33:5458–5470. doi: 10.1093/nar/gki855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong P, Iwasaki M, Somervaille TC, Ficara F, Carico C, Arnold C, et al. The miR-17-92 microRNA polycistron regulates MLL leukemia stem cell potential by modulating p21 expression. Cancer Res. 2010;70:3833–3842. doi: 10.1158/0008-5472.CAN-09-3268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivanovska I, Ball AS, Diaz RL, Magnus JF, Kibukawa M, Schelter JM, et al. MicroRNAs in the miR-106b family regulate p21/CDKN1A and promote cell cycle progression. Mol Cell Biol. 2008;28:2167–2174. doi: 10.1128/MCB.01977-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang B, Liu XX, He JR, Zhou CX, Guo M, He M, et al. Pathologically decreased miR-26a antagonizes apoptosis and facilitates carcinogenesis by targeting MTDH and EZH2 in breast cancer. Carcinogenesis. 2011;32:2–9. doi: 10.1093/carcin/bgq209. [DOI] [PubMed] [Google Scholar]
- Kim H, Huang W, Jiang X, Pennicooke B, Park PJ, Johnson MD. Integrative genome analysis reveals an oncomir/oncogene cluster regulating glioblastoma survivorship. Proc Natl Acad Sci USA. 2010;107:2183–2188. doi: 10.1073/pnas.0909896107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Viale A, De Franco F, Orleth A, Cambiaghi V, Giuliani V, Bossi D, et al. Cell-cycle restriction limits DNA damage and maintains self-renewal of leukaemia stem cells. Nature. 2009;457:51–56. doi: 10.1038/nature07618. [DOI] [PubMed] [Google Scholar]
- De Marchis ML, Ballarino M, Salvatori B, Puzzolo MC, Bozzoni I, Fatica A. A new molecular network comprising PU.1, interferon regulatory factor proteins and miR-342 stimulates ATRA-mediated granulocytic differentiation of Acute Promyelocitic Leukemia cells. Leukemia. 2009;23:856–862. doi: 10.1038/leu.2008.372. [DOI] [PubMed] [Google Scholar]
- Rosa A, Brivanlou AH. A regulatory circuitry comprised of miR-302 and the transcription factors OCT4 and NR2F2 regulates human embryonic stem cell differentiation. EMBO J. 2011;30:237–248. doi: 10.1038/emboj.2010.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.