Abstract
Excessive mitochondrial free radical production and the related mitogen-activated protein kinase P38 (P38 MAPK) activation are key regulators in the pathogenesis of high glucose-induced cell stress. Increasing evidence has emphasized the impact of hyperglycemia on neurons and the consequent neuronal stresses eventually resulting in neurodegeneration and neuronal death. In this study, we employed a novel mitochondria-targeted antioxidant, SS31 peptide, on high glucose-insulted neuroblastoma cells (SH-SY5Y). Our results showed that high glucose promoted significantly increased P38 phosphorylation which was efficiently suppressed by the application of the SS31 peptide under the experimental conditions. The inhibition of high glucose-induced P38 activation by the SS31 peptide was associated with the impact of the SS31 peptide on attenuating high glucose-induced mitochondrial ROS (reactive oxygen species) elevation and mitochondrial membrane potential collapse. The addition of SS31 peptide significantly attenuated high-gluose-induced apoptosis. Therefore, our study suggests that elimination of high glucose-induced mitochondrial oxidative stress helps to rescue SH-SY5Y cells from high glucose-related P38 MAPK pathway disturbances, and the SS31 peptide has the potential to serve as a new treatment strategy against hyperglycemia-instigated neuronal perturbations.
Keywords: high-glucose, SS31 peptide, antioxidant, mitogen-activated protein kinase P38, mitochondrial reactive oxygen species, neuroblastoma cell
Introduction
Dysregulated glucose metabolism underlies a variety of pathologies leading to impairments in multiple types of cells including neurons (1). Recently, emerging evidence has accentuated the impact of high blood glucose on neuronal function in vitro and in vivo suggesting the correlation between glucose dysmetabolism and diverse types of brain diseases including stroke and neurodegenerative diseases (1,2). Mitochondria are the main glucose-consuming sites in neurons and produce ATP by glycolysis through oxidation phosphorylation. Previous studies have revealed that high blood glucose substantially encumbers mitochondrial function and results in mitochondria-dependent apoptosis suggesting mitochondrial pathology stands in the nexus of glucose dysmetabolism and cell injuries (3,4). Among the many glucose dysmetabolism-induced mitochondrial dysfunctions, increased mitochondria oxidative stress is a predominant mitochondrial pathology and occurs early in response to high-glucose insult (5). Correspondingly, it has been widely reported that excessive reactive oxygen species (ROS) plays a key role in the pathogenesis of high glucose-associated pathological conditions such as neurodegeneration and diabetes (5,6). Oxidative stress is a strong inducer of the mitogen-activated protein kinase P38 (P38 MAPK) activation (7) and the activation of P38 MAPK signaling pathway is another prominent change in high glucose-exposed cells. The excessive activation of P38 MAPK is a critical mechanism underlying high glucose-induced cellular injuries eventually leading to apoptosis (8,9). Thus, elimination of high glucose-induced mitochondrial ROS may have the potential to attenuate P38 MAPK signaling disturbances.
The SS31 peptide (H-D-Arg-Dmt-Lys-Phe-NH2) is a novel cell permeable antioxidant specifically targeted to mitochondria and subsequently eliminates mitochondrial reactive oxygen species (ROS) and preserves ATP production against oxidizing insults (10,11). Yet, to the best of our knowledge, the effect of the SS31 peptide on P38 MAPK signaling perturbation in high glucose-insulted neurons has not yet been elucidated. In the present study, using a neuron-like cell line (human neuroblastoma SH-SY5Y) we demonstrated that the use of the SS31 peptide substantially suppressed high glucose-instigated mitochondrial oxidative stress and P38 MAPK activation in neuroblastoma cells which serves as evidence of the potential application of mitochondria-targeted antioxidants in the treatment of glucose dysmetabolism-induced neuronal stresses.
Materials and methods
Cell culture and treatment
Human neuroblastoma cells (SH-SY5Y) were obtained from the American Type Culture Collection (ATCC). Cells were cultured in DMEM (25 mM glucose, Gibco, USA) with 5% fetal bovine serum (Gibco), 2 mM L-glutamine, 100 U/ml penicillin in a 95% air, 5% CO2 atmosphere. Cells were starved 12 h before the experiments by replacing the medium with OptiMEM (Gibco) without fetal bovine serum, followed by treatment with D-glucose (Sigma) as vehicle (no additional glucose added) at indicated final concentrations. The SS31 peptide (Stealth Peptide International, China) at the indicated concentration was applied 30 min before the addition of glucose. Antimycin A (Sigma) treatment was conducted at 50 μM on cells for 2 h. Carbonylcyanide p-trifluoromethoxyphenylhydrazone (FCCP; Sigma) was used at 5 mM to treat cells for 90 min.
Intracellular and intra-mitochondrial ROS and mitochondrial membrane potential detection
Intracellular ROS was detected by using fluorescence probe 5-(and 6-) chloromethyl-20, 70-dichlorodihydrofluorescein diacetate (CM-DCF-DA; Invitrogen, USA) at 10 μM for 30 min, and intra-mitochondrial ROS was visualized by coincubation of MitoSox Red (Invitrogen) at 1 μM. To detect mitochondrial membrane potential, tetramethylrhodamine methyl ester (TMRM; Invitrogen) was used at 200 nM for 20 min followed by washing using culture medium. Cells, after appropriate staining were observed under a Zeiss Axiovert 200M inverted fluores,cence microscope with heating control and CO2 chamber. The intensity of fluorescent staining was calculated as (F1-F0). F1 is the intensity of the staining and F0 is the intensity of the background.
Western blotting
The proper amount of cell extracts was loaded and separated by SDS-PAGE and transferred to nitrocellulose membranes (Bio-Rad, USA). Immunoblotting was performed using antibodies against phospho P38 (pT180/pY182, BD; 1:1,000), P38 (Cell Signaling, USA; 1:3,000) or β-actin (Sigma; 1:5,000) overnight at 4˚C. Immunoreactive bands were visualized using the ECL system (Fisher Scientific, USA).
TUNEL assay
Cell apoptosis was measured by terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) method using the TUNEL Assay kit (Roche, Germany) following the manufacturer's instructions.
Statistics and data analysis
Statistical analysis was performed by one-way ANOVA and post-hoc ANOVA analysis when appropriate using SPSS software. The values are shown as mean ± SEM. Significance was defined as P<0.05. Analysis of immunoblotting and cell staining was conducted using NIH ImageJ software (1.62).
Results
High glucose induces P38 MAPK activation in SH-SY5Y cells in a dose-dependent manner
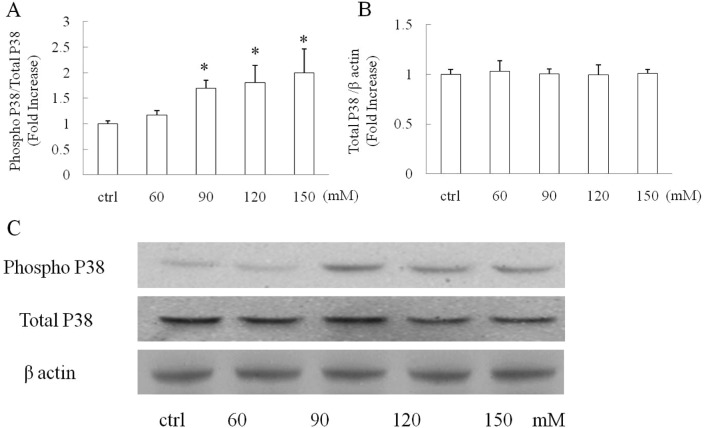
Cells were exposed to vehicle, 60, 90, 120 and 150 mM glucose for 90 min after starvation and then subjected to immunoblotting to detect the levels of phospho P38 and total P38 MAPK. β-actin was adopted as the loading control to verify that the same amount of proteins was loaded in each well. Our data showed that the P38 phosphorylation level was increased in a dose-dependent manner with increased glucose concentration. Glucose treatment (60 mM) induced a mild increase in P38 phosphorylation as compared with the vehicle group (Fig. 1A, P>0.05); whereas glucose at concentrations of 90 mM or higher significantly instigated P38 phosphorylation in the treated cells (Fig. 1A, P<0.05 vs. control group). In contrast, the level of total P38 was not changed in any group by comparison with β-actin suggesting that the increased P38 phosphorylation in the glucose treated-groups was not due to an increased level of total P38.
Figure 1.
Cells were exposed to glucose at the indicated concentrations for 90 min and then subjected to western blotting. The P38 phosphorylation level was significantly increased in cells exposed to (A) high concentration of glucose while (B) the total P38 level remained the same. (C) Representative immunoreactive bands of phospho P38, total P38 and β-actin. *P<0.05 vs. the control group. Data are derived from 3 independent experiments.
SS31 peptide protects mitochondrial membrane potential against high-glucose insult
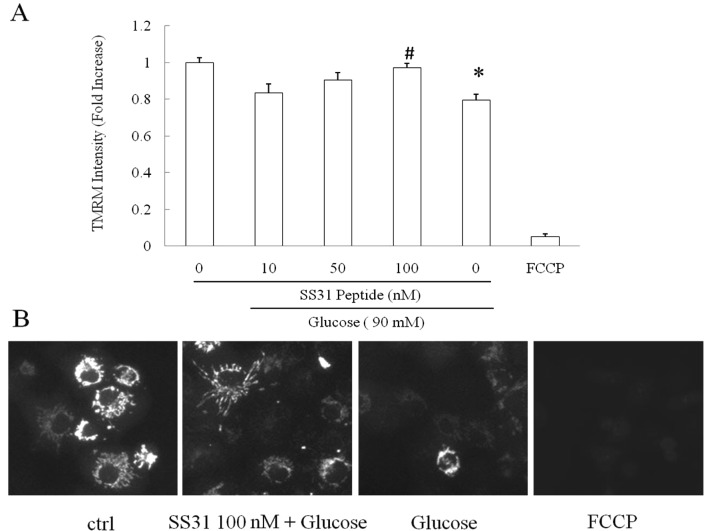
Mitochondrial membrane potential is a critical indicator of mitochondrial function, and decreased mitochondrial membrane potential is closely associated with elevated mitochondrial ROS production. Previous studies have shown that the SS31 peptide confers substantial protection on mitochondrial membrane potential against multiple insults (3,12,13). We next studied whether the SS31 peptide preserves mitochondrial membrane potential against high glucose in SH-SY5Y cells. FCCP, a strong mitochondrial uncoupler, was employed to treat cells at 5 mM as a positive control. High glucose substantially decreased mitochondrial membrane potential to 79.5% in comparison to the vehicle group (Fig. 2, P<0.0001); whereas the adoption of the SS31 peptide demonstrated a dose-dependent rescue of the high glucose-induced mitochondrial membrane potential collapse. Of note, the SS31 peptide at the concentration of 100 nM exhibited significant restoration of high glucose-insulted mitochondrial membrane potential decrease almost to the same level as that in the vehicle group (Fig. 2, vs. high glucose-treated group; P<0.0001); while the SS31 peptide at 50 nM also showed partial recovery as compare to the high glucose-treated group (Fig. 2A, P<0.05). FCCP-exposed cells demonstrated significantly lower mitochondrial membrane potential indicating the specificity of TMRM as a mitochondrial membrane potential indicator.
Figure 2.
SH-SY5Y cells were exposed to the SS31 peptide at 10, 50 and 100 nM for 30 min followed by the addition of glucose (final concentration 90 mM). FCCP was used as a positive control. (A) SS31 peptide significantly preserved mitochondrial membrane potential from high-glucose insult in a dose-dependent manner. *P<0.05 vs. control group and #P<0.05 vs. high-glucose group. (B) Representative images of TMRM. Data are derived from 3 independent experiments.
SS31 peptide suppresses high glucose-induced intracellular and intra-mitochondrial ROS production
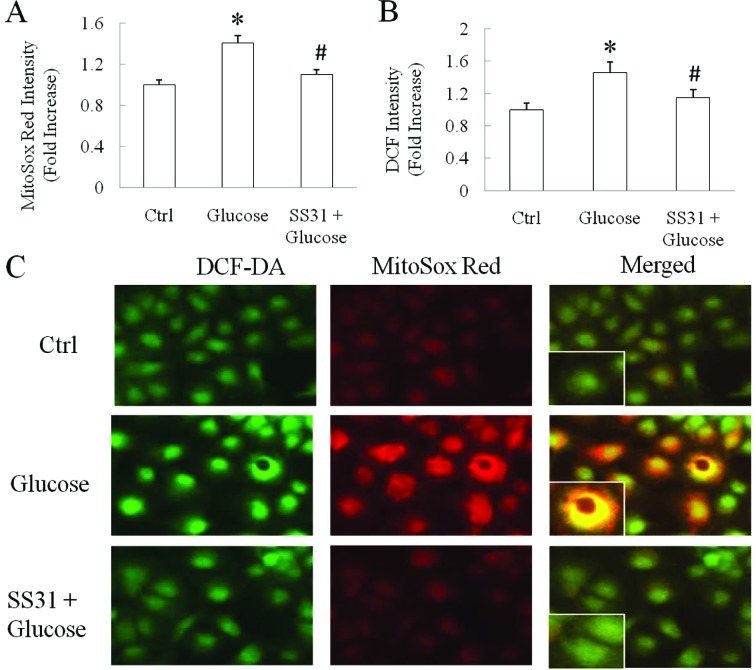
Mitochondrial membrane potential collapse is closely associated with mitochondrial ROS production. As the SS31 peptide at 100 nM significantly restored mitochondrial membrane potential in SH-SY5Y cells against high-glucose insult, we next observed the impact of the SS31 peptide at 100 nM on high glucose-induced cellular and mitochondrial ROS production. We compared the intracellular and intra-mitochondria ROS levels between the vehicle- and glucose-treated groups. Intracellular ROS was detected using DCF-DA and intra-mitochondrial ROS was shown as the increased intensity of MitoSox Red, a specific mitochondrial ROS indicator. The data showed that cells exposed to 90 mM glucose for 90 min underwent significantly increased intra-mitochondrial and intracellular ROS levels by 40.5% (Fig. 3A and C, P<0.0001) and 45.5% (Fig. 3B and C, P=0.0047), respectively, as compared with the vehicle group. In sharp contrast, the addition of the SS31 peptide significantly suppressed the high glucose-induced increase in intracellular and intra-mitochondrial ROS levels. In the SS31 peptide-treated group, the intra-mitochondrial ROS level was reduced by 28.0% (Fig. 3A and C, P=0.0008) while intracellular ROS level was decreased by 26.6% (Fig. 3B and C, P=0.0359) in comparison with the high glucose-treated group implicating the effect of SS31 peptide application on high glucose-instigated ROS generation.
Figure 3.
SH-SY5Y cells were exposed to SS31 peptide 100 nM for 30 min followed by 90 mM glucose for 90 min. The SS31 peptide significantly quenched the (A) mitochondrial ROS and (B) high glucose-induced cellular levels. (A) *P<0.0001 vs. control group and #P<0.05 vs. high-glucose group. (B) *P=0.0047 vs. control group and #P<0.05 vs. high-glucose group. (C) Representative images of DCF-DA (green) and MitoSox Red (red) staining in indicated groups. Inserted images are enlarged from the original images. Data are derived from 3 independent experiments and shown as fold increase.
Elevated mitochondrial ROS production instigates P38 MAPK activation in SH-SY5Y cells
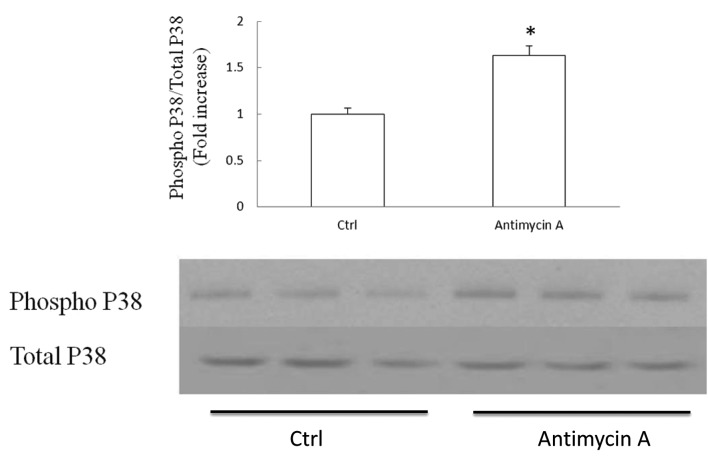
Oxidative stress is a known causative factor of P38 activation. Mitochondria are the major sites of ROS production. In order to examine whether P38 activation is related to mitochondrial ROS production in SH-SY5Y cells, we applied antimycin A to cultured cells. Antimycin A is the inhibitor of mitochondrial complex III and is a strong inducer of mitochondrial ROS production. Cells were exposed to antimycin A at 50 μM for 2 h and subjected to immunoblotting to detect the P38 phosphorylation level. The ratio of phospho P38 to total P38 was employed as the manifestation of P38 activation. Our data showed that antimycin A induced a 63% increase in P38 activation level in comparison to the control (Fig. 4, P<0.05). The result indicates that mitochondrial ROS production/release is associated with P38 activation in SH-SY5Y cells.
Figure 4.
SH-SY5Y cells were exposed to 50 μM antimycin A for 2 h and then subjected to immunoblotting for phospho P38 and total P38. Antimycin A induced a 63% increase in P38 activation as compared to the vehicle control (upper panel); *P<0.05. The lower panel shows representative immunoreactive bands. Data are derived from 3 independent experiments.
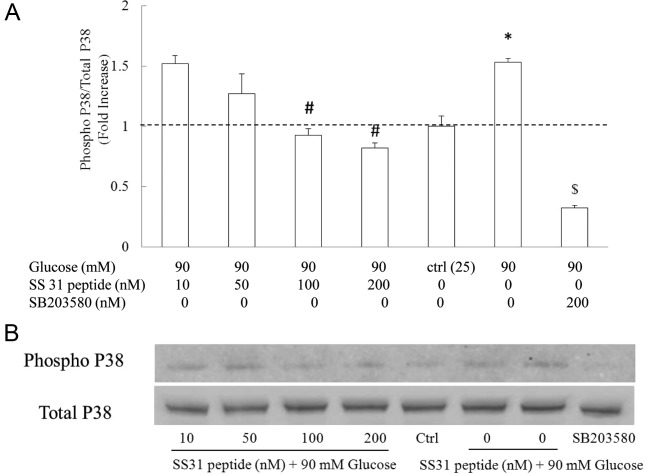
SS31 peptide inhibits high glucose-induced P38 phosphorylation in SH-SY5Y cells in a dose-dependent manner
Finally, we determined whether suppressed mitochondrial ROS subsequently inhibits high glucose-instigated P38 activation. We employed the SS31 peptide at 0, 10, 50 100 and 200 nM, respectively. Cells were exposed to the SS31 peptide at indicated concentrations for 30 min followed by incubation with glucose at 90 mM for 90 min. The coincubation of the SS31 peptide with high glucose markedly attenuated high glucose-induced P38 activation in a dose-dependent manner. As shown in Fig. 5, high-glucose treatment increased the P38 phosphorylation level to 2.33±0.14-fold as compared with the vehicle group (Fig. 5A, P<0.05). In sharp contrast, the application of the SS31 peptide at 100 and 200 nM substantially decreased P38 phosphorylation to a similar level in comparison with that in the vehicle control (P>0.05 vs. vehicle group), which was significantly lower than that in the high glucose-treated group (Fig. 5A, P<0.05). Although the SS31 peptide at 10 nM exhibited limited inhibition on high glucose-induced P38 activation, the SS31 peptide at 50 nM demonstrated partial protection on high glucose-induced P38 activation (Fig. 5A, P<0.05). The dose-dependent inhibition of the SS31 peptide on glucose-induced P38 activation was in correlation with the impact of the SS31 peptide on high glucose-instigated mitochondrial membrane potential and ROS production perturbations. The addition of P38 inhibitor SB203580 (200 nM) significantly suppressed high glucose-induced P38 activation (Fig. 5A, P<0.05 vs. high glucose-treated group). Thus, our data indicate a protective effect of the SS31 peptide on P38 activation in SH-SY5Y cells against high-glucose exposure.
Figure 5.
(A) SS31 peptide demonstrated a dose-dependent inhibition of high glucose-induced P38 activation. (B) Representative immunoreactive bands of phospho P38 and total P38. *P<0.05 vs. the control group and 50 nM SS31 peptide group; #P<0.05 vs. high glucose-treated group; $P<0.05 vs. all the other groups. Data are derived from 3 independent experiments.
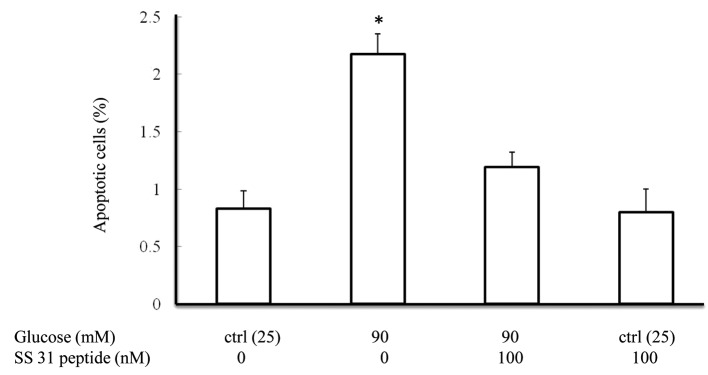
SS31 peptide attenuates high glucose-induced SH-SY5Y cell apoptosis
P38 activation has been implicated to be involved in apoptosis in response to cell injuries. We then assessed the protection of the SS31 peptide on apoptosis in the face of high-glucose insult. SH-SY5Y cells were exposed to 90 mM glucose in the presence or absence of 100 nM SS31 peptide for 24 h and then subjected to TUNEL assay. Our data showed that high glucose-induced a 2.6-fold increase in the percentage of apoptotic cells while the application of the SS31 peptide significantly ameliorated the high glucose-induced apoptosis (Fig. 6, P<0.05 vs. other groups). The SS31 peptide alone did not have a significant effect on cell apoptosis.
Figure 6.
SS31 peptide attenuates high glucose-induced apoptosis. Glucose (90 mM) induced increased apoptosis in SH-SY5Y cell after 24-h treatment which was significantly ameliorated by the SS31 peptide (100 nM). *P<0.05 vs. other groups.
Discussion
Hyperglycemia is a potential culprit underlying peripheral neuron changes in diabetes and neurodegeneration in the central nervous system (CNS) (6,14). The P38 MAPK signaling pathway plays a key role in controlling cell proliferation while excessive P38 activation leads to mitochondria-dependent apoptosis. It should be noted that P38 MAPK signaling activation is a predominant cell change in response to high-glucose insult and is significantly involved in high glucose-related cell injuries including decreased mitochondrial function and mitochondria-dependent apoptosis in the pathogenesis of hyperglycemia-associated diseases (9,15). Previous studies have shown that mitochondrial ROS is an initiator of P38 activation in cardiomyocytes and neurons under hypoxia and hyperglycemic conditions (16,17). The elimination of excessive cellular ROS has the potential to protect against high glucose-induced P38 activation. Indeed, various ROS scavengers such as VitE, SOD and lipid acid have been applied to treat hyperglycemia-associated cell stresses. However, the applications of these antioxidants are limited by multiple drawbacks such as high drug dose and various side effects (18–20). The SS31 peptide is a novel antioxidant specifically targeted to mitochondria and efficiently quenches mitochondrial ROS in many pathological conditions. Importantly, its relatively small-molecular weight and lipid solubility enable the peptide to rapidly penetrate cells (21). Our result showed that the addition of the SS31 peptide significantly reversed the high glucose-induced mitochondrial ROS elevation and mitochondrial membrane potential collapse. These findings are in agreement with many previous studies on the pharmaceutical effects of the SS31 peptide (10,13). Furthermore, by eliminating mitochondrial ROS and protecting mitochondrial membrane potential, P38 activation as well as apoptosis in high glucose-treated SH-SY5Y cells was substantially suppressed by the application of the SS31 peptide. The SS31 peptide protects cells from high-glucose insult. Notably, we adopted a neuron-like cell line, SH-SY5Y which demonstrates neuron biological features. To the best of our knowledge, to date, reports are limited on the effect of SS31 peptide against high glucose-induced mitochondrial ROS as well as neuronal dysfunctions. These results serve as important evidence for the application of this peptide in preventing neuronal stresses and injury related to hyperglycemia.
Collectively, we demonstrated that the nascent mitochondria-targeted antioxidant, SS31 peptide, substantially rescues high glucose-associated mitochondrial dysfunction and P38 activation in SH-SY5Y cells. This provides evidence that the SS31 peptide is a promising treatment strategy for rescuing neuronal function from hyperglycemia. Moreover, the small size of the SS31 peptide and its lipid solubility substantially enable the use of the SS31 peptide in treating hyperglycemia-instigated neuronal stresses in the CNS.
Acknowledgements
This study was supported by a grant from the Science Foundation of Shandong Province (Grant No. Q2006C15), China.
References
- 1.Das F, Dey N, Venkatesan B, Kasinath BS, Ghosh-Choudhury N, Choudhury GG. High-glucose upregulation of early-onset Parkinson's disease protein DJ-1 integrates the PRAS40/TORC1 axis to mesangial cell hypertrophy. Cell Signal. 2011;23:1311–1319. doi: 10.1016/j.cellsig.2011.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Matsuzaki T, Sasaki K, Tanizaki Y, et al. Insulin resistance is associated with the pathology of Alzheimer disease: the Hisayama study. Neurology. 2010;75:764–770. doi: 10.1212/WNL.0b013e3181eee25f. [DOI] [PubMed] [Google Scholar]
- 3.Li J, Chen X, Xiao W, et al. Mitochondria-targeted antioxidant peptide SS31 attenuates high-glucose-induced injury on human retinal endothelial cells. Biochem Biophys Res Commun. 2011;404:349–356. doi: 10.1016/j.bbrc.2010.11.122. [DOI] [PubMed] [Google Scholar]
- 4.Schiff M, Loublier S, Coulibaly A, Benit P, de Baulny HO, Rustin P. Mitochondria and diabetes mellitus: untangling a conflictive relationship? J Inher Metab Dis. 2009;32:684–698. doi: 10.1007/s10545-009-1263-0. [DOI] [PubMed] [Google Scholar]
- 5.Herlein JA, Fink BD, Sivitz WI. Superoxide production by mitochondria of insulin-sensitive tissues: mechanistic differences and effect of early diabetes. Metabolism. 2010;59:247–257. doi: 10.1016/j.metabol.2009.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang HL, Chou AH, Wu AS, et al. PARK6 PINK1 mutants are defective in maintaining mitochondrial membrane potential and inhibiting ROS formation of substantia nigra dopaminergic neurons. Biochimic Biophys Acta. 2011;1812:674–684. doi: 10.1016/j.bbadis.2011.03.007. [DOI] [PubMed] [Google Scholar]
- 7.Ravindran J, Gupta N, Agrawal M, Bala Bhaskar AS, Lakshmana Rao PV. Modulation of ROS/MAPK signaling pathways by okadaic acid leads to cell death via mitochondrial-mediated caspase-dependent mechanism. Apoptosis. 2011;16:145–161. doi: 10.1007/s10495-010-0554-0. [DOI] [PubMed] [Google Scholar]
- 8.Begum N, Ragolia L. High-glucose and insulin inhibit VSMC MKP-1 expression by blocking iNOS via P38 MAPK activation. Am J Physiol Cell Physiol. 2000;278:C81–C91. doi: 10.1152/ajpcell.2000.278.1.C81. [DOI] [PubMed] [Google Scholar]
- 9.Ren Y, Shi Y, Wang Y, et al. P38 MAPK pathway is involved in high-glucose-induced thioredoxin interacting protein induction in mouse mesangial cells. FEBS Lett. 2010;584:3480–3485. doi: 10.1016/j.febslet.2010.07.010. [DOI] [PubMed] [Google Scholar]
- 10.Carter EA, Bonab AA, Goverman J, et al. Evaluation of the antioxidant peptide SS31 for treatment of burn-induced insulin resistance. Int J Mol Med. 2011;28:589–594. doi: 10.3892/ijmm.2011.752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Reddy TP, Manczak M, Calkins MJ, et al. Toxicity of neurons treated with herbicides and neuroprotection by mitochondria-targeted antioxidant SS31. Inter J Environ Res Public Health. 2011;8:203–221. doi: 10.3390/ijerph8010203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Andersson DC, Fauconnier J, Yamada T, et al. Mitochondrial production of reactive oxygen species contributes to the beta-adrenergic stimulation of mouse cardiomycytes. J Physiol. 2011;589:1791–1801. doi: 10.1113/jphysiol.2010.202838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Manczak M, Mao P, Calkins MJ, et al. Mitochondria-targeted antioxidants protect against amyloid-beta toxicity in Alzheimer's disease neurons. J Alzheimers Dis. 2010;20(Suppl 2):S609–S631. doi: 10.3233/JAD-2010-100564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Toth C, Brussee V, Cheng C, Zochodne DW. Diabetes mellitus and the sensory neuron. J Neuropathol Exp Neurol. 2004;63:561–573. doi: 10.1093/jnen/63.6.561. [DOI] [PubMed] [Google Scholar]
- 15.Wilmer WA, Dixon CL, Hebert C. Chronic exposure of human mesangial cells to high-glucose environments activates the P38 MAPK pathway. Kidney Int. 2001;60:858–871. doi: 10.1046/j.1523-1755.2001.060003858.x. [DOI] [PubMed] [Google Scholar]
- 16.Kulisz A, Chen N, Chandel NS, Shao Z, Schumacker PT. Mitochondrial ROS initiate phosphorylation of p38 MAP kinase during hypoxia in cardiomyocytes. Am J Physiol Lung Cell Mol Physiol. 2002;282:L1324–L1329. doi: 10.1152/ajplung.00326.2001. [DOI] [PubMed] [Google Scholar]
- 17.Sharma R, Buras E, Terashima T, et al. Hyperglycemia induces oxidative stress and impairs axonal transport rates in mice. PloS One. 2010;5:e13463. doi: 10.1371/journal.pone.0013463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54:1615–1625. doi: 10.2337/diabetes.54.6.1615. [DOI] [PubMed] [Google Scholar]
- 19.Guerrero-Romero F, Rodriguez-Moran M. Complementary therapies for diabetes: the case for chromium, magnesium, and antioxidants. Arch Med Res. 2005;36:250–257. doi: 10.1016/j.arcmed.2005.01.004. [DOI] [PubMed] [Google Scholar]
- 20.Vincent AM, Russell JW, Low P, Feldman EL. Oxidative stress in the pathogenesis of diabetic neuropathy. Endocr Rev. 2004;25:612–628. doi: 10.1210/er.2003-0019. [DOI] [PubMed] [Google Scholar]
- 21.Cho S, Szeto HH, Kim E, Kim H, Tolhurst AT, Pinto JT. A novel cell-permeable antioxidant peptide, SS31, attenuates ischemic brain injury by down-regulating CD36. J Biol Chem. 2007;282:4634–4642. doi: 10.1074/jbc.M609388200. [DOI] [PubMed] [Google Scholar]