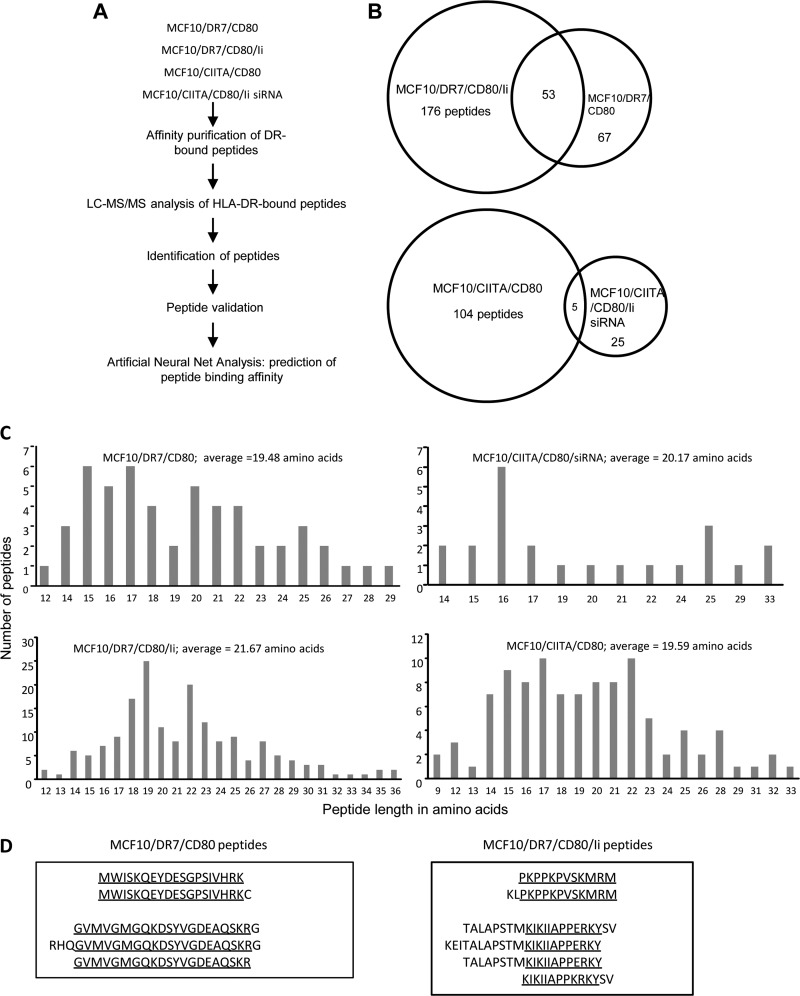
Fig. 2.
A, Experimental design for isolation and identification of HLA-DR-bound peptides. HLA-DR bound peptides from two independent preparations of DR+Ii− MCF10 vaccine cells and from DR+Ii+ MCF10 transfectants were purified using affinity chromatography, sequenced using LC-MS/MS, assigned to proteins using SEQUEST, and analyzed using ANN. Peptides with the highest predicted HLA-DR7 binding affinity were selected for further study. B, HLA-DR+ MCF10 vaccines and Ii+ transfectants present distinct and partially overlapping repertoires of peptides. Values are the number of peptides that are unique or shared between DR+Ii− MCF10 vaccine cells and DR+Ii+ MCF10 transfectants. C, Peptides isolated from HLA-DR7-transfected and CIITA-transfected Ii− and Ii+ MCF10 cells are similar in length. D, Peptides isolated from HLA-DR7-transfected Ii− and Ii+ MCF10 cells contain nested sequences. Data are from two independent analysis of each preparation of cells.