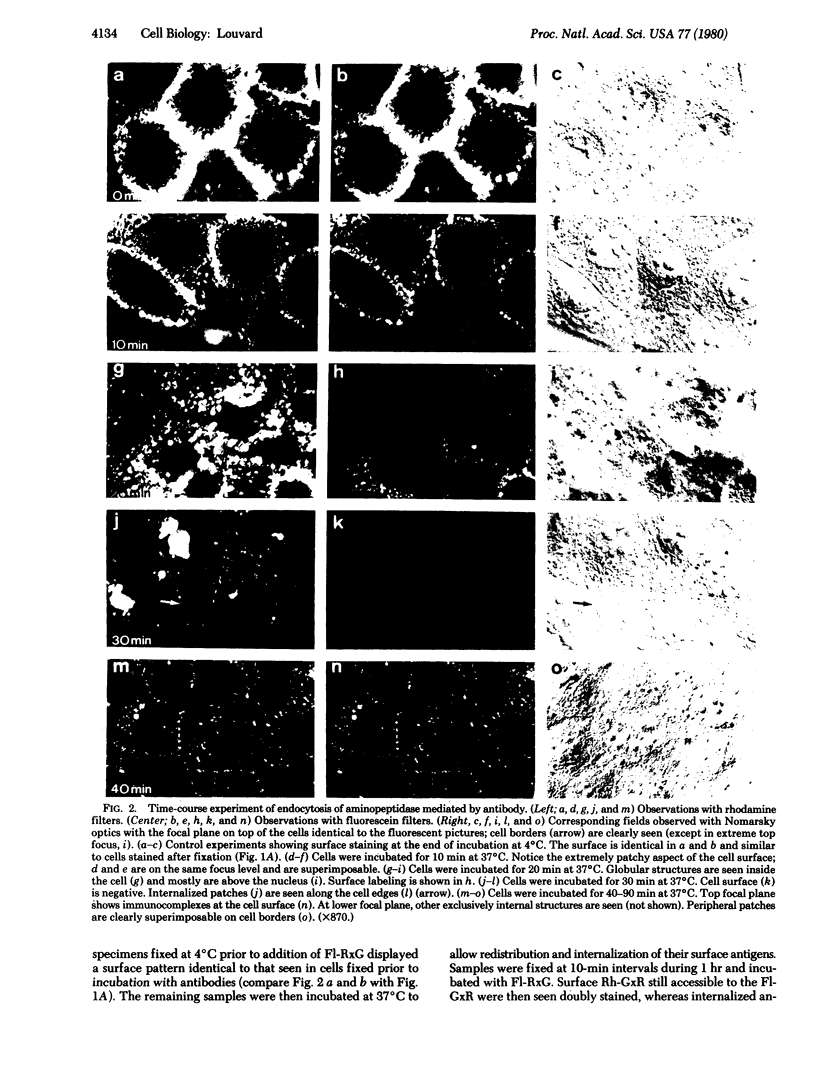
Abstract
A dog kidney epithelial cell line (MDCK), grown in monolayer, displayed in vitro an asymmetric localization of surface proteins. Aminopeptidase [alpha-aminoacylpeptide hydrolase (microsomal), EC 3.4.11.2] was found only in the apical face whereas Na+, K+-ATPase (ATP phosphohydrolase, EC 3.6.1.3) was found in the basolateral faces. These two faces are delineated by the junctional complex at which close cell-cell contact occurs. alpha-Actinin, a protein associated with plasma membranes, was concentrated near the region of cell-cell contact. When membrane proteins in the apical surface were crosslinked and subsequently removed from the surface by endocytosis, crosslinked antigens reappeared in the apical face at the region of cell-cell contact. Antigens that were not crosslinked were also (re)inserted in the same region. This process was not affected by cycloheximide, presumably because a large pool of apical membrane proteins (observed in small cytoplasmic vesicles) was used to replace the endocytosed antigens. It is psoposed that the region containing the junctional complex is involved in guiding apical membrane proteins to their final location.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ash J. F., Louvard D., Singer S. J. Antibody-induced linkages of plasma membrane proteins to intracellular actomyosin-containing filaments in cultured fibroblasts. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5584–5588. doi: 10.1073/pnas.74.12.5584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blakeslee D., Baines M. G. Immunofluorescence using dichlorotriazinylaminofluorescein (DTAF). I. Preparation and fractionation of labelled IgG. J Immunol Methods. 1976;13(3-4):305–320. doi: 10.1016/0022-1759(76)90078-8. [DOI] [PubMed] [Google Scholar]
- Brandtzaeg P. Conjugates of immunoglobulin G with different fluorochromes. I. Characterization by anionic-exchange chromatography. Scand J Immunol. 1973;2(3):273–290. doi: 10.1111/j.1365-3083.1973.tb02037.x. [DOI] [PubMed] [Google Scholar]
- Cereijido M., Ehrenfeld J., Meza I., Martínez-Palomo A. Structural and functional membrane polarity in cultured monolayers of MDCK cells. J Membr Biol. 1980;52(2):147–159. doi: 10.1007/BF01869120. [DOI] [PubMed] [Google Scholar]
- Cereijido M., Robbins E. S., Dolan W. J., Rotunno C. A., Sabatini D. D. Polarized monolayers formed by epithelial cells on a permeable and translucent support. J Cell Biol. 1978 Jun;77(3):853–880. doi: 10.1083/jcb.77.3.853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craig S. W., Pardo J. V. alpha-Actinin localization in the junctional complex of intestinal epithelial cells. J Cell Biol. 1979 Jan;80(1):203–210. doi: 10.1083/jcb.80.1.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujita M., Kawai K., Asano S., Nakao M. Protein components of two different regions of an intestinal epithelial cell membrane. Regional singularities. Biochim Biophys Acta. 1973 Apr 25;307(1):141–151. doi: 10.1016/0005-2736(73)90032-1. [DOI] [PubMed] [Google Scholar]
- Fujita M., Ota H., Kawai K., Matsui H., Nakao M. Differential isolation of microvillous and basolateral plasma membranes from intestinal mucosa: mutually exclusive distribution of digestive enzymes and ouabain-sensitive ATPase. Biochim Biophys Acta. 1972 Aug 9;274(2):336–347. doi: 10.1016/0005-2736(72)90181-2. [DOI] [PubMed] [Google Scholar]
- Geiger B. A 130K protein from chicken gizzard: its localization at the termini of microfilament bundles in cultured chicken cells. Cell. 1979 Sep;18(1):193–205. doi: 10.1016/0092-8674(79)90368-4. [DOI] [PubMed] [Google Scholar]
- Geiger B., Singer S. J. The participation of alpha-actinin in the capping of cell membrane components. Cell. 1979 Jan;16(1):213–222. doi: 10.1016/0092-8674(79)90202-2. [DOI] [PubMed] [Google Scholar]
- Geiger B., Tokuyasu K. T., Singer S. J. Immunocytochemical localization of alpha-actinin in intestinal epithelial cells. Proc Natl Acad Sci U S A. 1979 Jun;76(6):2833–2837. doi: 10.1073/pnas.76.6.2833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heggeness M. H., Ash J. F., Singer S. J. Transmembrane linkage of fibronectin to intracellular actin-containing filaments in cultured human fibroblasts. Ann N Y Acad Sci. 1978 Jun 20;312:414–417. doi: 10.1111/j.1749-6632.1978.tb16822.x. [DOI] [PubMed] [Google Scholar]
- Heggeness M. H., Ash J. F. Use of the avidin-biotin complex for the localization of actin and myosin with fluorescence microscopy. J Cell Biol. 1977 Jun;73(3):783–788. doi: 10.1083/jcb.73.3.783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hynes R. O., Destree A. T. Relationships between fibronectin (LETS protein) and actin. Cell. 1978 Nov;15(3):875–886. doi: 10.1016/0092-8674(78)90272-6. [DOI] [PubMed] [Google Scholar]
- Kyte J. Immunoferritin determination of the distribution of (Na+ + K+) ATPase over the plasma membranes of renal convoluted tubules. II. Proximal segment. J Cell Biol. 1976 Feb;68(2):304–318. doi: 10.1083/jcb.68.2.304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyte J. Purification of the sodium- and potassium-dependent adenosine triphosphatase from canine renal medulla. J Biol Chem. 1971 Jul 10;246(13):4157–4165. [PubMed] [Google Scholar]
- Lazarides E., Burridge K. Alpha-actinin: immunofluorescent localization of a muscle structural protein in nonmuscle cells. Cell. 1975 Nov;6(3):289–298. doi: 10.1016/0092-8674(75)90180-4. [DOI] [PubMed] [Google Scholar]
- Louvard D., Maroux S., Baratti J., Desnuelle P., Mutaftschiev S. On the preparation and some properties of closed membrane vesicles from hog duodenal and jejunal brush border. Biochim Biophys Acta. 1973 Feb 16;291(3):747–763. doi: 10.1016/0005-2736(73)90478-1. [DOI] [PubMed] [Google Scholar]
- MARCUS P. I. Dynamics of surface modification in myxovirus-infected cells. Cold Spring Harb Symp Quant Biol. 1962;27:351–365. doi: 10.1101/sqb.1962.027.001.033. [DOI] [PubMed] [Google Scholar]
- Misfeldt D. S., Hamamoto S. T., Pitelka D. R. Transepithelial transport in cell culture. Proc Natl Acad Sci U S A. 1976 Apr;73(4):1212–1216. doi: 10.1073/pnas.73.4.1212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pisam M., Ripoche P. Redistribution of surface macromolecules in dissociated epithelial cells. J Cell Biol. 1976 Dec;71(3):907–920. doi: 10.1083/jcb.71.3.907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson J. C., Simmons N. L. Demonstration of protein asymmetries in the plasma membrane of cultured renal (MDCK) epithelial cells by lactoperoxidase-mediated iodination. FEBS Lett. 1979 Sep 15;105(2):201–204. doi: 10.1016/0014-5793(79)80611-0. [DOI] [PubMed] [Google Scholar]
- Rodriguez Boulan E., Sabatini D. D. Asymmetric budding of viruses in epithelial monlayers: a model system for study of epithelial polarity. Proc Natl Acad Sci U S A. 1978 Oct;75(10):5071–5075. doi: 10.1073/pnas.75.10.5071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider Y. J., Tulkens P., de Duve C., Trouet A. Fate of plasma membrane during endocytosis. I. Uptake and processing of anti-plasma membrane and control immunoglobulins by cultured fibroblasts. J Cell Biol. 1979 Aug;82(2):449–465. doi: 10.1083/jcb.82.2.449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokuyasu K. T., Singer S. J. Improved procedures for immunoferritin labeling of ultrathin frozen sections. J Cell Biol. 1976 Dec;71(3):894–906. doi: 10.1083/jcb.71.3.894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vannier C., Louvard D., Maroux S., Desnuelle P. Structural and topological homology between porcine intestinal and renal brush border aminopeptidase. Biochim Biophys Acta. 1976 Nov 11;455(1):185–199. doi: 10.1016/0005-2736(76)90163-2. [DOI] [PubMed] [Google Scholar]