Abstract
Clinical studies reported hypomagnesaemia in long-term omeprazole usage that was probably due to intestinal Mg2+ wasting. Our previous report demonstrated the inhibitory effect of omeprazole on passive Mg2+ transport across Caco-2 monolayers. The present study aimed to identify the underlying mechanism of omeprazole suppression of passive Mg2+ absorption. By using Caco-2 monolayers, we demonstrated a potent inhibitory effect of omeprazole on passive Mg2+, but not Ca2+, transport across Caco-2 monolayers. Omeprazole shifted the %maximum passive Mg2+ transport-Mg2+ concentration curves to the right, and increased the half maximal effective concentration of those dose-response curves, indicating a lower Mg2+ affinity of the paracellular channel. By continually monitoring the apical pH, we showed that omeprazole suppressed apical acid accumulation. Neomycin and spermine had no effect on passive Mg2+ transport of either control or omeprazole treated monolayers, indicating that omeprazole suppressed passive Mg2+ transport in a calcium sensing receptor (CaSR)-independent manner. The results of western blot analysis showed that omeprazole significantly suppressed claudin (Cldn)-7 and -12, but not Cldn-2, expression in Caco-2 cells. By using apical solution of pH 5.5, 6.0, 6.5, and 7.0, we found that apical acidity markedly increased passive Mg2+ transport, Mg2+ affinity of the paracellular channel, and Cldn-7 and -12 expression in Caco-2 monolayers. Apical acidity abolished the inhibitory effect of omeprazole on passive Mg2+ transport and Cldn-7 and -12 expression. Our results provided the evidence for the regulation of intestinal passive Mg2+ absorption by luminal acidity-induced increase in Cldn-7 and -12 expression.
Keywords: claudins, intestines, magnesium, omeprazole, proton pump inhibitors, tight junctions
Introduction
Magnesium (Mg2+) is an essential co-factor of numerous enzymatic reactions in the human body. Normal Mg2+ homeostasis is tightly regulated by the harmonious function of the intestinal absorption and renal excretion (Konrad et al., 2004; Quamme, 2008). Since dietary intake is the sole source of Mg2+ in humans, intestinal absorption plays a vital role in the regulation of normal Mg2+ balance. Different intestinal segments contribute unequally to Mg2+ absorption. In percentage of the total input, duodenum absorbs 11%, jejunum 22%, ileum 56%, and large intestine 11% (McCarthy and Kumar, 1999). Paracellular passive Mg2+ transport is the major mechanism responsible for approximately 90% of total intestinal Mg2+ absorption (Quamme, 2008).
There have previously been a number of case reports of severe hypomagnesemia in patients with long-term use of proton pump inhibitor (PPI) that probably resulted from intestinal Mg2+ wasting (Epstein et al., 2006; Cundy and Dissanayake, 2008; Shabajee et al., 2008; Broeren et al., 2009; Kuipers et al., 2009; Hoorn et al., 2010; Mackay and Bladon, 2010). Recently, we reported an inhibitory effect of the popular PPI omeprazole on the paracellular passive, but not active, Mg2+ transport in human enterocyte-like Caco-2 monolayers (Thongon and Krishnamra, 2011). However the underlying cellular mechanism of this omeprazole action on paracellular channel still had not been elucidated.
The acidic environment of the intestinal lumen is known to have effects on the secretory and absorption activities of the intestine. The luminal acidity along the entire human small bowel varied between pH 5.5-7.0 (Nugent et al., 2001). Heijnen and coworkers (1993) reported that luminal acidic pH increased Mg2+ absorption in the ileum. Recently, elevation of extracellular pH was found to increase the sensitivity of CaSR (Quinn et al., 2004; Doroszewicz et al., 2005), and the activation CaSR in turn, suppressed paracellular passive Mg2+ transport (Ikari et al., 2008). Thus, omeprazole suppressed paracellular passive Mg2+ absorption at least in part by modulating the luminal acidity and CaSR sensitivity (Thongon and Krishnamra, 2011).
Claudins (Cldn) are the integral membrane proteins of the tight junction with ~23 members which were expressed in a tissue specific manner (Furuse and Tsukita, 2006). Monomeric and heteromeric combination of Cldn in each epithelium created unique paracellular channel for paracellular ion transport (Tang and Goodenough, 2003; Krause et al., 2008). Cldn-2 and -12 were reported to act as paracellular channels for divalent cation i.e., Ca2+ (Fujita et al., 2008). In addition, paracellular Cldn-7 channel was highly permeable to Na+, but not Cl- (Alexandre et al., 2005). Our previous finding reported that omeprazole inhibited paracellular Mg2+ and Na+ channels (Thongon and Krishnamra, 2011). Therefore, omeprazole probably disturbed the expression and/or function of Cldn-2, -7, and -12, which were highly expressed in the jejunum, ileum, and Caco-2 cells (Fujita et al., 2006, 2008). The present study, therefore, aimed to identify the underlying mechanism of the omeprazole-suppressed intestinal paracellular Mg2+ absorption. We hypothesized that omeprazole disturbed luminal acidity and increased CaSR activity. The activation of CaSR suppressed of paracellular Mg2+ channel that formed by Cldn in intestinal epithelium. Experiments were performed in Caco-2 monolayer, which was accepted as a suitable intestinal model for studying Mg2+ transport (Ekmekcioglu et al., 2000; Thongon and Krishnamra, 2011).
Results
Omeprazole decreased passive Mg2+ transport across Caco-2 monolayers
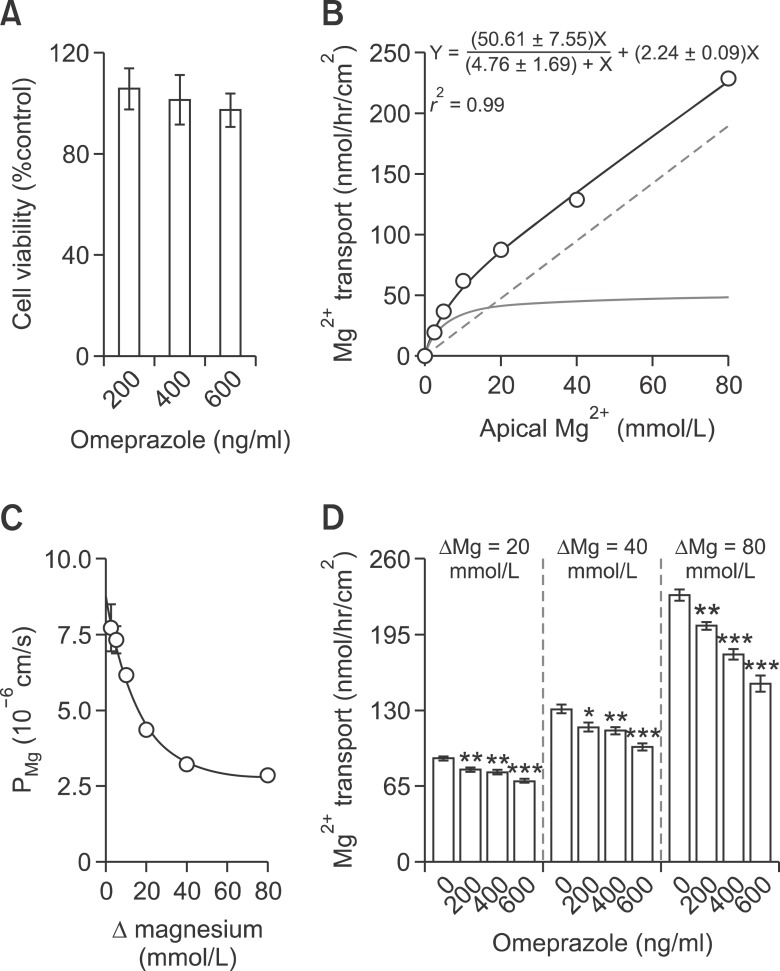
Since omeprazole was reported to have a potent apoptotic effect on human polymorphonuclear leucocytes (Capodicasa et al., 2008), cell viability assay was performed by incubating Caco-2 cells with the omeprazole at concentrations resembling those found in the human plasma of, i.e., 200 and 400 ng/ml (Macek et al., 2007), as well as a supra-physiological concentration of 600 ng/ml. As demonstrated in Figure 1A, neither 200, 400, nor 600 ng/ml omeprazole had cytotoxic effect on Caco-2 cells. Figures 1B and 1C depicted the apical to basolateral Mg2+ transport and absolute Mg2+ permeability (PMg) of Caco-2 monolayers (Figures 1B and 1C) in the present transwell transporting setup which were comparable to those of previous modified Ussing chamber set up (Thongon and Krishmanra, 2011). Omeprazole significantly suppressed the non-saturable passive Mg2+ transport in the presence of 20, 40, and 80 mmol/L (Figure 1D) Mg2+ concentration gradients. Therefore, the Mg2+ flux setup used in the present study was suitable for studying intestinal Mg2+ absorption.
Figure 1.
Omeprazole suppressed passive Mg2+ transport across Caco-2 monolayers. Relative cell viability of omeprazole - treated cells (A). Apical to basolateral Mg2+ transport of control monolayers (B). Light solid line represent the saturable active component. Dash line represent the non-saturable passive component. Paracellular PMg of control monolayers (C). Δ represent Mg2+ concentration gradient of apical and basolateral solution. Paracellular passive Mg2+ transport under 20, 40, or 80 (D) mmol/L Mg2+ concentration gradient. *P < 0.05, **P < 0.01, ***P < 0.001 vs control group. For each data point, n = 6.
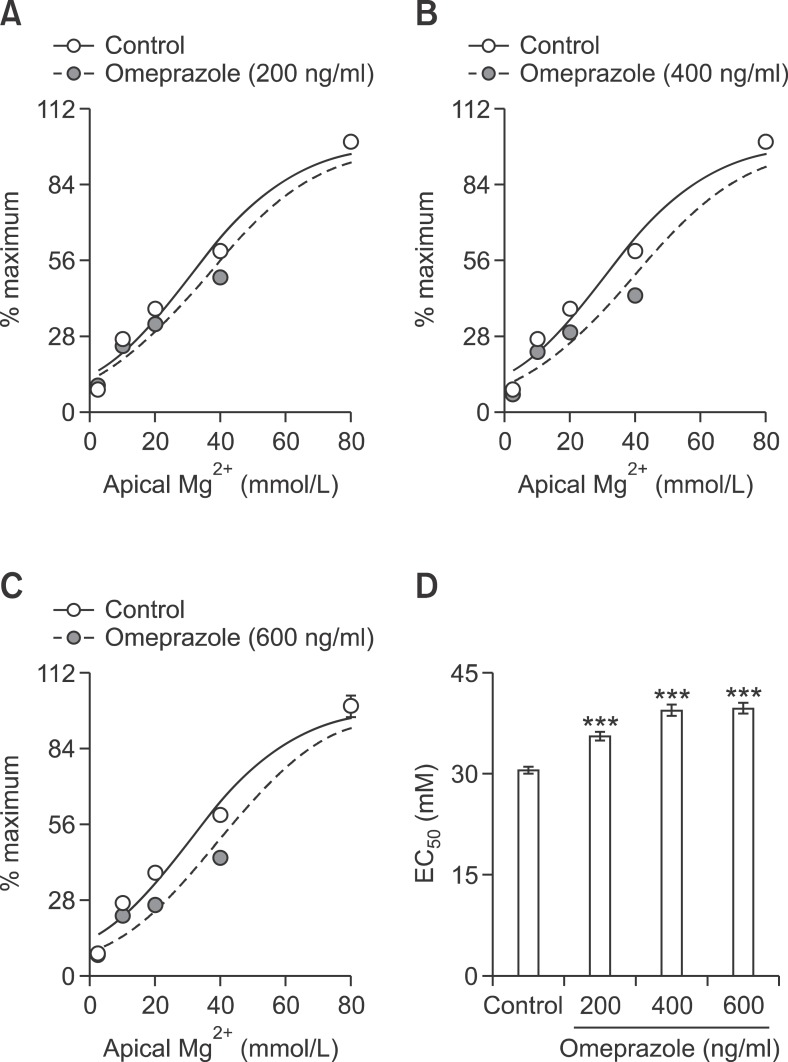
The relationships of percent change of passive Mg2+ transport and apical Mg2+ concentration were compared among control and omeprazole treated groups (Figures 2A-2C). Omeprazole shifted the %change - apical Mg2+ concentration curves to the right. Omeprazole at 200, 400, and 600 ng/ml significantly increased the EC50 from 30.54 ± 1.13 to 35.54 ± 1.41, 39.39 ± 1.83, and 39.74 ± 1.84 mmol/L, respectively (Figure 2D). These results indicated that omeprazole decreased affinity of paracellular channel for Mg2+.
Figure 2.
Omeprazole decreased Mg2+ affinity of the paracellular channels of Caco-2 monolayers. %maximum passive Mg2+ transport - Mg2+ concentration curve of control and with omeprazole at 200 (A), 400 (B), or 600 (C) ng/ml. EC50 of the dose - response curve of control and omeprazole trated monolayers (D). ***P < 0.001 vs control group. For each data point, n = 6.
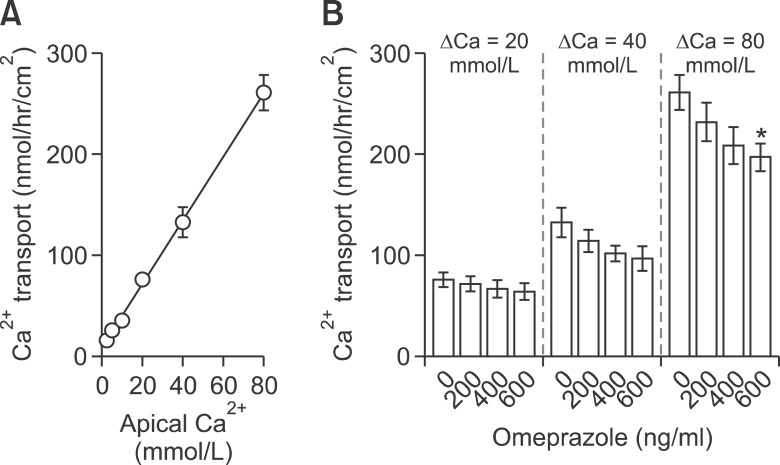
Since passive transport of Mg2+ and Ca2+ probably involved similar tight junction-associated proteins, the effect of omeprazole on passive Ca2+ transport across Caco-2 monolayers was also examined. The apical Ca2+ concentration - Ca2+ transport showed a liner relationship (Y = (3.16 ± 0.14)X + (8.47 ± 5.34), r2 = 0.94) (Figure 3A). In the presence of 20 and 40 mmol/L Ca2+ concentration gradients (Figure 3B), omeprazole showed the tendency to decrease Ca2+ transport, but the reductions were not statistically significant. Only at Ca2+ concentration gradient of 80 mmol/L with the concentration of 600 ng/ml that omeprazole significantly decreased passive Ca2+ transport. These results indicated that omeprazole had less inhibitory effect on passive Ca2+ transport than on passive Mg2+ absorption.
Figure 3.
Omeprazole had less effect on the paracellular passive Ca2+ transport across Caco-2 monolayers. Passive Ca2+ transport of control monolayers (A). Paracellular passive Ca2+ transport under 20, 40, or 80 (B) mmol/L Ca2+ concentration gradients. ΔCa represent Ca2+ concentration gradient of apical and basolateral solution. *P < 0.05 vs control group. For each data point, n = 10.
Omeprazole disturbed apical acidity of Caco-2 monolayers
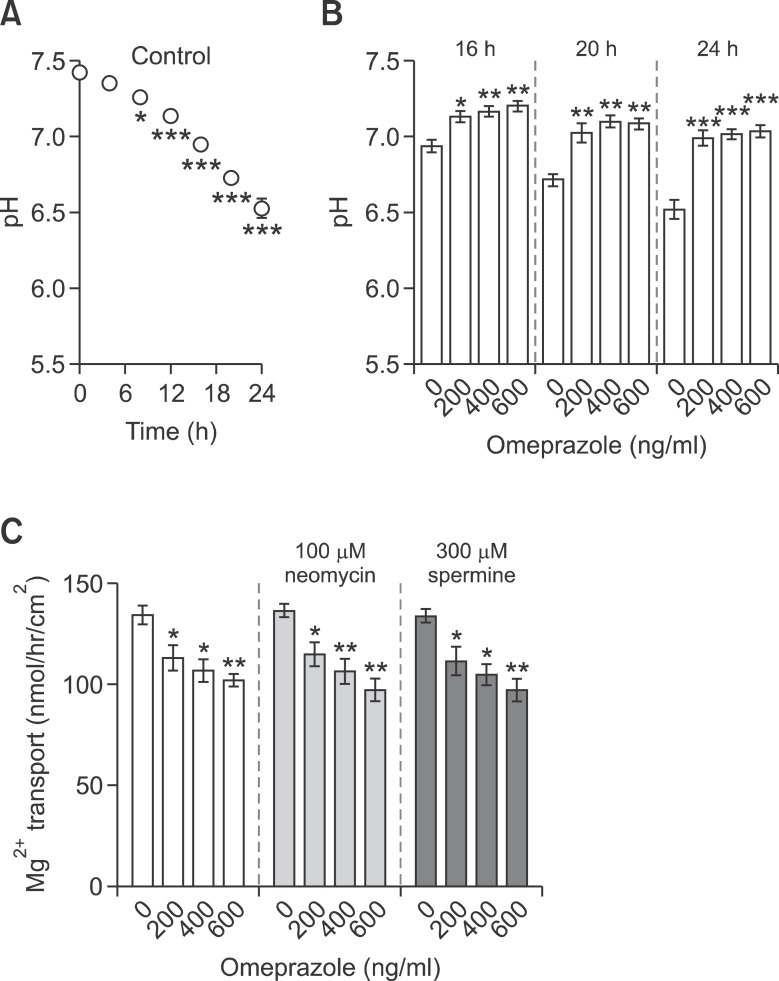
Since luminal pH could modulate intestinal Mg2+ absorption (Heijnen et al., 1993), the pH of apical culture media of control and omeprazole-treated monolayers were continually measured for 24 h after culture media change. In the control condition, apical pH significantly decreased from 8 h to 24 h after media change (Figure 4A). These results indicated the apical acid secretion and accumulation that was probably due to the activity of the proton pump in Caco-2 monolayers (Abrahamse et al., 1992). Omeprazole at 200, 400, and 600 ng/ml clearly raised the apical pH at 16 h, 20 h, and 24 h (Figure 4B), suggesting that a potent PPI omeprazole could suppress apical acid secretion by Caco-2 monolayers.
Figure 4.
Omeprazole disturbed apical acidity of Caco-2 monolayers. Accumulative apical pH values at various time points after replacing media with fresh one for 14d control cells (A). Apical pH of the media of control and omeprazole exposed Caco-2 cells at 16 h, 20 h, and 24 h (B) after media change. Passive Mg2+ transport (40 mmol/L concentration gradient) of control and omeprazole-treated groups that were incubated with CaSR agonists neomycin or spermine (C). *P < 0.05, **P < 0.01, ***P < 0.001 vs control group. For each data point, n = 6.
Omeprazole suppressed passive Mg2+ transport by a CaSR-independent mechanism
Since an increase in the extracellular pH could enhance CaSR sensitivity (Quinn et al., 2004; Doroszewicz et al., 2005), and activation of CaSR in turn, decreased paracellular Mg2+ transport (Ikari et al., 2008), possible involvement of CaSR in passive Mg2+ transport was studied by using the polycationic CaSR agonists neomycin and spermine (Sigma) (Ye et al., 1997). As demonstrated in Figure 4C, neither 100 µM neomycin nor 300 µM spermine affected passive Mg2+ transport in control and omeprazole-treated monolayers. These results indicated that omeprazole-suppressed passive Mg2+ transport was CaSR - independent.
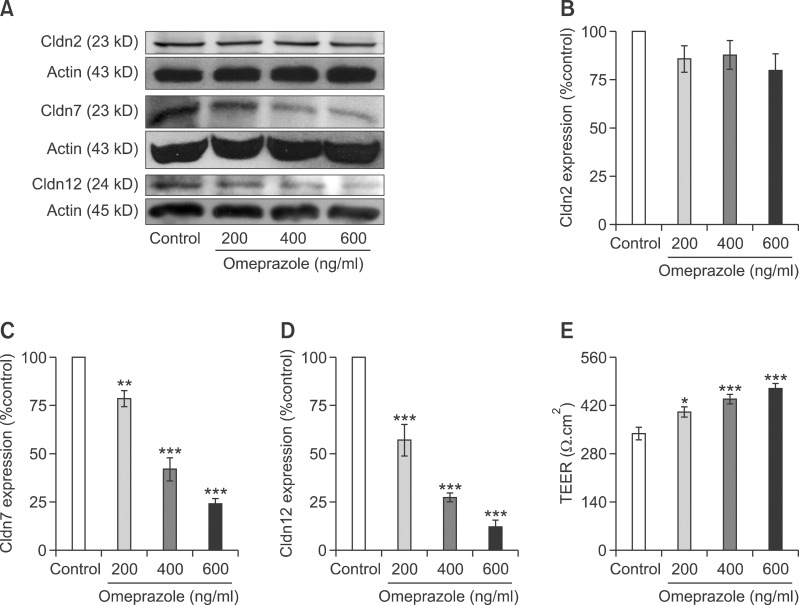
Omeprazole suppressed Cldn-7 and -12 expression
It was widely accepted that paracellular channel Cldn regulated paracellular passive ion transport across the epithelium (Tang and Goodenough, 2003; Krause et al., 2008). Therefore, the expressions of Cldn-2, -7, and -12 that was highly expressed in the jejunum, ileum, and Caco-2 cells (Fujita et al., 2006, 2008) were studied. The results of immunoblot and densitometric analysis demonstrated that omeprazole significantly reduced the expression of Cldn-7 (Figures 5A and 5C) and -12 (Figures 5A and 5D), but not of Cldn-2 (Figures 5A and 5B), in a concentration-dependent manner. Since the transepithelial electrical resistance (TEER) inversely related to the paracellular cation conductances (Tang and Goodenough, 2003; Thongon and Krishnamra, 2011), TEER of control and omeprazole-exposed monolayers were observed. As demonstrated in Figure 5E, omeprazole significantly increased TEER of Caco-2 monolayers.
Figure 5.
Omeprazole decreased Cldn-7 and -12 expression. The quantitative immunobloting analysis of Cldn-2, -7, and -12 in control and omeprazole-exposed Caco-2 cells (A). Representative densitometric analysis of Cldn-2 (B), -7 (C), and -12 (D) expression in control and omeprazole exposed cells. Trans-epithelial electrical resistance of control and omeprazole-exposed Caco-2 monolayers (E). *P < 0.05, **P < 0.01, ***P < 0.001 vs control group. For each data point, n = 5.
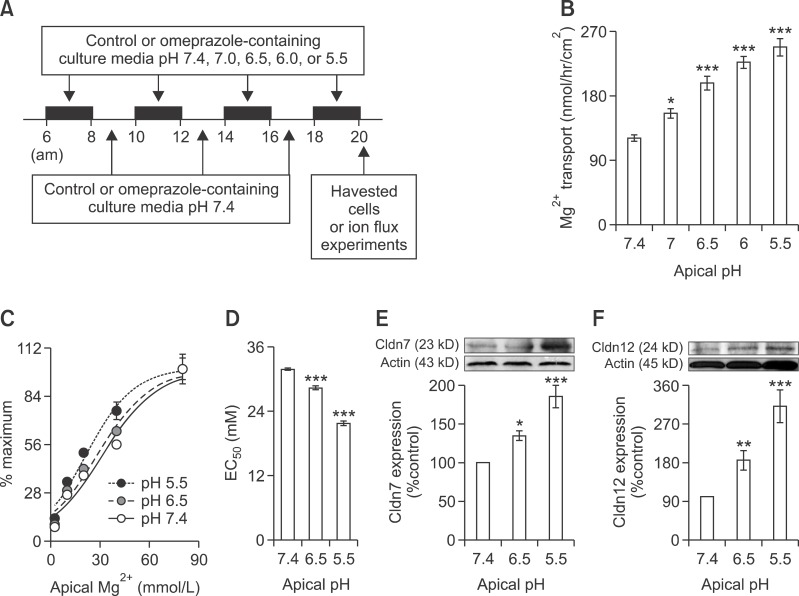
Apical acidity enhanced passive Mg2+ transport and Cldn-7 and -12 expression
To study the effect of apical acidity on the intestinal Mg2+ absorption and Cldn expression, Caco-2 monolayers were exposed for 2 h, 4 times a day to culture media with pH 7.4, 7.0, 6.5, 6.0, or 5.5 (Figure 6A) from day 7 to day 14 after seeding. In the control condition, apical acidity significantly increased passive Mg2+ transport at 40 mmol/L Mg2+ concentration gradient (Figure 6B). The relationship of percent change of passive Mg2+ transport and apical Mg2+ concentration in control condition at various apical pH was shown (Figure 6C). Apical acidic pH of 6.5 and 5.5 markedly shifted the relationship curve to the left (Figure 6C). Apical acidity of 6.5 and 5.5 decreased EC50 from 31.78 ± 1.11 at pH 7.4 to 28.32 ± 1.55 and 21.69 ± 1.80 mmol/L, respectively (Figure 6D). In addition, apical acidity significantly increased Cldn-7 (Figure 6E) and -12 (Figure 6F) expression in Caco-2 cells.
Figure 6.
Apical acidity enhanced passive Mg2+ absorption and Cld-7 and -12 expression. Representative time line for apical acidic media treatment (A). Passive Mg2+ transport (40 mmol/L concentration gradient) of control monolayers (B). %maximum passive Mg2+ transport - Mg2+ concentration curve of control monolayers at pH 7.4, 6.5, and 5.5 (C). EC50 of the dose - response curve of control monolayers at pH 7.4, 6.5, and 5.5 (D). For each data point, n = 6. Immunobloting and densitometric analysis of Cldn-7 (E) and -12 (F). For each data point, n = 5. *P < 0.05, **P < 0.01, ***P < 0.001 vs control group (n = 5).
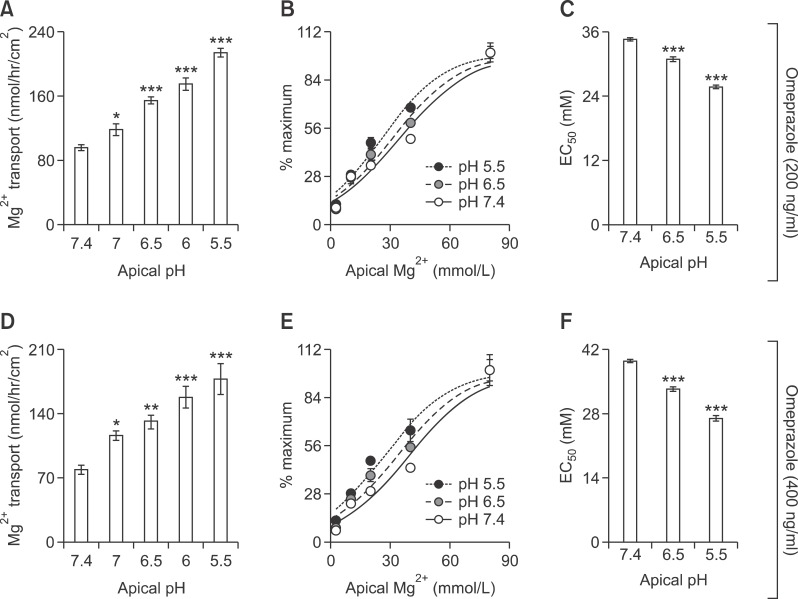
Apical acidity restored omeprazole effect on Caco-2 monolayers
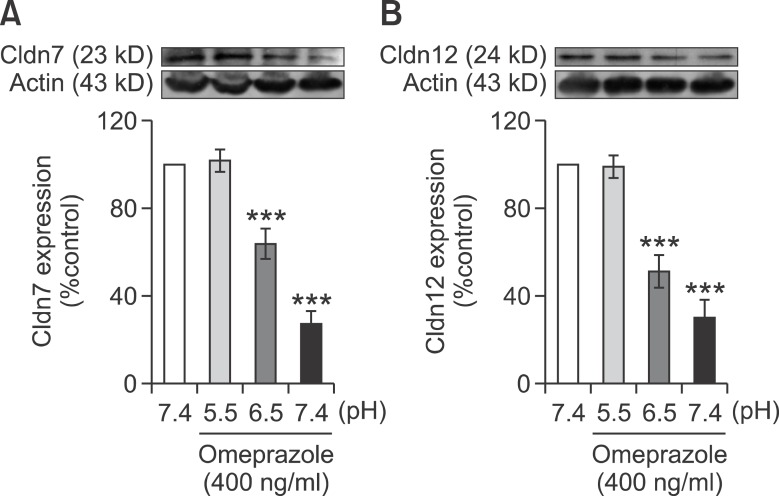
Since apical acidity enhanced passive Mg2+ transport as well as the expression of Cldn-7 and -12 in control Caco-2 monolayers (Figure 6), this experiment aimed to elucidate the effect of low apical pH on Mg2+ transport and Cldn expression in omeprazole-exposed monolayers. Similar to the control condition, apical acidity markedly increased the paracellular passive Mg2+ transport (40 mmol/L concentration gradient) of both 200 (Figure 7A) and 400 ng/ml omeprazole treated monolayers (Figure 7D). In the presence of 40 mmol/L Mg2+ concentration gradient, the rate of passive Mg2+ transport of 200 ng/ml omeprazole-treated monolayers at pH 7.0 (118 ± 7.25, Figure 7A), of 400 ng/ml omeprazole-treated monolayers pH 7.0 (116.60 ± 5.12, Figure 7D), and pH 6.5 (132.30 ± 6.01, Figure 7D) were not significantly different from that of control monolayers at pH 7.4 (132.20 ± 3.96, Figure 1E). These results indicated that apical acidity could restore the inhibitory effect of omeprazole on passive Mg2+ absorption. Apical acidity also shifted the dose-response curve of 200 (Figure 7B) and 400 ng/ml omeprazole (Figure 7E) treated group to the left. The EC50 of of 200 (Figure 7C) and 400 ng/ml (Figure 7F) omeprazole treated monolayers also decreased with the increase in acidity. At apical pH 6.5, EC50 of 200 (30.99 ± 1.67 mmol/L, Figure 7B) and 400 ng/ml omeprazole (33.30 ± 1.99 mmol/L, Figure 7E) were similar to that of control monolayers at pH 7.4 (Figure 6D). In addition to the stimulatory effect on Mg2+ flux, apical acidity also abolished the effect of omeprazole and normalized Cldn-7 and -12 expression in 400 ng/ml omeprazole treated monolayers (Figure 8). These results indicated that apical acidity abolished the inhibitory effect of omeprazole on passive Mg2+ transport and Cldn expression.
Figure 7.
Apical acidity decreases omeprazole effect on passive Mg2+ absorption. Passive Mg2+ transport (40 mmol/L concentration gradient) of 200 (A) or 400 ng/ml omeprazole exposed monolayers at various apical pH (D). %maximum passive Mg2+ transport - Mg2+ concentration curve of 200 (B) or 400 ng/ml omeprazole exposed monolayers at pH 7.4, 6.5, and 5.5 (E). EC50 of the dose - response curve of 200 (C) or 400 ng/ml omeprazole exposed monolayers at pH 7.4, 6.5, and 5.5 (F). *P < 0.05, **P < 0.01, ***P < 0.001 vs control group. For each data point, n = 6.
Figure 8.
Apical acidity restored the inhibitory effect of omeprazole effect on Cldn-7 and -12 expression. Immunobloting and densitometric analysis of Cldn-7 (A) and -12 (B) of control and 400 ng/ml omeprazole exposed cells. ***P < 0.001 vs control group, n = 5.
Discussion
Proton pump inhibitors (PPI) have been as therapeutics for acid-related gastrointestinal diseases. PPI binds and inhibits acid secretion from parietal cells of the gastric mucosa, thus allows mucosal ulcers and inflammation to heal. However, prolonged PPI usage in chronic acid-gastric diseases led to hypocalcaemia and hypomagnesaemia were probably caused by intestinal wasting (Epstein et al., 2006; Cundy and Dissanayake, 2008; Shabajee et al., 2008; Broeren et al., 2009; Kuipers et al., 2009; Hoorn et al., 2010; Mackay and Bladon, 2010). While hypocalcaemia could be remedied by elemental Ca2+ supplement, hypomagnesaemia could be treated only by withdrawal of PPI (Epstein et al., 2006; Shabajee et al., 2008). On the other hand, short-term PPI administration had no effect on intestinal Ca2+ and Mg2+ absorption (Serfaty-Lacrosniere et al., 1995; Hansen et al., 2010). In the present study, we clearly showed that omeprazole had an inhibitory effect on the passive Mg2+ absorption (Figure 1), but with much less effect on passive Ca2+ absorption (Figure 3). Therefore, omeprazole possibly inhibited the function of paracellular Mg2+ channels that were proposed to be created by the tight junction associated claudins (Cldn) (Simon et al., 1999; Tang and Goodenough, 2003). However the effect of omeprazole on the active Ca2+ transport could not be excluded by the present study.
Among the biological cations, Mg2+ is the most charge dense cation with high hydration energy of about -1,922 kj/mol versus -1,592 kj/mol of Ca2+. Therefore, Mg2+ tightly coordinates with 6-7 H2O molecules and has the highest hydration radius (Wolf and Cittadini, 2003; Wolf et al., 2003). In order to permeate the transporting epithelium, Mg2+ must partially or completely be stripped of the bound hydration shell by its transporting protein (Moomaw and Maguire, 2008). The relationship between %maximum passive Mg2+ transport and Mg2+ concentration showed that omeprazole shifted the curve to the right and increased EC50 (Figure 2), indicating a lower affinity of the paracellular channel for Mg2+. In addition, the electrical field within the paracellular pore could mediate the dehydration process and determine the charge selectivity (Yu et al., 2009). Omeprazole was previously found to decrease the negative electric field strength and cation selectivity of the paracellular cation channels in Caco-2 monolayers (Thongon and Krishnamra, 2011). Moreover, the Arrhenius plot showed a higher activation energy for passive Mg2+ transport of the omeprazole-exposed monolayers (Thongon and Krishnamra, 2011). Therefore, it appeared that omeprazole inhibited the function of paracellular Mg2+ channels in the intestinal epithelium by decreasing Mg2+ affinity and disturbing the dehydration process.
Electrostatic interactions in the paracellular pore can determine conductivity of ion transport through the pore (Tang and Goodenough, 2003; Yu et al., 2009). Therefore, changing the electrostatic interaction for instance, by lower apical pH markedly increases the paracellular ion permeability (Tang and Goodenough, 2003). In the gastrointestinal tract, luminal acidity present along the entire small intestine (Nugent et al., 2001) was required for passive Mg2+ absorption (Heijnen et al., 1993). In the present study, %maximum passive Mg2+ transport - Mg2+ concentration curves were shifted to the left (Figure 6C) and EC50 was decreased (Figure 6D) by apical acidic condition (pH 7.0-5.5), indicating enhanced paracellular passive Mg2+ absorption (Figure 6B). On the other hand, inhibition of proton pumps in Caco-2 cells (Abrahamse et al., 1992) by omeprazole decreased apical acidity (Figure 4) and paracellular passive Mg2+ transport (Figure 1) across the Caco-2 monolayers. Therefore, omeprazole, by inhibiting gastric acid secretion, disturbed the luminal acidity and suppressed the intestinal Mg2+ absorption. In long-term omeprazole usage, continuous wasting of intestinal Mg2+ gradually depleted Mg2+ store in the human body, which then led to hypomagnesaemia. However, the inhibitory effect of omeprazole on the paracelluar passive Mg2+ transport, as well as Mg2+ affinity of the paracellular channel, was normalized by physiological luminal acidic condition (Figure 7). Therefore, oral Mg2+ supplement using acid-containing capsule that released Mg2+ and acid in the distal small intestine probably resolved hypomagnesaemia in long-term omeprazole usage.
Extracellular CaSR were expressed along the entire length of the gastrointestinal tract (Geibel and Hebert, 2009) and also in Caco-2 monolayers (Davies et al., 2008). CaSR played a role in gastric acid secretion, intestinal fluid absorption and secretion, and colonic fluid secretion (Geibel and Hebert, 2009). In the renal epithelium, CaSR activation suppressed the paracellular passive Mg2+ transport by controlling Cldn-16 function in the tight junction (Ikari et al., 2008). In the present study, CaSR agonists neomycin and apermine were found to have no effect on passive Mg2+ transport in both control and omeprazole treated monolayers (Figure 4C). Our results agreed with a previous study that showed regulation of paracellular passive Ca2+ transport across Caco-2 monolayers by extracellular Ca2+ in a CaSR-independent mechanism (Davies et al., 2008). Therefore, omeprazole was likely to inhibit the intestinal passive Mg2+ absorption in a CaSR-independent manner.
Among the 23 members of claudins, Cldn-16 had been proposed as the paracellular channel for Mg2+ in kidney (Simon et al., 1999). However Cldn-16 was not detected along the intestinal tract (Fujita et al., 2006), suggesting that other Cldn might be involved in passive Mg2+ absorption. Since, omeprazole suppressed paracellular passive Mg2+ transport as well as the expression of Cldn-7 and -12 in Caco-2 monolayers (Figure 5), thus, monomeric or heteromeric combination of Cldn-7 and -12 possibly contributed to the intestinal paracellular Mg2+ absorption. Cldn contained 4 trans-membrane domains, 1 intracellular loop, and 2 extracellular loops (Krause et al., 2008). Paracellular channels were thought to be formed by the extracellular loop 1 (ECL1), negative or positive amino acid residues of which determined the charge selectivity (Yu et al., 2009). In ECL1, Cldn-7 contained two negatively charged residues (positions 38, and 53) and Cldn12 contained four negatively charged residues (positions 62, 66, 71 and 74), thus, both were likely to act as paracellular cation transporting proteins (Alexandre et al., 2005: Fujita et al., 2006, 2008).
It is known that Cldn expression can be modulated by acid. In the esophageal reflux model, marked apical acidity (pH 2) was shown to suppress Cldn-4 expression in the squamous epithelial (Oshima et al., 2012). Moreover, apical gastric acid exposure down-regulated Cldn-3 expression in the esophageal epithelium (Oguro et al., 2011). Our laboratory previously showed that chronic metabolic acidosis up-regulated mRNA expression of Cldn-2, -3, -6, -8, -11, -12, -14, -19 and -22 in the duodenal enterocytes (Charoenphandhu et al., 2007). In bone lining cells, chronic metabolic acidosis also suppressed Cldn-14 expression but increased Cldn-15 and -16 expression (Wongdee et al., 2010). In addition, chronic metabolic acidosis suppressed Cldn-2 expression in renal epithelium (Balkovetz et al., 2009). Thus, it seemed that acid modulated Cldn expression in a tissue specific manner. In the present study, apical acidity within the physiological level i.e., pH 6.5 and 5.5 up-regulated Cldn-7 and -12 expression in Caco-2 cells (Figures 6E and 6F). Apical acidity also normalized Cld-7 and -12 expression in omeprazole-exposed Caco-2 cells (Figure 8). Since Cldn-7 and -12 were cation-permeable proteins (Alexandre et al., 2005: Fujita et al., 2008), an increased in paracellular passive Mg2+ absorption (Figures 6 and 7) possibly resulted from the up-regulation of Cldn-7 and -12 in apical acid-exposed epithelium. Therefore, luminal acidity probably exerted its effect on normal intestinal uptake of Mg2+ in Cldn-7 and -12 dependent mechanism.
Methods
Cell culture
Caco-2 cells (ATCC No. HTB-37) were grown as previously described (Thongon et al., 2008). For ion flux studies, the Caco-2 monolayers were developed by seeding cells (1.0 × 106 cells/cm2) onto the permeable polyester Transwell-clear inserts (Corning, Corning, NY) and maintained for 14 days. For western blot analysis, cells were plated (5.0 × 105 cells/well) on 6-well plate and maintained for 14 days. For MTT reduction assay, cells were seeded (1.0 × 104 cells/well) on 96-well plate (Corning) and maintained for the same period of time. In the omeprazole-treated group, the monolayers were grown in media containing 200, 400, or 600, ng/ml omeprazole (Calbiochem, San Diego, CA).
Measurements of Ca2+ and Mg2+ fluxes
Apical to basolateral paracellular Ca2+ and Mg2+ fluxes studies were performed at a humidified atmosphere with 5% CO2. After removal of the culture media, the apical and basolateral side of Caco-2 monolayers on transwell were added with apical and basolateral solutions, respectively. At 1 and 2 h, 500 µl solution was collected from the basolateral side for measurement of Ca2+ or Mg2+ concentration. Fluxes and permeabilities of Ca2+ and Mg2+ across the Caco-2 monolayers were calculated as previously described (Thongon and Krishnamra, 2011). The relationship of Mg2+ transport and apical Mg2+ concentration was fitted with modified Michaelis-Menten equation plus linear component (Thongon and Krishnamra, 2011).
Bathing solution
For Ca2+ transport studies, the basolateral solution contained (in mM) 1 MgCl2, 4.5 KCl, 12 D-glucose, 2.5 L-glutamine, 250 mannitol, and 10 HEPES pH 7.4; whereas, the apical solution contained (in mM) 1 MgCl2, 4.5 KCl, 12 D-glucose, 2.5 L-glutamine and 10 HEPES pH 7.4, with either 2.5 CaCl2 and 242 mannitol, 5 CaCl2 and 235 mannitol, 10 CaCl2 and 230 mannitol, 20 CaCl2 and 200 mannitol, 40 CaCl2 and 115 mannitol, or 80 CaCl2 and 90 mannitol.
For Mg2+ transport studies, the basolateral solution was (in mM) 1.25 CaCl2, 4.5 KCl, 12 D-glucose, 2.5 L-glutamine, 250 D-mannitol, and 10 HEPES pH 7.4; the apical solution was (in mM) 2.5 CaCl2, 4.5 KCl, 12 D-glucose, 2.5 L-glutamine, and 10 HEPES pH 7.4, containing either 2.5 MgCl2 and 242 mannitol, 5 MgCl2 and 235 mannitol, 10 MgCl2 and 230 mannitol, 20 MgCl2 and 200 mannitol, 40 MgCl2 and 115 mannitol, or 80 MgCl2 and 90 mannitol.
All solutions with an osmolality of 290-295 mmol/kg H2O were gassed with 5% CO2 in 95% O2 30 min prior to use and maintained at 37℃. In the pH dependent ion flux studies, HEPES of pH 7.4 in apical solution was substituted with same concentration of HEPES pH 7.0, HEPES pH 6.5, HEPES pH 6.0, or HEPES pH 5.5. All chemicals were purchased from Sigma (Sigma, St. Louis, MO).
Measurements of Ca2+ and Mg2+ concentrations
Mg2+ concentration was determined by Xylidyl Blue colorimetric assay (pH 10.5) as previously described (Thongon and Krishnamra, 2011). Ca2+ concentration was determined by Arsenazo III colorimetric assay (pH 6.5) according to the method of Tang and Goodenough (2003). All chemicals were purchased from Sigma.
MTT reduction assay
Caco-2 cells were washed with PBS and treated with 1 mg/ml MTT solution (sigma) for 3 h in a humidified atmosphere containing 5% CO2 at 37℃. Formazan crystals in cells were solubilized with DMSO and subjected to colorimetric analysis using multi-mode microplate reader at 540 nM (BioTek Instruments, Inc).
Trans-epithelial electrical resistance
Trans-epithelial electrical resistance (TEER) was determined as previously described (Thongon et al., 2008).
Western blot analysis
Caco-2 cells were lysed in RIPA buffer (Sigma) with gentle shaking (seesaw mode) for 20 min at 4℃. After cells were scraped with Cell Scraper (Corning), lysates were sonicated, centrifuged at 12,000 g for 15 min, and then heated for 5 min at 95℃. Proteins (60 µg) or Cruz Marker™ Molecular Weight Standards were loaded and separated on 12.5% SDS-PAGE gel, then transferred to a polyvinylidene difluoride membrane (PVDF; Amersham, Buckinghamshire, UK). Membranes were blocked with 5% nonfat milk overnight at 4℃ and probed overnight at 4℃ with 1:500 rabbit polyclonal antibodies (Santa Cruz Biotechnology, Santa Cruz, CA) raised against human claudin-2, -7, -12. Membranes were also reprobed with 1:5,000 anti-β-actin monoclonal antibodies (Santa Cruz Biotechnology). After 2 h incubation at 25℃ with 1:5,000 HRP-conjugated secondary antibodies (Santa Cruz Biotechnology), blots were visualized by Pierce ECL western blotting substrate (Thermo Scientific Pierce Protein Research, Rockford, IL) and captured on Hyperfilm™ (Amersham). Densitometric analysis was performed using ImageJ for Mac Os X (Rasband, 1997-2011).
Statistical analysis
Results were expressed as means ± SE. Two sets of data were compared using the unpaired Student's t-test. One-way analysis of variance (ANOVA) with Dunnett's post test was employed for multiple sets of data. Linear regression was performed to obtain the passive Ca2+ transport - apical Ca2+ concentration relationship. Non-linear regression was performed to elucidate the %maximum - apical Mg2+ concentration relationship. The curve of PMg-Δmagnesium relationship was obtained using one phase exponential decay equation. The level of significance was P < 0.05. All data were analyzed by GraphPad Prism (GraphPad Software Inc.).
Acknowledgements
This study was supported by the Thailand Research Fund and Higher Education Commission (No. MRG5380003) and Higher Education Commission (No.44495) to N. Thongon. We thank Dr. Siriporn Chamniansawat, Ms. Penpisut Boonthongthalerng, Mr. Phongthong Kunfai, and Ms. Onchuda Koobkratok of the Facuty of Allied Health Sciences, Burapha University for their technical assistance.
Abbreviations
- CaSR
calcium sensing receptor
- Cldn
claudin
- ΔCa
concentration gradient of Ca2+
- ΔMg
concentration gradient of Mg2+
- EC50
half maximal effective concentration
- ECL1
extracellular loop 1
- PMg
magnesium permeability
- PPI
proton pump inhibitor
- r2
coefficient of determination
- RIPA buffer
radio-immunoprecipitation assay buffer
- TEER
trans-epithelial electrical resistance
References
- 1.Abrahamse SL, Bindels RJ, van Os CH. The colon carcinoma cell line Caco-2 contains an H+/K+-ATPase that contributes to intracellular pH regulation. Pflügers Arch. 1992;421:591–597. doi: 10.1007/BF00375056. [DOI] [PubMed] [Google Scholar]
- 2.Alexandre MD, Lu Q, Chen YH. Overexpression of claudin-7 decreases the paracellular Cl- conductance and increases the paracellular Na+ conductance in LLC-PK1 cells. J Cell Sci. 2005;118:2683–2693. doi: 10.1242/jcs.02406. [DOI] [PubMed] [Google Scholar]
- 3.Alexandre MD, Jeansonne BG, Renegar RH, Tatum R, Chen YH. The first extracellular domain of claudin-7 affects paracellular Cl- permeability. Biochem Biophys Res Commun. 2007;357:87–91. doi: 10.1016/j.bbrc.2007.03.078. [DOI] [PubMed] [Google Scholar]
- 4.Balkovetz DF, Chumley P, Amlal H. Downregulation of claudin-2 expression in renal epithelial cells by metabolic acidosis. Am J Physiol Renal Physiol. 2009;297:F604–F611. doi: 10.1152/ajprenal.00043.2009. [DOI] [PubMed] [Google Scholar]
- 5.Broeren MA, Geerdink EA, Vader HL, van den Wall Bake AW. Hypomagnesemia induced by several proton-pump inhibitors. Ann Intern Med. 2009;151:755–756. doi: 10.7326/0003-4819-151-10-200911170-00016. [DOI] [PubMed] [Google Scholar]
- 6.Capodicasa E, Cornacchione P, Natalini B, Bartoli A, Coaccioli S, Marconi P, Scaringi L. Omeprazole induces apoptosis in normal human polymorphonuclear leucocytes. Int J Immunopathol Pharmacol. 2008;21:73–85. doi: 10.1177/039463200802100109. [DOI] [PubMed] [Google Scholar]
- 7.Charoenphandhu N, Wongdee K, Tudpor K, Pandaranandaka J, Krishnamra N. Chronic metabolic acidosis upregulated claudin mRNA expression in the duodenal enterocytes of female rats. Life Sci. 2007;80:1729–1737. doi: 10.1016/j.lfs.2007.01.063. [DOI] [PubMed] [Google Scholar]
- 8.Cundy T, Dissanayake A. Severe hypomagnesaemia in long-term users of proton-pump inhibitors. Clin Endocrinol (Oxf) 2008;69:338–341. doi: 10.1111/j.1365-2265.2008.03194.x. [DOI] [PubMed] [Google Scholar]
- 9.Davies SL, Gibbons CE, Steward MC, Ward DT. Extracellular calcium- and magnesium-mediated regulation of passive calcium transport across Caco-2 monolayers. Biochim Biophys Acta. 2008;1778:2318–2324. doi: 10.1016/j.bbamem.2008.05.013. [DOI] [PubMed] [Google Scholar]
- 10.Doroszewicz J, Walderger P, Jeck N, Seyberth H, Waldegger S. pH dependence of extracellular calcium sensing receptor activity determined by a novel technique. Kidney Int. 2005;67:187–192. doi: 10.1111/j.1523-1755.2005.00069.x. [DOI] [PubMed] [Google Scholar]
- 11.Ekmekcioglu C, Ekmekcioglu A, Marktl W. Magnesium transport from aqueous solutions across Caco-2 cells--an experimental model for intestinal bioavailability studies. Physiological considerations and recommendations. Magnes Res. 2000;13:93–102. [PubMed] [Google Scholar]
- 12.Epstein M, McGrath S, Law F. Proton-pump inhibitors and hypomagnesemic hypoparathyroidism. N Engl J Med. 2006;355:1834–1836. doi: 10.1056/NEJMc066308. [DOI] [PubMed] [Google Scholar]
- 13.Fujita H, Chiba H, Yokozaki H, Sakai N, Sugimoto K, Wada T, Kojima T, Yamashita T, Sawada N. Differential expression and subcellular localization of claudin-7, -8, -12, -13, and -15 along the mouse intestine. J Histochem Cytochem. 2006;54:933–944. doi: 10.1369/jhc.6A6944.2006. [DOI] [PubMed] [Google Scholar]
- 14.Fujita H, Sugimoto K, Inatomi S, Maeda T, Osanai M, Uchiyama Y, Yamamoto Y, Wada T, Kojima T, Yokozaki H, Yamashita T, Kato S, Sawada N, Chiba H. Tight junction proteins claudin-2 and -12 are critical for vitamin D-dependent Ca2+ absorption between enterocytes. Mol Biol Cell. 2008;19:1912–1921. doi: 10.1091/mbc.E07-09-0973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Furuse M, Tsukita S. Claudins in occluding junctions of humans and flies. Trends Cell Biol. 2006;16:181–188. doi: 10.1016/j.tcb.2006.02.006. [DOI] [PubMed] [Google Scholar]
- 16.Geibel JP, Hebert SC. The functions and roles of the extracellular Ca2+-sensing receptor along the gastrointestinal tract. Annu Rev Physiol. 2009;71:205–217. doi: 10.1146/annurev.physiol.010908.163128. [DOI] [PubMed] [Google Scholar]
- 17.Hansen KE, Jones AN, Lindstrom MJ, Davis LA, Ziegler TE, Penniston KL, Alvig AL, Shafer MM. Do proton pump inhibitors decrease calcium absorption? J Bone Miner Res. 2010;25:2786–2795. doi: 10.1002/jbmr.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Heijnen AM, Brink EJ, Lemmens AG, Beynen AC. Ileal pH and apparent absorption of magnesium in rats fed on diets containing either lactose or lactulose. Br J Nutr. 1993;70:747–756. doi: 10.1079/bjn19930170. [DOI] [PubMed] [Google Scholar]
- 19.Hoorn EJ, van der Hoek J, de Man RA, Kuipers EJ, Bolwerk C, Zietse R. A case series of proton pump inhibitor-induced hypomagnesemia. Am J Kidney Dis. 2010;56:112–116. doi: 10.1053/j.ajkd.2009.11.019. [DOI] [PubMed] [Google Scholar]
- 20.Ikari A, Okude C, Sawada H, Sasaki Y, Yamazaki Y, Sugatani J, Degawa M, Miwa M. Activation of a polyvalent cation-sensing receptor decreases magnesium transport via claudin-16. Biochim Biophys Acta. 2008;1778:283–290. doi: 10.1016/j.bbamem.2007.10.002. [DOI] [PubMed] [Google Scholar]
- 21.Konrad M, Schlingmann KP, Gudermann T. Insights into the molecular nature of magnesium homeostasis. Am J Physiol Renal Physiol. 2004;286:F599–F605. doi: 10.1152/ajprenal.00312.2003. [DOI] [PubMed] [Google Scholar]
- 22.Krause G, Winkler L, Mueller SL, Haseloff RF, Piontek J, Blasig IE. Structure and function of claudins. Biochim Biophys Acta. 2008;1778:631–645. doi: 10.1016/j.bbamem.2007.10.018. [DOI] [PubMed] [Google Scholar]
- 23.Kuipers MT, Thang HD, Arntzenius AB. Hypomagnesaemia due to use of proton pump inhibitors-a review. Neth J Med. 2009;67:169–172. [PubMed] [Google Scholar]
- 24.Macek J, Klíma J, Ptácek P. Rapid determination of omeprazole in human plasma by protein precipitation and liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2007;852:282–287. doi: 10.1016/j.jchromb.2007.01.026. [DOI] [PubMed] [Google Scholar]
- 25.Mackay JD, Bladon PT. Hypomagnesaemia due to proton-pump inhibitor therapy: a clinical case series. QJM. 2010;103:387–395. doi: 10.1093/qjmed/hcq021. [DOI] [PubMed] [Google Scholar]
- 26.McCarthy JM, Kumar R. Divalent cation metabilism: Magnesium. In: Schrier RW, Berl T, Bonventre JV, editors. Atlas of Diseases of the Kidney. volume 1. Philadaphia, PA: Current Medicine Inc; 1999. 4.1-4.12. [Google Scholar]
- 27.Moomaw AS, Maguire ME. The unique nature of Mg2+ channels. Physiology. 2008;23:275–285. doi: 10.1152/physiol.00019.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nugent SG, Kumar D, Rampton DS, Evans DF. Intestinal luminal pH in inflammatory bowel disease: possible determinants and implications for therapy with aminosalicylates and other drugs. Gut. 2001;48:571–577. doi: 10.1136/gut.48.4.571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Oguro M, Koike M, Ueno T, Asaoka D, Mori H, Nagahara A, Uchiyama Y, Watanabe S. Dissociation and dispersion of claudin-3 from the tight junction could be one of the most sensitive indicators of reflux esophagitis in a rat model of the disease. J Gastroenterol. 2011;46:629–638. doi: 10.1007/s00535-011-0390-1. [DOI] [PubMed] [Google Scholar]
- 30.Oshima T, Koseki J, Chen X, Matsumoto T, Miwa H. Acid modulates the squamous epithelial barrier function by modulating the localization of claudins in the superficial layers. Lab Invest. 2012;92:22–31. doi: 10.1038/labinvest.2011.139. [DOI] [PubMed] [Google Scholar]
- 31.Quamme GA. Recent developments in intestinal magnesium absorption. Curr Opin Gastroenterol. 2008;24:230–235. doi: 10.1097/MOG.0b013e3282f37b59. [DOI] [PubMed] [Google Scholar]
- 32.Quinn SJ, Bai M, Brown EM. pH Sensing by the calcium-sensing Receptor. J Biol Chem. 2004;279:37241–37249. doi: 10.1074/jbc.M404520200. [DOI] [PubMed] [Google Scholar]
- 33.Rasband WS. ImageJ. Bethesda, Maryland, USA: U.S. National Institutes of Health; 1997-2011. imagej.nih.gov/ij/ [Google Scholar]
- 34.Serfaty-Lacrosniere C, Wood RJ, Voytko D, Saltzman JR, Pedrosa M, Sepe TE, Russell RR. Hypochlorhydria from short-term omeprazole treatment does not inhibit intestinal absorption of calcium, phosphorus, magnesium or zinc from food in humans. J Am Coll Nutr. 1995;14:364–368. doi: 10.1080/07315724.1995.10718522. [DOI] [PubMed] [Google Scholar]
- 35.Shabajee N, Lamb EJ, Sturgess I, Sumathipala RW. Omeprazole and refractory hypomagnesaemia. BMJ. 2008;337:a425. doi: 10.1136/bmj.39505.738981.BE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Simon DB, Lu Y, Choate KA, Velazquez H, Al-Sabban E, Prega M, Casari G, Bettiniel A, Colussi G, Rodiguez-Soriano J, McCredie D, Milford D, Sanjad S, Lifton RP. Paracellin-1, a renal tight junction protein required for paracellular Mg2+ resorption. Science. 1999;285:103–106. doi: 10.1126/science.285.5424.103. [DOI] [PubMed] [Google Scholar]
- 37.Tang VW, Goodenough DA. Paracellular ion channel at the tight junction. Biophys J. 2003;84:1660–1673. doi: 10.1016/S0006-3495(03)74975-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thongon N, Nakkrasae LI, Thongbunchoo J, Krishnamra N, Charoenphandhu N. Prolactin stimulates transepithelial calcium transport and modulates paracellular permselectivity in Caco-2 monolayer: mediation by PKC and ROCK pathways. Am J Physiol Cell Physiol. 2008;294:C1158–C1168. doi: 10.1152/ajpcell.00020.2008. [DOI] [PubMed] [Google Scholar]
- 39.Thongon N, Krishnamra N. Omeprazole decreases magnesium transport across Caco-2 monolayers. World J Gastroenterol. 2011;17:1574–1583. doi: 10.3748/wjg.v17.i12.1574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wolf FI, Cittadini A. Chemistry and biochemistry of magnesium. Mol Aspects Med. 2003;24:3–9. doi: 10.1016/s0098-2997(02)00087-0. [DOI] [PubMed] [Google Scholar]
- 41.Wolf FI, Torsello A, Fasanella S, Cittadini A. Cell physiology of magnesium. Mol Aspects Med. 2003;24:11–26. doi: 10.1016/s0098-2997(02)00088-2. [DOI] [PubMed] [Google Scholar]
- 42.Wongdee K, Riengrojpitak S, Krishnamra N, Charoenphandhu N. Claudin expression in the bone-lining cells of female rats exposed to long-standing acidemia. Exp Mol Pathol. 2010;88:305–310. doi: 10.1016/j.yexmp.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 43.Ye C, Ho-Pao CL, Kanazirska M, Quinn S, Seidman CE, Seidman JG, Brown EM, Vassilev PM. Deficient cation channel regulation in neurons from mice with targeted disruption of the extracellular Ca2+-sensing receptor gene. Brain Res Bull. 1997;44:75–84. doi: 10.1016/s0361-9230(97)00088-9. [DOI] [PubMed] [Google Scholar]
- 44.Yu AS, Cheng MH, Angelow S, Gunzel D, Kanzawa SA, Scheeberger EE, Fromm M, Coalson RD. Molecular basis for cation selectivity in claudin-2 base paracellular pore: Identification of electrostatic site. J Gen Physiol. 2009;133:111–127. doi: 10.1085/jgp.200810154. [DOI] [PMC free article] [PubMed] [Google Scholar]