Abstract
This study investigated the effects of polymer dispersion and hydration conditions on hypromellose (HPMC) film properties, such as strength, oxygen permeability, water vapor transmission, clarity, and haze. The focus of the study was to build a better understanding of the impact that changes to HPMC dispersion and hydration conditions have on performance properties of the resulting films. This understanding could potentially lead to more flexible formulation guidelines for formulators. Films of HPMC 2906 (USP) were produced from aqueous solutions prepared using various formulation conditions. Results showed that tensile properties and oxygen permeability were not significantly affected by the variables used. The differences observed in water vapor transmission are unlikely to affect practical application of the material. However, the differences observed in clarity and haze at 50°C hydration temperature could affect the appearance of a capsule or coated tablet. Several methods were used to determine whether loss of optical properties was due to surface phenomena or bulk defects within a film. Results indicated that the cloudy appearance was primarily due to surface roughness. Based on this information, there is some flexibility in formulation conditions; however, hydration temperatures greater than 25°C are not recommended.
KEY WORDS: film properties, formulation conditions, HPMC, hypromellose, polymer hydration
INTRODUCTION
Hypromellose (HPMC) is a common excipient used in pharmaceutical films, such as tablet coatings and hard capsule shells. The process of preparing formulations of HPMC to produce films that meet high-quality requirements can be an exacting process. Standard preparation protocols call for the polymer to be first dispersed in hot water (>80°C) and then hydrated at lower temperatures (<10°C) (1). If the powder is added directly to cold water, lumps may form in which the outer layer of polymer begins to hydrate, forming a shell around the dry powder, which can result in extended polymer hydration times.
Such exacting formulation conditions can be difficult to maintain, prompting a desire from formulators for more flexibility in the formulation process. The focus of this study was to build a better understanding of HPMC dispersion and hydration, and the impact that changes to these processes have on the performance properties of the resulting films. A secondary emphasis was to determine if the recommended formulation conditions for HPMC could be broadened to make the formulation process more flexible.
BACKGROUND
Several film properties are assessed when considering formulations for film-forming polymers, including mechanical strength, permeation, and optical properties (2–5). Strength is a particularly important property for films used to form capsule shells or tablet coatings. The film must be able to protect the contents during filling, packaging, shipping, and storage processes.
In addition to mechanical strength, low oxygen permeability and water vapor transmission are desired properties for capsule shells, since certain active pharmaceutical ingredients react with either water or oxygen. Gasses travel through the films by two methods, capillary flow and diffusion. Capillary flow occurs when a gas passes through pores within a film. Transmission of water vapor occurs when the substance dissolves on the surface, diffuses through the film, and evaporates on the other side (6). Both processes are affected by the structure of the polymer composing the film and the affinity of the gas to the material. For example, water vapor would have high transmission through a hydrophilic material such as HPMC. Capillary flow would be increased by defects within the film that allow the gas to easily pass through. The presence of additives or moisture in the film can also affect the interaction of the gas with the material and therefore the permeation/transmission rates. For example, the rate of oxygen permeation through an HPMC film is sensitive to the humidity of the testing conditions (7).
Although strength and gas permeation/transmission properties are critical to protecting the contents of a capsule or tablet, the initial perception of the customer will be based on the appearance of the film. In cases where a clear film is desired, a lack of clarity or the presence of haze would be a negative feature. Typically, as clarity decreases, haze increases. Clarity is determined by small-angle light scattering; haze is determined by large-angle light scattering.
Poor optical quality can be due to defects that exist either on the surface or within the bulk of the film (8). Inconsistent structure within the bulk of the film can lead to a non-uniform refractive index, resulting in poor optical quality. Crystallization within the film can also create defects that will alter the optical properties of the material (9–11).
Several methods exist for investigating the source of poor optical quality. Optical properties can be compared to film thickness. If the defects lie on the surface of the film, these properties would remain constant as film thickness is varied. If the reason for poor optical quality is within the bulk of the film, optical properties would probably correlate with film thickness. An alternative method is to coat the surface of the film with oil that has a refractive index similar to the film material. The oil will fill any surface defects but not affect those within the bulk of the film. If the optical quality of the oil-coated film improves, this would indicate the defects are on the surface. Analysis of surface morphology may be conducted through either contact (profilometry) or noncontact (interferometry) methods, both of which provide information on surface roughness. Increased surface roughness can affect both the optical properties of the film and also the visual appearance of the film texture (12).
MATERIALS AND METHODS
Materials
The films in this study were made from low-viscosity (low molecular weight) HPMC 2906 (METHOCEL1 F5 Premium cellulose ether, The Dow Chemical Company, Midland, MI, USA).
Hypromellose Solution Preparation
All formulations were prepared using the conventional “hot–cold” method in which the HPMC powder is dispersed in water at an elevated temperature and then cooled for hydration of the polymer (1). Temperature during dispersion and hydration was maintained with a water bath monitored with a thermocouple to within a degree of the target. The solutions (20% HPMC in water) were prepared by quickly adding the powder to a 500-mL jar containing water at the dispersion temperature (60 or 80°C) and equipped with a three-blade stirring shaft. During the powder addition, the stirring rate was set to 600 rpm to efficiently draw the powder into the water by creating a vortex. After powder addition, the rate was reduced to 400 rpm to reduce the number of bubbles incorporated into the solution. Stirring during the dispersion phase was continued for 1 h. Following dispersion, the water bath was adjusted to the hydration temperature (4°C, 25°C, or 50°C). Timing of the hydration phase began once the solution had reached the appropriate temperature, typically 5–15 min, depending on the magnitude of the temperature drop. Once the hydration temperature was achieved, stirring was stopped. Samples were removed at 1, 3, and 5 h. Before films were prepared, dissolved gas was removed from the solution by placing the sample under vacuum for 2 min followed by centrifugation at 2,800 rpm for 5 min to remove bubbles.
Preparation of Films
Films were hand-drawn on glass plates using the 40-mil gap option of a multiple clearance application square (BYK, Columbia, MD, USA). The HPMC solution (at hydration temperature) was poured into the square near the edge of the glass plate, and the square was pulled steadily down the glass to minimize formation of defects. Films were dried overnight under ambient conditions, typically about 21°C with relative humidity ranging from 20% to 40%. Thermogravimetric analysis (TGA 2950, DuPont Instruments, Cincinnati, OH, USA) was used to confirm that the water content of the films was consistent with dry films, about 6% moisture content. After removal from the glass, the films were stored in a laboratory with constant temperature and humidity (22°C, 50% relative humidity) for at least 18 h prior to analysis to equilibrate the water content. For all film testing, samples were taken from areas of the film that did not contain any obvious defects or bubbles. Film thickness is reported in the unit mil or 0.001 in.
Tensile Strength Measurements
Tensile strength was measured using an Instron universal testing machine (Model 4201/5501R, Instron, Norwood, MA, USA) following ASTM method D-638 with an extension rate of 0.2 in./min. Samples were cut from the film using a type IV die. Thickness of each sample was measured prior to testing and was generally between 4 and 5 mil.
Oxygen Permeability and Water Vapor Transmission Rate Measurements
Oxygen permeability was measured on an Ox-Tran 2/21 system (Mocon, Minneapolis, MN, USA). Test conditions were 10% oxygen, 23°C, and 50% relative humidity. Films were masked to a 1-in. diameter testing area, which was normalized to a 5 cm2 testing area in final calculations. The permeability was normalized for the film thickness to yield a value with units of cc∙mil/[100 in2∙day∙atm]. Four samples were tested for each condition at an HPMC hydration time of 5 h.
Water vapor transmission rates were measured using a dry cup method. Two grams of calcium chloride was weighed into a 4-oz jar and allowed to equilibrate at 50% relative humidity and 73°F with a closed cap for 1 h. Next, a small amount of vacuum grease was placed around the edge of the jar’s mouth. Films were cut into 1.3-in. diameter circles using a metal punch. The film samples were placed over the mouth of the jar, and a 1-in. diameter, open-hole lid was placed on top. The lid was tightened enough to form a seal, but not enough to damage the film. The jars were then placed in a temperature/humidity chamber equipped with an Environ-Cab controller (Lab-Line Instrument, Inc., Melrose Park, IL, USA) set at 75% relative humidity and 25°C. The total weight of the jar, lid, film, and calcium chloride was recorded and measured again approximately every 24 h for 5 days. The rate was normalized for film thickness and to a 5 cm2 testing area to yield a value with units of grams per meter per square centimeter per minute. Four samples were tested for each condition at an HPMC hydration time of 5 h.
Clarity and Haze Measurements
Clarity was measured on a clarity meter (Model CL-100, Zebedee Corporation, Moore, SC, USA). Haze was measured at four places on each film using a Haze-guard unit (BYK Gardner, Columbia, MD, USA) equipped with a CIE-C light source. The thickness of each film was measured prior to analysis.
Film Surface Property Measurements
To check for surface vs bulk defects, a drop of silicone oil was placed on the surface of both the clear and cloudy films. Silicone oil has a refractive index near that of HPMC film (n ~ 1.52). Microscopy was used to collect images of each film type (clear or cloudy) at five or 10 times magnification. Comparison of the areas coated with oil to those without can indicate if the defects are on the surface or within the bulk of the film.
For surface roughness determinations, each film was mounted to a glass slide and imaged using a Tencor P-15 stylus profilometer (KLA-Tencor Corporation, Milpitas, CA, USA). A 1,000 × 1,000 μm area was imaged with a 1.0-mg load, 200-μm/s scan speed, 50-Hz sampling rate, 4-μm y-spacing, and 131 μm/0.0781 Å range/resolution. Data were processed and analyzed using SPIP v.5.1.5 software (Image Metrology, Hørsholm, Denmark). Images were plane-fit and filtered for noise before roughness analysis.
A white light interferometer (Model NT9100, Wyko Corporation, Tucson, AZ, USA) was also used to analyze the surface characteristics of the films. To ensure that analysis was representative of the whole film, several locations on each film were tested, avoiding any areas of visible defects, such as bubbles. Each scan analyzes an area of approximately 0.1 x 0.1 mm. The images presented are of representative locations and numerical results show an average of at least three locations.
RESULTS AND DISCUSSION
Film Mechanical Properties
Mechanical strength is a key property for excipient films used in capsules and tablet coatings. The capsules must be able to withstand the filling process and both capsules and tablet coatings must provide protection of the contents during shipping and storage. In this study, tensile properties were used as a measure of strength. Figure 1 shows average Young’s modulus for each formulation condition. There were no clear trends when the dispersion or hydration temperature was varied, and extending the hydration time from 1 to 3 or 5 h had no significant effect on the modulus. This data set was analyzed using the Tukey–Kramer test for statistically relevant similarity, and results showed no difference between the data from any formulation condition to a 95% confidence level. Maximum tensile stress and strain were also measured, with the data compared in Figs. 2 and 3. These properties exhibited a relatively large standard deviation compared to Young’s modulus, an inherent difficulty when measuring tensile properties of films, but there were no clear trends across the range of conditions tested.
Fig. 1.

Effect of formulation conditions on Young’s modulus
Fig. 2.

Effect of formulation conditions on maximum tensile stress
Fig. 3.

Effect of formulation conditions on maximum tensile strain
Oxygen Permeability and Water Vapor Transmission
Both oxygen and water vapor can affect the contents of capsules and tablets, making low permeability for these substances a highly desirable property. Oxygen permeation through hypromellose films is known to be highly affected by the water content of the films (7), which is determined by the humidity of the testing conditions. For this study, humidity was held at a constant value of 50% for all measurements and therefore should not affect permeability results. However, even more than humidity, permeability is influenced by defects in the film that create pores through which the gas can transmit. Therefore, changes in polymer structure due to formulation conditions could be reflected by changes in oxygen permeability. Figure 4 shows that oxygen permeability was not affected by the different formulation conditions studied, indicating that no set of formulation conditions was more likely to produce a defect sensitive to oxygen permeation than another. Because no difference was seen between oxygen permeability at the low and high hydration temperatures and because this measurement is very time-consuming, hydration data were not collected at the middle hydration temperature, 25°C.
Fig. 4.

Effect of formulation conditions on oxygen permeability at 5 h hydration time, 50% RH, 23°C
Water vapor transmission rate (WVTR) of films produced from formulations dispersed at 60°C was higher for both hydration temperatures than formulations dispersed at 80°C (Fig. 5), a statistically significant difference with a confidence level of 95%. The average WVTR of formulations hydrated at 50°C was higher than those hydrated at 4°C, within the same dispersion condition, but the large standard deviations limited the amount of statistically significant conclusions that could be drawn from these data. Again, this measurement was limited to the high and low hydration temperatures because the additional time required to collect data at the middle temperature, 25°C, was not likely to further elucidate the results. The introduction of this paper describes two methods by which water vapor can travel through a film, by capillary flow through pores (defects) in the film, or by a process in which the vapor diffuses through the polymer structure of the film. Either of these mechanisms could be affected by a change in the film. Defects in the film surface will give more opportunity for capillary flow, and a change in the polymer structure composing the film may affect the interaction of water with the polymer, therefore changing the diffusion rates (6). Despite the variability observed with dispersion conditions, based on the authors’ experience, these results are not likely to affect practical application of the material.
Fig. 5.

Effect of formulation conditions on water vapor transmission at 5 h hydration time, 75% RH, 25°C
The oxygen permeation and water vapor transmission analysis of the film produced from different dispersion and hydration conditions indicate that these properties do not vary enough within this range of conditions to be a primary measure of film quality. This is a positive result in regards to formulation flexibility when preparing films, but not a good discriminating factor.
Clarity and Haze
Varying the dispersion temperature did not affect either clarity or haze (Figs. 6 and 7), but when the formulation was hydrated and cast at 50°C, the films had a much lower level of clarity relative to hydration at 4°C or 25°C (Fig. 6). Clarity of films hydrated at 50°C did not appreciably change when the hydration time was lengthened. Analysis of the clarity results using the Tukey–Kramer test revealed statistically significant differences only between hydration temperatures, not when dispersion temperature or hydration time was changed, with the exception of the samples hydrated at 50°C. The clarity values for this set of data were low enough that the films would not be usable for capsules or tablet coatings. Haze analysis of the films revealed the same trends, a decrease in optical quality (increase in haze) with increased hydration temperatures (Fig. 7). Even though all haze values were low, when the hydration temperature was increased to 50°C the haze results were higher at a statistically significant level compared to conditions at 4°C or 25°C, demonstrated by the Tukey–Kramer test.
Fig. 6.

Effect of formulation conditions on film clarity
Fig. 7.
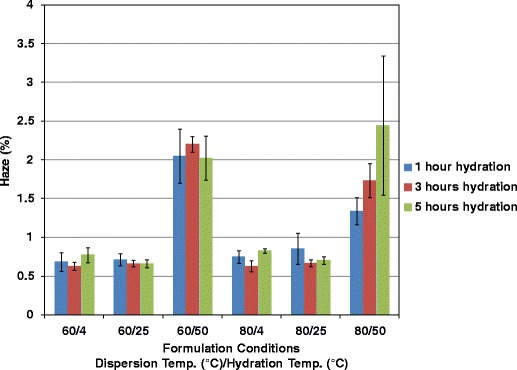
Effect of formulation conditions on film haze
Clarity was also measured on films with a range of thicknesses. Films were cast at wet thicknesses ranging from 5 to 50 mil, producing dry films from 0.5 to 8 mil. There was no indication that the clarity varied across this range of film thicknesses for either the clear films (formulation hydrated at 4°C) or the cloudy films (hydrated at 50°C; Fig. 8).
Fig. 8.

Clarity of films produced from HPMC solution hydrated at 4°C or 50°C
The reason for the difference observed in optical quality between films produced at different formulation conditions is not obvious. A key question is whether the optical effects are surface effects or lie within the bulk of the HPMC film. Several internal Dow studies have determined that, in the case of polyethylene films, observed haze is either due to surface roughness caused as the film exits the die or from crystallization near the film surface. For HPMC, surface defects could occur as the casting bar is pulled across the formulation to create the films. At higher hydration temperatures, the solution is above the temperature at which thermal gelation occurs and this more viscous, gelled solution is less likely to produce a smooth surface upon film casting. This is consistent with general HPMC solution properties in which the viscosity initially decreases upon heating, but at a point referred to as the “incipient gelation temperature” the viscosity undergoes a steep rise (13). For an HPMC formulation containing 20% METHOCEL F5 Premium cellulose ether, this point is near 36.5°C. Casting films from the more viscous solution above this temperature may be contributing to the observed surface defects. In addition, higher hydration temperatures may slow the hydration process or not allow for complete hydration. This may change the polymer structure within the film, which could reduce optical quality.
Bulk vs. Surface Optical Effects
The fact that there was no change in clarity across a range of film thicknesses for either the clear films (formulation hydrated at 4°C) or the cloudy films (hydrated at 50°C; Fig. 8) is strong evidence in support of a surface effect. A simple microscopy test provided further support. When a drop of oil placed on the clear and cloudy films was examined under a light microscopy, the oil layer removed the appearance of defects on the cloudy film, smoothing any roughness on the surface and resulting in fewer defects to deflect the light. On the clear film, the oil did not have an obvious impact on the appearance. These results give further evidence that the lower optical quality of the cloudy films is due to surface effects (Fig. 9).
Fig. 9.

An oil test performed by placing a drop of oil on the surface of a cloudy and a clear film
White light interferometry and profilometry were used to measure surface roughness. Figure 10 shows the interferometry analysis of a clear film sample hydrated at 4°C and a cloudy film hydrated at 50°C. Table I lists the average numerical results. The images and surface roughness parameters reveal a difference between the side of the film dried against the glass substrate and the side exposed to air, and an even greater difference between the exposed surface of the clear and cloudy films. The surface roughness parameters Sa (average height of the surface roughness) and Sq (root mean square of the roughness) target average deviation from the plane of the surface (11). The exposed side of the cloudy film exhibited values that were about 10 times greater than for the exposed side of the clear film. Sz and St both relate to the height difference between the greatest peaks and valleys of the surface. The difference between the glass side of the clear and cloudy films was not as great for these parameters, but the cloudy film did appear to have more peaks than the clear film.
Fig. 10.

White light interferometry images of a clear film and a cloudy film. All images are of a 95 × 127 μm area of film
Table I.
Surface Roughness Parameters, Mean Deviation (S a) and Root Mean Square Deviation (S q), Obtained with use of a White Light Interferometer or Profilometer
| Method | Film | S a (nm) | S q (nm) |
|---|---|---|---|
| Interferometry | Clear, glass side | 5 | 6 |
| Cloudy, glass side | 6 | 8 | |
| Clear, exposed side | 10 | 20 | |
| Cloudy, exposed side | 140 | 170 | |
| Profilometry | Clear, exposed side | 10 | 20 |
| Cloudy, exposed side | 400 | 500 |
Profilometry was used to examine another set of similar samples, examining only the side of the film exposed to air during drying. Figure 11 shows a three-dimensional image of each film. Note that the scale of the z-axis is different for each sample in order to show detail. The cloudy film is displayed with a surface depth of 3,300 nm, while the clear film, which was much smoother with smaller defects, is displayed at a surface depth of 200 nm. Data in Table I indicate a 40-fold increase in surface roughness for the cloudy film.
Fig. 11.

Three-dimensional image of each film
These results indicate that hydration temperature was the critical factor affecting optical quality, increasing surface roughness of the HPMC film. Although the upper limit for hydration temperature in this study (50°C) is well above normal solution preparation temperatures, the positive results seen at 25°C hydration could enable more flexibility in the formulation process, which now requires a hydration temperature of <10°C.
CONCLUSIONS
Hypromellose films were prepared using a range of dispersion and hydration conditions. Although physical properties of the films were generally unaffected by the range of conditions tested, optical properties were negatively affected by high hydration temperatures (50°C). A more moderate increase in hydration temperature to 25°C did not affect optical properties. Film clarity was not affected by film thickness, indicating that the change in optical properties was a surface effect. This finding was confirmed by microscopy, interferometry, and profilometry tests, which indicated surface roughness as the cause of lower optical quality. Therefore, although there can be some flexibility in the formulation conditions for HPMC solution preparation, hydration temperatures greater than 25°C are not recommended.
Acknowledgments
We would like to thank Bob Gunther and Sheri Clark for assistance with measuring clarity and haze properties of the films, Ellen Keene for conducting the microscopy oil test, and Meagan Blake for profilometry analysis of the films.
Footnotes
™Trademark of The Dow Chemical Company (“Dow”) or an affiliated company of Dow
References
- 1.The Dow Chemical Company. Methocel Cellulose Ethers Technical Handbook. Form No. 192-01062-0902 AMS, Sept. 2002.
- 2.Bae HJ, Cha DS, Whiteside WS, Park HJ. Film and pharmaceutical hard capsule formation properties of mungbean, waterchestnut, and sweet potato starches. Food Chem. 2008;106(1):96–105. doi: 10.1016/j.foodchem.2007.05.070. [DOI] [Google Scholar]
- 3.Al-Tabakha MM. HPMC capsules: current status and future prospects. J Pharm Pharm Sci. 2010;13(3):428–42. doi: 10.18433/j3k881. [DOI] [PubMed] [Google Scholar]
- 4.Felton LA, Timmins GS. A nondestructive technique to determine the rate of oxygen permeation into solid dosage forms. Pharm Dev Technol. 2006;11(1):141–7. doi: 10.1080/10837450600561208. [DOI] [PubMed] [Google Scholar]
- 5.Missaghi S, Fassihi R. Evaluation and comparison of physicomechanical characteristics of gelatin and hypromellose capsules. Drug Devel Ind Pharm. 2006;32(7):829–38. doi: 10.1080/03639040600608888. [DOI] [PubMed] [Google Scholar]
- 6.Debeaufort F, Voilley A, Meares P. Water vapor permeability and diffusivity through methylcellulose edible films. J Membrane Sci. 1994;91(6):125–33. doi: 10.1016/0376-7388(94)00024-7. [DOI] [Google Scholar]
- 7.Laksmana F, Hartman Kok P, Frijlink H, Vromans H, Van Der Voort Maarschalk K. Gas permeation related to the moisture sorption in films of glassy hydrophilic polymers. J Appl Polym Sci. 2010;116(6):3310–7. [Google Scholar]
- 8.Johnson M, Wilkes G, Sukhadia A, Rohlfing D. Optical properties of blown and cast polyethylene films: surface versus bulk structural considerations. J Appl Polym Sci. 2000;77(13):2845–64. doi: 10.1002/1097-4628(20000923)77:13<2845::AID-APP6>3.0.CO;2-7. [DOI] [Google Scholar]
- 9.Larena A, Pinto G. The effect of surface roughness and crystallinity on the light scattering of polyethylene tubular blown films. Polym Eng Sci. 1993;33(12):742–7. doi: 10.1002/pen.760331204. [DOI] [Google Scholar]
- 10.Stehling FC, Speed CS, Westerman L. Causes of haze of low-density polyethylene blown films. Macromolecules. 1981;14(3):698–708. doi: 10.1021/ma50004a047. [DOI] [Google Scholar]
- 11.Ashizawa H, Spruiell JE, White JL. An investigation of optical clarity and crystalline orientation in polyethylene tubular film. Polym Eng Sci. 1984;24(13):1035–42. doi: 10.1002/pen.760241307. [DOI] [Google Scholar]
- 12.Seitavoupio P, Heinamaki J, Rantanen J, Yliruusi J. Monitoring tablet surface roughness during the film coating process. Pharm Sci Technol. 2006;7(2):E1–6. doi: 10.1208/pt070231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sarkar N. Thermal gelation properties of methyl and hydroxypropyl methylcellulose. J Appl Polym Sci. 1979;24(4):1073–87. doi: 10.1002/app.1979.070240420. [DOI] [Google Scholar]


