Table 3.
Interaction Energies for Complexes between Models a–c and a Water Molecule
| System[a] | Interaction Energy (ΔE)[b] | |
|---|---|---|
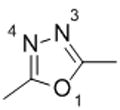
|
Compound 12 | |
| W:O1 (a)[c] | −2.03 | |
| W:N3 (a) | −6.12 | |
| W:N4 (a) | −6.12 | |
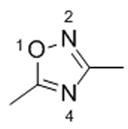
|
Compound 13 | |
| W:O1 (b) | −3.68 | |
| W:N2 (b) | −4.77 | |
| W:N4 (b) | −5.08 | |
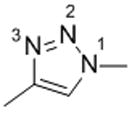
|
Compound 11 | |
| W:N2 (c) | −5.75 | |
| W:N3 (c) | −5.95 |
Schematic representation of the molecule models.
Interaction Energy between molecule and water is in kcal/mol.
Complexes are designated as W:X(m) where W stands for “water” molecule, “X” for the heteroatoms and “m” for molecule model.
