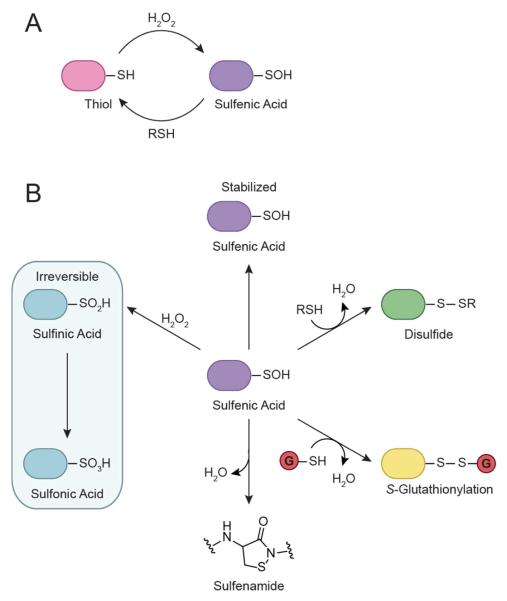
Figure 2.
Oxidative modification of cysteine residues by hydrogen peroxide (H2O2). (A) The initial reaction product of a thiolate with H2O2 yields sulfenic acid (RSOH). This modification, also known as sulfenylation, is reversible and can be directly reduced back to the thiol form or indirectly through disulfide bond formation. (B) Sulfenic acids can be stabilized by the protein microenvironment and/or undergo subsequent modification. For example, they can condense with a second cysteine in the same or different protein to generate a disulfide bond. Alternatively, reaction with the low molecular weight thiol glutathione (GSH, red circle) affords a mixed disulfide through a process known as S-glutathionylation. In a few proteins, such as PTP1B, nucleophilic attack of a backbone amide on RSOH results in sulfenamide formation. Sulfenyl groups can also oxidize further to the sulfinic (RSO2H) and/or sulfonic (RSO3H) acid form under conditions of high oxidative stress.