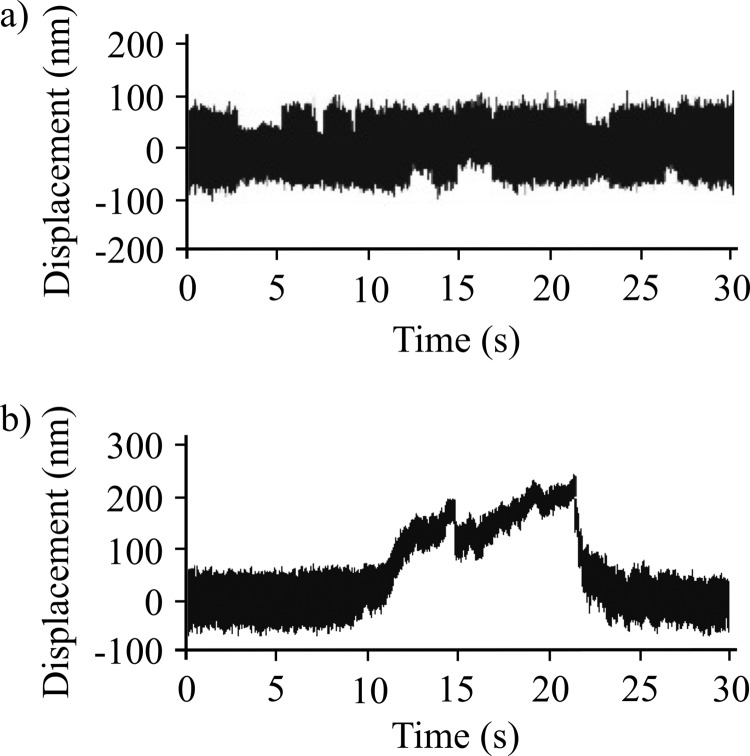
FIGURE 7.
Optical trap analysis of NMIIB nonmuscle filament-actin interactions. a, bead position record of full-length NMIIB-actin interaction. In this case the myosin molecules were deposited on nitrocellulose surface in high salt (300 mm KCl AB buffer) buffer, and as a result single, two-headed myosin molecules are probed. No signs of processive runs were observed. b, bead position record of a NMIIB filament-actin interaction. Prior to sample deposition the ionic strength of the buffer was lowered to 150 mm, and as a result the myosin molecules formed filaments. The fluorescent GFP-RLC-labeled NMIIB filaments were clearly visible on top of the pedestals, and during the measurements these pedestals were targeted. The NMIIB filaments show robust motility.